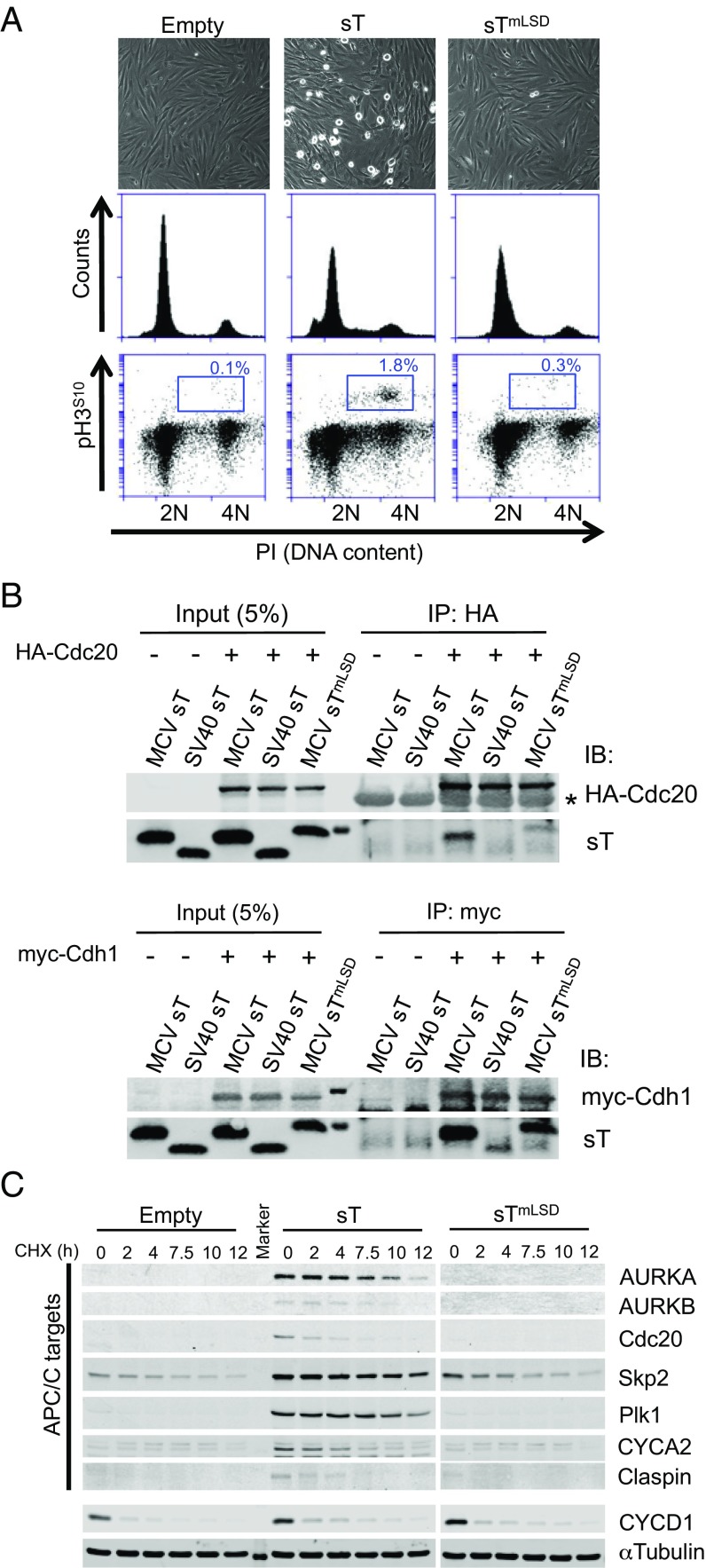
Fig. 1.
MCV sT promotes mitosis by targeting APC/C E3-ubiquitin ligases. (A) MCV sT induces cellular mitogenesis. BJ-T cells stably transduced with MCV sT have increased mitotic rounding and a 6–18-fold increase in pH3S10+ mitotic cells compared with empty vector or sTmLSD transduced cells. (B) MCV sT interacts with APC/C E3 ligase substrate recognition subunit Cdc20 and Cdh1 proteins. HA-tagged Cdc20 or myc-tagged Cdh1 expression plasmids were cotransfected with MCV sT, MCV sTmLSD, or SV40 sT expression plasmids into 293 cells and immunoprecipitated 48 h later with anti-HA or anti-myc antibodies, followed by immunoblotting using mixed anti-MCV sT (CM8E6) and anti-SV40 sT (PAb419) antibodies. Cdc20 interaction with MCV sT was nearly eliminated in the sTmLSD mutant protein, whereas partial interaction was retained between MCV sTmLSD and myc-Cdh1 proteins. Weak interaction between SV40 sT and myc-Cdh1 only was detected. Asterisk indicates IgG heavy chain. (C) APC/C target proteins (AURKA/B, Cdc20, Skp2, Plk1, CYCA2, and claspin) are stabilized by MCV sT expression. BJ-T cells were treated with CHX (100 µg/mL) to inhibit new protein synthesis and harvested at the indicated time points. The half-lives of proteins regulated by APC/C are extended by expression of MCV sT but not empty vector or MCV sTmLSD controls. CYCD1 is not directly regulated by Cdh1, and its half-life was unchanged by MCV sT expression. A representative α-tubulin loading control is shown. Representative results are shown from three independent experiments.