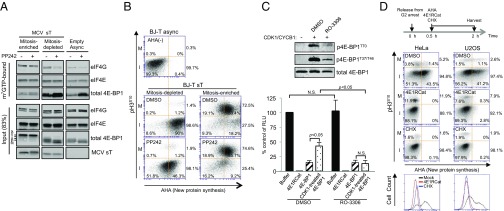
Fig. 5.
eIF4F formation on the m7GTP cap and direct measurement of cap-dependent protein translation during mitosis (M) and interphase (I). (A) eIF4F formation on the m7GTP cap is PP242-independent for mitosis-enriched cells but PP242-sensitive for mitosis-depleted cells. BJ-T cells expressing MCV sT were harvested by mitotic shake-off to enrich for mitotic (nonadherent) and nonmitotic (adherent) populations and compared with asynchronous empty vector BJ-T cells without shake-off. Lysates were bound to m7GTP-resin, precipitated, and immunoblotted. Mitosis-enriched cell eIF4G binding, as well as 4E-BP1 binding, to the eIF4E/cap complex was unaffected by mTOR inhibition. For mitosis-depleted cells, eIF4G binding was reduced and 4E-BP1 binding was increased by mTOR inhibition. Representative results from two independent experiments are shown. (B) Nascent protein synthesis during mitosis is resistant to mTOR inhibition in BJ-T cells. BJ-T cells stably expressing sT were labeled with AHA for 45 min in methionine-depleted media, separated by mitotic shake-off as in A, reacted with Click-iT Alexa Fluor 488 alkyne after permeabilization, and new protein synthesis was measured by flow cytometry. Relative mitotic protein synthesis was determined by dividing percentage of pH3S10+-AHA+ cells by percentage of total pH3S10+ cells. Likewise, interphase protein synthesis was determined by dividing percentage of pH3S10–-AHA+ cells by percentage of total pH3S10– cells. Approximately 91% of pH3S10– adherent interphase cells showed AHA incorporation that was sensitive to mTOR inhibition. Only 74% of pH3S10+-positive mitotic cells were positive for AHA uptake, but this new protein synthesis was resistant to PP242 treatment. Baseline fluorescence was determined in asynchronous BJ-T cells without AHA incubation. I, interphase pH3S10– cells; M, mitotic pH3S10+ cells. (C) In vitro capped mRNA translation is inhibited by 4E1RCat and activated by CDK1/CYCB1. Capped and polyadenylated luciferase mRNA was generated in a T7 polymerase reaction and used to produce luciferase protein in rabbit reticulocyte lysate. The addition of 4E1RCat abolished luciferase translation, whereas addition of GST–4E-BP1 reduced translation to 15% of buffer control (averages for three independent experiments with SEM shown). When GST–4E-BP1 was phosphorylated by CDK1/CYCB1 in kinase reaction buffer, translation increased to 43% of buffer control. This effect was eliminated by RO-3306 pretreatment. Insert shows GST–4E-BP1 phosphorylation immunoblot. (D) Mitotic translation is primarily cap-dependent for HeLa and U2OS. HeLa or U2OS cells were synchronized for 24 h at the G2/M boundary with RO-3306 (10 µM), released by washing, and incubated with 25 µM of AHA for 90 min in methionine-depleted media, and then harvested 2 h after release. Harvested cells were permeabilized and reacted with Alexa Fluor 488 alkyne to measure AHA incorporation into protein. DMSO vehicle control, CHX (100 µg/mL), or 4E1RCat (50 µM) were added together with AHA 30 min after release. Vertical bar represents maximum AHA incorporation after CHX translation inhibition. Fewer mitotic (26%) than interphase (42%) HeLa cells were positive for new protein synthesis, but all cells were sensitive to 4E1RCat inhibition of cap-dependent translation. For U2OS, cell numbers positive for total mitotic and interphase translation were identical (42%), and cap-dependent translation represented 73% and 85% of mitotic and interphase translation, respectively. Bottom panel shows 4N-gated AHA positivity for treated cells, which shows that 4E1RCat inhibition (cap-dependent) is similar to CHX (total) translation inhibition. Representative results are shown for one of three independent experiments.