Figure 3.
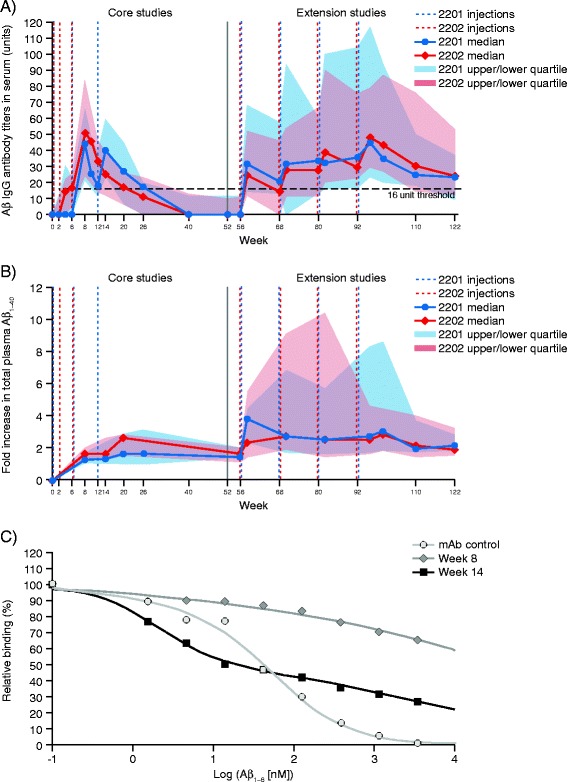
Amyloid-β-specific antibody response. (A) Median and interquartile range (IQR) Q1–Q3 Aβ-IgG titers (units) in serum by study and visit (pooled safety analysis set).* (B) Median and IQR Q1–3 fold increase in Aβ1–40 in plasma,† by study and visit (pooled safety analysis set).* (C) Mean Aβ-IgG affinity maturation between second and third injections compared with similar N-terminal monoclonal antibody control. *Only patients who received CAD106 in the core studies and entered the extension studies are included. At each time point, only patients with a value at baseline and that time point are included. †Values below the LLOQ (LLOQ = 8.93) were set to 67 pg/ml. Binding of Aβ1–6 to the serum samples containing either a reference antibody or to samples from one representative serological responder to CAD106 from study 2201 at Week 8 (2 weeks after the second injection, Aβ-IgG titers = 48.5 U) and from the same patient at Week 14 (2 weeks after the third injection, Aβ-IgG titers = 278 U). Aβ, Amyloid-β; IgG, Immunoglobulin G; IQR, Interquartile range; LLOQ, Lower limit of quantification; mAb, Monoclonal antibody.
