Figure 5. PI3-kinase can interact with B-TPM of CagA during co-culture.
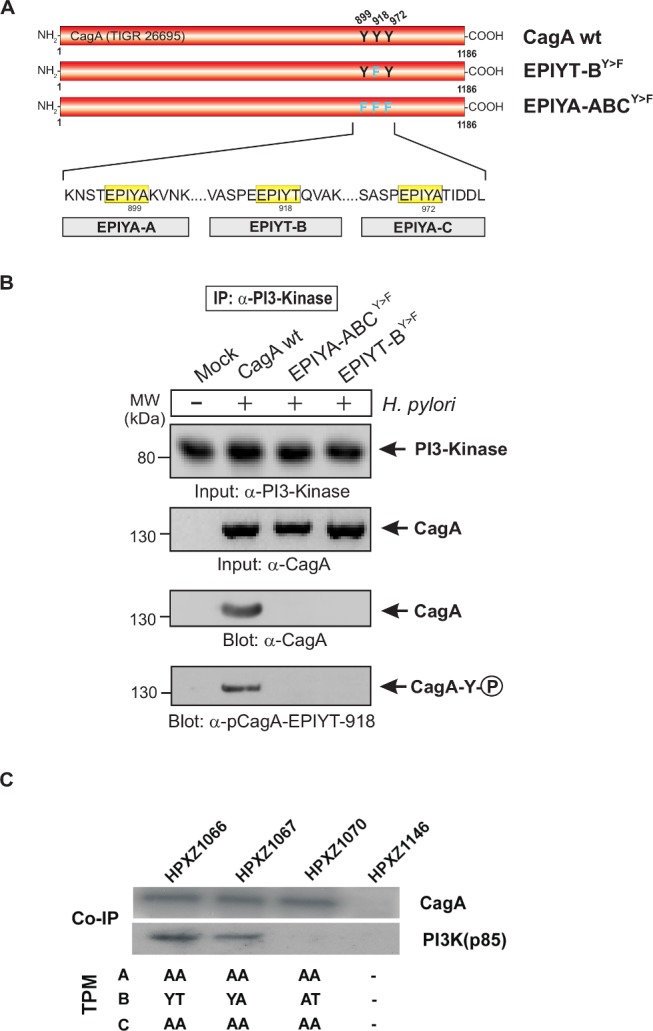
Panel A: Site-directed mutagenesis of CagA TPM-motifs A, B and C was performed to generate the indicated phospho-resistant variants. Tyrosine residues in adjacent TPM-motifs were replaced by phenylalanines. The resulting single and triple mutants were named as indicated and complemented into the H. pylori ΔcagA mutant. Panel B: AGS cells were co-cultured with the various CagA-expressing H. pylori strains for 6 h as indicated. Cell extracts were harvested and subjected to reverse immunoprecipitation (IP) using α-PI3-kinase antibodies. All samples contained similar amounts of PI3-kinase in the input control. CagA presence and phosphorylation at the EPIYT-site in the IPs was examined using phospho-specific α-pCagA-EPIYT-918 and α-CagA antibodies (arrows). Only the lane with H. pylori expressing CagA wt revealed a signal for CagA and phosphorylation at EPIYT-918 in the IP, indicating that phosphorylated EPIYT-B is necessary for the interaction with PI3-kinase. Panel C: After 24 h co-culture of AGS cells with the isogenic H. pylori strains containing the engineered CagA molecules, whole cell lysates were subjected to immunoprecipitation with an anti-CagA antibody. The anti-CagA immunoprecipitates (IP) were separated on SDS-PAGE, followed by western blot with anti-PI3-kinase (p85), which indicated that the engineered CagA B-EPIYA and CagA B-EPIYT molecules have different affinity to the PI3-kinase protein in AGS cells.
