Abstract
Stress is now recognized as a universal premorbid factor associated with many risk factors of various chronic diseases. Acute stress may induce an individual’s adaptive response to environmental demands. However, chronic, excessive stress causes cumulative negative impacts on health outcomes through “allostatic load”. Thus, monitoring the quantified levels of long-term stress mediators would provide a timely opportunity for prevention or earlier intervention of stressrelated chronic illnesses. Although either acute or chronic stress could be quantified through measurement of changes in physiological parameters such as heart rate, blood pressure, and levels of various metabolic hormones, it is still elusive to interpret whether the changes in circulating levels of stress mediators such as cortisol can reflect the acute, chronic, or diurnal variations. Both serum and salivary cortisol levels reveal acute changes at a single point in time, but the overall long-term systemic cortisol exposure is difficult to evaluate due to circadian variations and its protein-binding capacity. Scalp hair has a fairy predictable growth rate of approximately 1 cm/month, and the most 1 cm segment approximates the last month’s cortisol production as the mean value. The analysis of cortisol in hair is a highly promising technique for the retrospective assessment of chronic stress. [BMB Reports 2015; 48(4): 209-216]
Keywords: Allostasis, Cortisol, Hair, Mass spectrometry, Metabolomics, Stress
INTRODUCTION
Stress can lead to both physical and psychological health issues. Some stress can be beneficial at times by producing a boost that provides the drive and energy to help people get through situations like exams or work deadlines. However, an extreme amount of stress can lead to negative consequences and adversely affect the immune, cardiovascular, neuroendocrine, and central nervous systems (1). In particular, chronic stress can have a serious impact due to sustained high levels of the chemicals released in the “fight or flight” response, which involves endocrine system releasing glucocorticoids (2, 3).
Cortisol, which is synthesized from cholesterol, is the main glucocorticoid in the zona fasciculate of human adrenal cortex. Its secretion in response to biochemical stress contributes to the well-characterized suppression of the hypothalamic-pituitary-adrenal (HPA) axis on health and cognition events (4-6). Since the vast majority of cortisol actions rely on binding to cytosolic receptors, only a small fraction of unbound, free cortisol is revealed to be biologically active. It comes out of the mitochondrion, migrates out of the cell into the extracellular space and into the bloodstream. Due to its low molecular weight and lipophilic nature, unbound cortisol enters the cells through passive diffusion, which makes it feasible to measure the free cortisol in many body fluids (7).
In general, cortisol levels in blood increase during the early morning (highest at about 8 a.m.) and decrease slightly in the evening and during the early phase of sleep (8). The timing of blood sampling is therefore very important. While its assessment in sweat or tears is only of theoretical importance and urinary cortisol of decreasing interest, salivary cortisol may have some advantages over the assessment of cortisol in blood (9, 10). Since the hormone levels in biological fluids fluctuate on a daily basis, cortisol extracted from the hair fiber has been investigated (11-13). This review discusses on the methods involved in mass spectrometry-based metabolomic studies for identification of biomarkers in chronic stress, which is more focused on hair cortisol. Comparative statistical analyses of crucial aspects are also included to facilitate the understanding of recent advances in the metabolic platform on mining biomarkers.
STRESS AND THE ADRENAL GLAND
The two adrenal glands are located on top of the kidneys, and these glands produce hormones in response to stress. Each adrenal gland consists of a central area, called the medulla, and an outer area of the cortex (Fig. 1). In case of the apparent threat, the hypothalamus sends direct signals via the sympathetic nervous system to the adrenal glands, causing them to release a catecholamine and epinephrine (same as adrenaline). It leads to an urgent action by stimulating faster breathing and heart rates. The adrenal medullar also secrete another catecholamine, norephinephrine, which works with epinephrine to stimulate liver cells to release glucose to make more fuel available for cellular respiration. These hormones have short-term effects as the nerve impulses are sent from the hypothalamus. Due to the short half-life of blood catecholamine, meticulous care must be taken to obtain blood samples consistently vis-à-vis the stress immersion experience (14).
Fig. 1. The differential stress hormones secreted by adrenal cortex and medullar. Catecholamines cause general physiological changes that prepare the body for physical activity (fight-or-flight response) in the shortterm response. Some typical effects include increases in heart rate, blood pressure, and blood glucose levels, and other general reactions of the sympathetic nervous system. Corticoids are involved in a wide range of physiological processes including chronic stress response, immune response, and regulations of inflammation, carbohydrate metabolism, protein catabolism, blood electrolyte levels, and behavior. Data are taken from “https://www.studyblue.com/notes/note/n/07-adrenalglands/deck/1109539”.
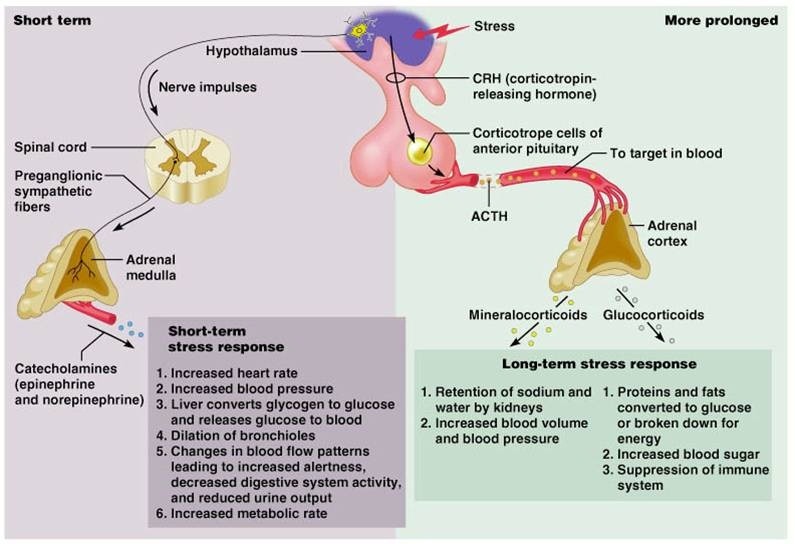
Finding a “gold standard” biomarker for chronic stress has been proven to be challenging, given its complex etiology and highly individual manifestations, while the biomarkers of acute stress have been well-defined and are primarily used to assess the release of catecholamine. Hormones secreted by the adrenal cortex provide a slower, longer-acting (chronic) response to the stress. In this event, the hypothalamus secretes a releasing hormone which causes the anterior pituitary to secrete an adrenal-stimulating hormone, adrenocorticotrophic hormone (ACTH); and this signals the cells in the adrenal cortex to produce and secrete corticosteroids. Among them, mineralocorticoids, like aldosterone, can regulate water and sodium re-absorption in the kidneys. It also regulates the active secretion of potassium in the principal cells of the cortical collecting tubule and protons via proton ATPases in the luminal membrane of the intercalated cells of the collecting tubule, which results in an increase of blood pressure and blood volume. Glucocorticoids promote fat and protein breakdown and glucose synthesis. Cortisol is the major glucocorticoids, and it regulates or supports a variety of important cardiovascular, metabolic, immunologic, and homeostatic functions (1).
ALLOSTASIS AND ALLOSTATIC LOAD
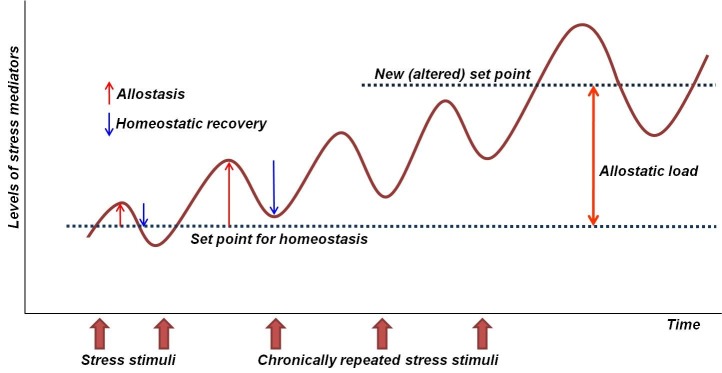
The term, stress, was originally adopted from engineering (measure of the internal forces induced by deformation of a body), but it is now referred to ‘threats or anticipation of threats to an organism’s homeostasis’ (15). Thus stress events could be understood as any stimuli that cause alterations in homeostasis for adaptation to the environment. These changes in homeostasis are referred to as ‘allostasis’, which can be exemplified by increased heart rate or blood pressure and enhanced systemic metabolism. In general, allostasis can be adaptive or maladaptive depending on its degree or contextual relevance; mediators of allostasis, such as metabolic hormones, could contribute to healthy adaptation and pathophysiology (16). The concept of ‘allostatic load’ indicates an altered, ‘new set point’ of homeostasis, resulting from cumulative effects of allostatic responses which are chronic, excessive, or poorly regulated (15, 16). For example, the increased serum glucose level is responsible for a single acute stressful event, which can be called as ‘allostatic response’. Also, diabetes (insulin resistance) resulting from repetitive chronic stress can be understood as an ‘allostatic overload’, in which the baseline fasting glucose level has been newly set to a higher level than before (Fig. 2). Therefore, the biomarkers of allostatic load, if available, could be used for measuring and predicting the cumulative biological risks of impending illnesses, and they are ideal indicators for detecting the ‘pre-phase’ of illnesses (6, 17, 18).
Fig. 2. Stress, allostasis, and allostatic load. Stress is any stimulus inducing either adaptive or maladaptive allostasis (changes in homeostasis) of stress mediators, which constitute the autonomic nervous system (blood pressure, catecholamines), metabolic hormones (cortisol, insulin), and pro- & anti-inflammatory cytokines. If stress stimuli are excessive and repetitive, recovery to the original homeostatic levels may be incomplete (indicated by the second blue arrow). As a result, chronic stress can make a body system to anticipate, as if such a new (stressful) environment would persist, demanding a newly defined set point for future adaptation. Thus, the difference between the new and old set points can be understood as a ‘cumulative burden of adaptation to stress’- i.e., allostatic load. Examples of allostatic load may be found in the primary mediators (hypercortisolemia, increased inflammatory cytokines), secondary outcomes (elevated blood pressure, overweight, insulin resistance), or tertiary outcomes (hypertension, diabetes, obesity, coronary heart disease, neurodegenerative disorders). For more details, see reference # 6.
Mediators of allostasis constitute the autonomic nervous system (catecholamines), metabolic hormones, and various cytokines (15, 16). Among these, cortisol is the one that has been paid great attention based on the concept of ‘glucocorticoid cascade hypothesis’ (19). This hypothesis explains how and why the cortisol actions could be related to pathophysiology upon overload of stress. Psychological and physical stresses increase the circulating cortisol levels. In the acute state, increased cortisol induces adaptive responses via enhancing catabolic processes to supply more energy to the body. In general, increased cortisol levels return to the basal levels by feedback inhibition mechanisms through the hypothalamus, prefrontal cortex, and most importantly, hippocampus (20). When stressful stimuli are repeated chronically, circulating cortisol is maintained at higher levels over a prolonged period. Chronically elevated cortisol levels now causes damages on hippocampal and cortical neurons (19), which are the main regions where the feedback inhibition starts. As a result, even when stress stimuli disappear, cortisol levels could be maintained at higher levels beyond the physiologically normal range due to a vicious cycle caused by the already damaged feedback mechanism.
Consequently, a great number of studies have explored the potential of utilizing cortisol as a biomarker for various chronic illnesses or their pre-disease states (21, 22). Currently, it seems that such role of cortisol, as one of biomarkers for the allostatic load, may be largely accepted (6). However, there are still controversial issues regarding the sampling and measurement methods of these hormones that are fluctuated by environmental contexts as well as by the circadian cycle. However, the most important issues might be the possibility that the level measured once at a current point could reflect the past history of an individual’s stress load, just as HbA1c indicating the degree of ‘glucose load’ for the past 3 months. The level of cortisol in scalp hair is now considered as a promising biomarker for assessing the averaged level of one’s past stress burden during a given period, and it will be further discussed in this paper. Its utility has been demonstrated across diverse clinical settings from neonates to old age, indicated by associations of hair cortisol levels with babies’ stress in the neonatal intensive care unit (23), children’s stress at school entry (24), and various metabolic and neuropsychiatric disorders including acute myocardial infarction (25), heart failure (26), metabolic syndrome (27), and post-traumatic stress disorder (28).
ADVANCES IN CORTISOL DETECTION IN METABOLITE PROFILING
Because more than 90% of circulating cortisol in human serum is protein-bound, changes in the binding proteins can alter the levels of serum total cortisol without influencing the free concentrations of cortisol. Total cortisol could be misleadingly lower than anticipated, resulting in the incorrect interpretation that adrenal function is impaired. This is important because the current standards for defining normal adrenal functions are based on healthy people who have normal levels of binding proteins. Measuring serum free cortisol levels in critical illnesses may help to prevent the unnecessary use of glucocorticoid therapy (21). Although the free cortisol hypothesis has been widely used, it has also been suggested that cortisol binding globulin (CBG)-bound cortisol may have physiological effects on target tissues (22).
There is a high correlation between salivary cortisol levels and unbound cortisol in plasma and serum, which remains high during the circadian cycle and under different dynamic test such as ACTH stimulation (22, 29). Since free cortisol represents the biologically active hormone fraction, salivary cortisol measures have early been considered as a better method than serum cortisol for the evaluation of adrenocortical function (9). Recently, the late-night salivary cortisol has been showing a superior diagnostic performance as the primary biochemical diagnostic test for Cushing’s syndrome (10). Cortisol in biological fluids has been extensively evaluated with cortisone, which may reveal the activity of 11β-hydroxysteroid dehydrogenase (11β-HSD) by mass spectrometry-based metabolite profiling techniques (30-32). These analytical methods have shown the acceptable analytical sensitivity and the selectivity in trace amounts of biological samples such as urine and serum. However, the methods have still hampered from incorrect physiological levels of adrenal steroids, including cortisol, due to the sampling problems with circadian variation. In general, acute cortisol levels fluctuate markedly depending on many physiological factors including circadian rhythmicity, and it may provide a rather poor reflection of normal, chronic cortisol secretion (12, 13).
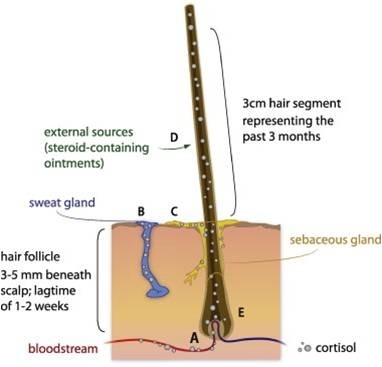
In contrast to the biological fluids, hair can provide biological information of long-term exposure because its growth rate is about 1 cm/month (Fig. 3). The hormones are mainly delivered from the blood circulation to the capillaries of the dermal papilla, which is located in the hair follicles (33). This phenomenon enables retrospective examination of cortisol production at the times when a stressor is most salient, without needing to take a sample right at that time. Alternatively, it can provide a baseline cortisol assessment for a time period during which the stress has not yet occurred. In addition, its non-invasive nature in sampling and easy storage at room temperature has been highlighted as advantage in clinical applications (34-36). In the hair analysis, steroid hormones are extracted by solubilization or digestion of the hair matrix with alkaline hydrolysis for androgen and estrogens (37, 38). Also, the acid hydrolysis or methanol extraction can enable an analysis of corticoids in hair due to its insufficient chemical stability (11). When 62 biologically active steroids were analyzed by an optimized extraction technique, and only 20 hair steroids, including cortisol and cortisone, were quantitatively detectable (11).
Fig. 3. A proposed mechanism of cortisol incorporation into hair and retrospective reflection of its chronic secretion. Cortisol may be incorporated into hair via passive diffusion from (A) blood capillary, (B) sweat, and (C) sebum, as well as from (D) external sources. Data are taken from reference #12.
STRESS-MEDIATED HAIR CORTISOL IN ENDOCRINE SYSTEM
Because more than 70% of diseases are believed to be stress-related, prediction of chronic stress is an important step in reducing the incidence of chronic illnesses. Hair cortisol may provide an objective measurement of stress over time rather than just ‘a day in life’. The symptoms of the metabolic syndrome resemble those of Cushing’s syndrome, a disease that is characterized by hypercortisolism. One of the questions raised is whether chronically elevated cortisol concentrations play a role in the development of obesity and the metabolic syndromes. The increased hair cortisol levels are also associated with children’s obesity caused by long-term activation of HPA-axis (39), which is in accordance with the urinary levels of cortisol in obese children (40). The risk of cardiovascular disease (CVD) is associated with the increase in hair cortisol levels by 2.7-times, which was similar to the risk associated with hypertension or obesity. This suggests that high cortisol levels in a long-term might be an important risk factor for CVD (41).
Mitotane, an anti-neoplastic agent for adrenocortical cancer (ACC), increases CBG and induces CYP3A4 activity, which leads to high doses of hydrocortisone; however, there has been no efficient biomarker to evaluate this therapy. Hair cortisol levels were higher in ACC patients compared to healthy individuals, and they were associated with body mass index (42). However, there was no correlation between hair cortisol levels and hydrocortisone doses. As a measure of long-term cortisol exposure, the hair cortisol analysis in patients receiving glucocorticoid replacement therapy may be a useful tool. Also, the hair cortisol content is correlated with hydrocortisone dose in the patients with adrenal insufficiency, who had significantly higher subjective stress scores than the control subjects (43).
METABOLOMIC IDENTIFICATIONS IN CHRONIC STRESS
The main benefit of metabolomic strategy is the high likelihood of identifying unpredicted changes in metabolic profiles cued by abnormal conditions (44). In particular, the metabolomic information may offer novel diagnostic indicators and therapeutic targets in clinical applications (45), as well as in mechanistic studies heading to elucidate metabolic modules that can regulate dysfunctional processes in disease statuses (46).
Among dysfunctional heath conditions, chronic diseases are mostly composed of subtle and long-term dysregulations of cellular and physiological function that is often not measurable even during disease onset (47). The stress-inducing abnormal status is chronically developed with unpredictable combination of various types of etiological factors. Therefore, the molecular characterization as well as clinical definition may not be clear at specific disease course and it may not be homologous across individuals (48). The diversity of pathological traits induced by chronic stress make it even harder to properly diagnose the abnormality and classify the progress stage. Most of the studies are mainly focused on targeted single molecule and one-point interaction, which may be insufficient to reflect the dynamics and systematic effects of chronic stress (49). In this context, metabolite profiling can be an effective tool for biomarker discovery and understanding of molecular mechanism, particularly for the cases where a diagnostic/prognostic indicator is unknown and molecular mechanism remains veiled. Recently, a chronic unpredictable mild stress in animal model showed aberrant profiling of amino acids that were grouped into neurotransmitters and branched-chain amino acids (50). The metabolic signatures under acute and chronic stress in rat model and the biochemical cues are also closely associated with behavior and physiological readouts (49).
DATA MINING IN METABOLOMICS
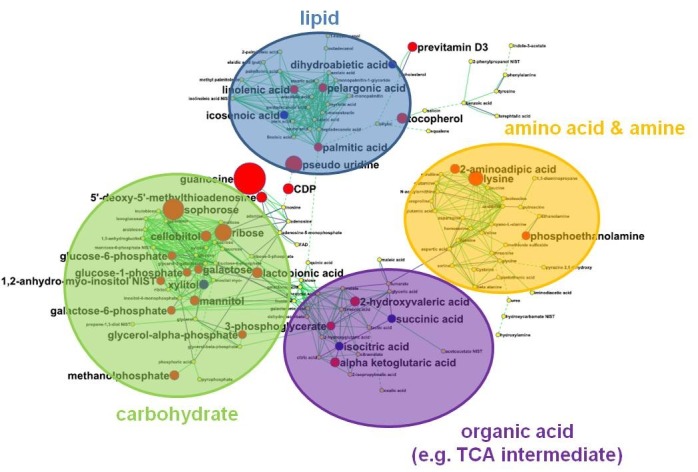
To capture disease-specific metabolic signatures, many studies, including the case studies described above, primarily explored on the multiple molecular constituents (metabolites), robustly managed variability and heterogeneity of disease progress, and defined the individuality of metabolic contents, which could lead to well-defined diagnoses and prognoses. In this context, multivariate statistics such as principal component analysis (PCA) or partial least squared algorithm is a useful data mining tool that can distinguish different groups with minimized loss of information (51). By this nature, unidentified variation source caused by transient (time-course) or individual specificity can be handled within reconstructed statistical model, which in turn offers more robust candidates for diagnostic/prognostic information in clinical cases (Fig. 4).
Fig. 4. Reconstructed metabolic network for systematic screening of therapeutic target point. The re-modeled metabolic structure is composed of metabolites, which consists of node (e.g. metabolite) and edge (e.g. correlation, structural similarity). The model may be extended to protein information via reaction pair that has already been built in the assembly, which could unravel “hidden” or veiled metabolic modulation, particularly in chronic disease. In this example, node color and size indicate significant difference and fold change compared to control (e.g. disease vs control), while edge presents two layers of information on structural similarity and reaction likelihood, which leads to automatic rearrangement as seen in the figure indicating proximity of biochemical module.
In many studies, metabolomic approach has been limitedly applied to biomarker discovery with either single target or group of metabolites. Yet, an ideal exploratory use of the technology would not only be able to capture the metabolite changes associated with pathology, but it would also elucidate the molecular mechanism responsible for the dysfunction and propose the logical candidate for regulating the abnormality. Pathway (enrichment) analysis, a systematic approach for data mining from metabolite sets, can provide metabolic pathway-wise information rather than the readouts of individual or few metabolite contents (52). This bioinformatics tool, which is rooted in gene ontology enrichment analysis, assigns groups of metabolites at the level of metabolic pathway. The analysis provides P value that presents significant difference at the levels of metabolic pathways and information on pathway centrality (pathway importance), which demonstrates how much connectivity is formed surrounding the metabolites of interest in the pathway (53). The output can be further investigated for mechanistic understanding of disease, and it can be applied for targeting and controlling the functional points of disease process. However, although metabolic signature can be identified at the level of either single molecule, groups of metabolites, or metabolic pathway, metabolite assessment cannot by itself provide a complete understanding of disease process. To gain comprehensive causality and mechanism in given experimental designs, integrative analysis should be accompanied by other molecular information such as mRNA expression levels and protein abundances (54). Since metabolomics joins systems with biology, mRNA has been the first partner for integrative analysis mainly due to the well-establishment of technical platform and advancements in statistical analysis. However, the expression level of mRNA showed a low correlation with enzyme activity and protein expression levels, which is a direct molecular companion with metabolites, the substrate, and products. Thus protein information is more suitable counterpart of metabolite readout (55).
Most importantly, in order to have molecular information (mRNA, protein, and metabolite) located at the cornerstone of clinical application, various types of molecular levels should be linked to physiological traits (e.g. clinical parameter) in a statistically sound manner. This has been mainly done by ‘qualitative’ assessment in which the final readout of molecular behaviors is linked to logical phenotypes of disease. For example, aberrant glycolytic activity can be associated with tumor metabolism, in which glycolytic intermediates and key enzymes are overexpressed in tumor cells. But in the case of chronic disease, we may not be able to detect “the standardized feature” like cancer metabolism case. Alternatively, we can track and correlate the quantitative traits of pathology with molecular dynamics, which can bear the variability caused by individuality and differential disease progress. One relevant statistical approach is canonical correlation analysis (CCA), which has been often applied in psychological, climate, and ecological studies to enumerate the correlations between two distinct data sets measured on the identical experimental units (56-59). This statistics can analyze significant relations between two different dataset (e.g. metabolite contents and the associated clinical parameters). It is similar as PCA, in the way that CCA seeks for linear combinations of the variables to reduce the dimension of the data sets; but at the same time, it explores to maximize the correlation between the two variates (58).
CONCLUDING REMARKS
Because single cortisol assessments are strongly affected by the acute context of the measurement situation (time of day, day of the week, and other circumstances such as distress for blood sampling), the assessment of long-term cortisol secretion from the biological fluids, such as blood, urine, and saliva, is highly labor-intensive and rather difficult to be implemented in physical and psychological studies. Hair growth patterns also vary across different regions of the scalp, and the precise mechanisms of substances incorporating into hair are still incompletely understood. But a growing observation supports the notion that hair cortisol analysis provides a valid and reliable reflection of long-term cortisol secretion (12, 13, 27). We mainly discussed about the advantages of hair analysis as an index of chronic cortisol response. Due to their association with various aspects of biochemical stress in adrenal steroids and other steroid metabolites, advantages of hair analysis may not only be restricted to cortisol (35). Further research on establishing the reference values of hair cortisol may be needed to enhance the current knowledge on particular aspects of chronic stress and other metabolic changes in many endocrine diseases.
References
- 1.Anderson NB. Levels of analysis in health sciences: A framework for integrating sociobehavioral and biomedical research. Ann N Y Acad Sci. (1998);840:563–576. doi: 10.1111/j.1749-6632.1998.tb09595.x. [DOI] [PubMed] [Google Scholar]
- 2.Edwards C. Sixty tears after hench-Corticosteroids and chronic inflammatory disease. J Clin Endocrinol Metab. (2012);97:1443–1451. doi: 10.1210/jc.2011-2879. [DOI] [PubMed] [Google Scholar]
- 3.Whirledge S, Cidlowski A. A role for glucocorticoids in stress-impaired reproduction: Beyond the hypothalamus and pituitary. Endocrinology. (2013);154:4450–4468. doi: 10.1210/en.2013-1652. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Oswald LM, Zandi P, Nestadt G, Potash JB, Kalaydjian AE, Wand GS. Relationship between cortisol response to stress and personality. Neuropsychopharmacology. (2006);31:1583–1591. doi: 10.1038/sj.npp.1301012. [DOI] [PubMed] [Google Scholar]
- 5.Staab CA, Maser E. 11β-Hydroxysteroid dehydrogenase type 1 is an important regulator at the interface of obesity and inflammation. J Steroid Biochem Mol Biol. (2010);119:56–72. doi: 10.1016/j.jsbmb.2009.12.013. [DOI] [PubMed] [Google Scholar]
- 6.Juster RP, McEwen BS, Lupien SJ. Allostatic load biomarkers of chronic stress and impact on health and cognition. Neurosci Biobehav Rev. (2010);35:2–16. doi: 10.1016/j.neubiorev.2009.10.002. [DOI] [PubMed] [Google Scholar]
- 7.Miller WL. Steroidogenic enzymes. Endocr Rev. (2008);13:1–18. doi: 10.1159/000134751. [DOI] [PubMed] [Google Scholar]
- 8.Weitzman ED, Fukushima D, Nogeire C, Roffwarg H, Gallagher TF, Hellman L. Twenty-four hour pattern of the episodic secretion of cortisol in normal subjects. J Clin Endocrinol Metab. (1971);33:14–22. doi: 10.1210/jcem-33-1-14. [DOI] [PubMed] [Google Scholar]
- 9.Hellhammer DH, Wüst S, Kudielka BM. Salivary cortisol as a biomarker in stress research. Psychoneuroendocrinology. (2009);34:163–171. doi: 10.1016/j.psyneuen.2008.10.026. [DOI] [PubMed] [Google Scholar]
- 10.Elias PCL, Martinez EZ, Barone BFC, Mermejo LM, Castro M, Moreira AC. Late-night salivary cortisol has a better performance than urinary free cortisol in the diagnosis of Cushing’s syndrome. J Clin Endocrinol Metab. (2014);99:2045–2051. doi: 10.1210/jc.2013-4262. [DOI] [PubMed] [Google Scholar]
- 11.Jung HJ, Kim SJ, Lee WY, Chung BC, Choi MH. Gas chromatography/mass spectrometry based hair steroid profiling may reveal pathogenesis in hair follicles of the scalp. Rapid Commun Mass Spectrom. (2011);25:1184–1192. doi: 10.1002/rcm.4975. [DOI] [PubMed] [Google Scholar]
- 12.Stalder T, Kirschbaum C. Analysis of cortisol in hair - State of the art and future directions. Brain Behav Immun. (2012);26:1019–1029. doi: 10.1016/j.bbi.2012.02.002. [DOI] [PubMed] [Google Scholar]
- 13.Russell E, Koren G, Rieder M, Uum SV. Hair cortisol as a biological marker of chronic stress: Current status, future directions and unanswered questions. Psychoneuroendocrinology. (2012);37:589–601. doi: 10.1016/j.psyneuen.2011.09.009. [DOI] [PubMed] [Google Scholar]
- 14.Dimsdale JE, Moss J. Short-term catecholamine response to psychological stress. Psychosom Med. (1980);42:493–497. doi: 10.1097/00006842-198009000-00003. [DOI] [PubMed] [Google Scholar]
- 15.Karatsoreos IN, McEwen BS. Psychobiological allostasis: resistance, resilience and vulnerability. Tr Cognit Sci. (2011);15:576–584. doi: 10.1016/j.tics.2011.10.005. [DOI] [PubMed] [Google Scholar]
- 16.McEwen BS. Protective and damaging effects of stress mediators. N Engl J Med. (1998);338:171–179. doi: 10.1056/NEJM199801153380307. [DOI] [PubMed] [Google Scholar]
- 17.Seeman TE, McEwen BS, Rowe JW, Singer BH. Allostatic load as a marker of cumulative biological risk: MacArthur studies of successful aging. Proc Natl Acad Sci U S A. (2001);98:4770–4775. doi: 10.1073/pnas.081072698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Staufenbiel SM, Penninx BW, Spijker AT, Elzinga BM, van Rossum EF. Hair cortisol, stress exposure, and mental health in humans: a systematic review. Psychoneuroendocrinology. (2013);38:1220–1235. doi: 10.1016/j.psyneuen.2012.11.015. [DOI] [PubMed] [Google Scholar]
- 19.Sapolsky RM, Krey LC, McEwen BS. The Neuroendocrinology of Stress and Aging: The Glucocorticoid Cascade Hypothesis. Endocr Rev. (1986);7:284–301. doi: 10.1210/edrv-7-3-284. [DOI] [PubMed] [Google Scholar]
- 20.Mizoguchi K, Ishige A, Aburada M, Tabira T. Chronic stress attenuates glucocorticoid negative feedback: involvement of the prefrontal cortex and hippocampus. Neuroscience. (2003);119:887–897. doi: 10.1016/S0306-4522(03)00105-2. [DOI] [PubMed] [Google Scholar]
- 21.Hamrahian AH, Oseni TS, Arafah BM. Measurement of serum free cortisol in critically ill patients. N Engl J Med. (2004);350:1629–1638. doi: 10.1056/NEJMoa020266. [DOI] [PubMed] [Google Scholar]
- 22.Levine A, Zagoory-Sharon O, Feldman R, Lewis JG, Weller A. Measuring cortisol in human psychobiological studies. Physiol Behav. (2007);90:43–53. doi: 10.1016/j.physbeh.2006.08.025. [DOI] [PubMed] [Google Scholar]
- 23.Yamada J, Stevens B, de Silva N, et al. Hair cortisol as a potential biologic marker of chronic stress in hospitalized neonates. Neonatology. (2007);92:42–49. doi: 10.1159/000100085. [DOI] [PubMed] [Google Scholar]
- 24.Groeneveld MG, Vermeer HJ, Linting M, Noppe G, van Rossum EF, van IJzendoorn MH. Children's hair cortisol as a biomarker of stress at school entry. Stress. (2013);16:711–715. doi: 10.3109/10253890.2013.817553. [DOI] [PubMed] [Google Scholar]
- 25.Pereg D, Gow R, Mosseri M. Hair cortisol and the risk for acute myocardial infarction in adult men. Stress. (2011);14:73–81. doi: 10.3109/10253890.2010.511352. [DOI] [PubMed] [Google Scholar]
- 26.Pereg D, Chan J, Russell E, et al. Cortisol and testosterone in hair as biological markers of systolic heart failure. Psychoneuroendocrinology. (2013);38:2875–2882. doi: 10.1016/j.psyneuen.2013.07.015. [DOI] [PubMed] [Google Scholar]
- 27.Stalder T, Kirschbaum C, Alexander N, et al. Cortisol in hair and the metabolic syndrome. J Clin Endocrinol Metab. (2013);98:2573–2580. doi: 10.1210/jc.2013-1056. [DOI] [PubMed] [Google Scholar]
- 28.Steudte S, Kirschbaum C, Gao W, et al. Hair cortisol as a biomarker of traumatization in healthy individuals and posttraumatic stress disorder patients. Biol Psychiatry. (2013);74:639–646. doi: 10.1016/j.biopsych.2013.03.011. [DOI] [PubMed] [Google Scholar]
- 29.Vining RF, McGinley RA, Symons RG. Hormones in saliva: mode of entry and consequent implications for clinical interpretation. Clin Chem. (1983);29:1752–1756. [PubMed] [Google Scholar]
- 30.Cho HJ, Kim JD, Lee WY, Chung BC, Choi MH. Quantitative metabolic profiling of 21 endogenous corticosteroids in urine by liquid chromatography-triple quadrupole-mass spectrometry. Anal Chim Acta. (2009);632:101–108. doi: 10.1016/j.aca.2008.10.059. [DOI] [PubMed] [Google Scholar]
- 31.Moon JY, Jung HJ, Moon MH, Chung BC, Choi MH. Heat-map visualization of gas chromatographymass spectrometry based quantitative signatures on steroid metabolism. J Am Soc Mass Spectrom. (2009);20:1626–1637. doi: 10.1016/j.jasms.2009.04.020. [DOI] [PubMed] [Google Scholar]
- 32.Maier B, Vogeser M. Target analyte quantification by isotope dilution LC-MS/MS directly referring to internal standard concentrations-validation for serum cortisol measurement. Clin Chem Lab Med. (2013);51:833–837. doi: 10.1515/cclm-2012-0276. [DOI] [PubMed] [Google Scholar]
- 33.Randall VA. Androgens and human hair growth. Clin Endocrinol (Oxf) (1994);40:439–457. doi: 10.1111/j.1365-2265.1994.tb02483.x. [DOI] [PubMed] [Google Scholar]
- 34.Choi MH, Kim KR, Kim IS, Lho DS, Chung BC. Increased hair polyamine levels in patients with Alzheimer’s disease. Ann Neurol. (2001);50:128. doi: 10.1002/ana.1086. [DOI] [PubMed] [Google Scholar]
- 35.Cho SH, Choi MH, Sim WY, Lee WY, Chung BC. Metabolic alterations of DHEA and cholesterol sulphates in the hair of patients with acne measured by liquid chromatography-mass spectrometry. Exp Dermatol. (2010);19:694–696. doi: 10.1111/j.1600-0625.2010.01094.x. [DOI] [PubMed] [Google Scholar]
- 36.Choi MH, Kim SJ, Lew BL, Sim WY, Chung BC. Hair steroid profiling reveals racial differences in male pattern baldness between Korean and Caucasian populations. J Invest Dermatol. (2013);133:822–824. doi: 10.1038/jid.2012.349. [DOI] [PubMed] [Google Scholar]
- 37.Choi MH, Chung BC. GC-MS determination of steroids related to androgen biosynthesis in human hair with pentafluorophenyldimethylsilyl-trimethylsilyl derivatisation. Analyst. (1999);124:1297–1300. doi: 10.1039/a903912k. [DOI] [PubMed] [Google Scholar]
- 38.Choi MH, Kim KR, Chung BC. Determination of estrone and 17β-estradiol in human hair by gas chromatography-mass spectrometry. Analyst. (2000);125:711–714. doi: 10.1039/a909107f. [DOI] [PubMed] [Google Scholar]
- 39.Veldhorst MA, Noppe G, Jongejan MH, et al. Increased scalp hair cortisol concentrations in obese children. J Clin Endocrinol Metab. (2014);99:285–290. doi: 10.1210/jc.2013-2924. [DOI] [PubMed] [Google Scholar]
- 40.Reinehr T, Kulle A, Wolters B, et al. Relationship between 24-hour urinary free cortisol concentrations and metabolic syndrome in obese children. J Clin Endocrinol Metab. (2014);99:2391–2399. doi: 10.1210/jc.2013-4398. [DOI] [PubMed] [Google Scholar]
- 41.Manenschijn L, Schaap L, van Schoor NM, et al. High long-term cortisol levels, measured in scalp hair, are associated with a history of cardiovascular disease. J Clin Endocrinol Metab. (2013);98:2078–2083. doi: 10.1210/jc.2012-3663. [DOI] [PubMed] [Google Scholar]
- 42.Manenschijn L, Quinkler M, van Rossum EF. Hair cortisol measurement in mitotane-treated adrenocortical cancer patients. Horm Metab Res. (2014);46:299–304. doi: 10.1055/s-0034-1370961. [DOI] [PubMed] [Google Scholar]
- 43.Gow R1, Koren G, Rieder M, Van Uum S. Hair cortisol content in patients with adrenal insufficiency on hydrocortisone replacement therapy. Clin Endocrinol (Oxf) (2011);74:687–693. doi: 10.1111/j.1365-2265.2011.04001.x. [DOI] [PubMed] [Google Scholar]
- 44.Lee do Y, Kind T, Yoon YR, Fiehn O, Liu KH. Comparative evaluation of extraction methods for simultaneous mass-spectrometric analysis of complex lipids and primary metabolites from human blood plasma. Anal Bioanal Chem. (2014);406:7275–7286. doi: 10.1007/s00216-014-8124-x. [DOI] [PubMed] [Google Scholar]
- 45.Crews B, Wikoff WR, Patti GJ, et al. Variability analysis of human plasma and cerebral spinal fluid reveals statistical significance of changes in mass spectrometry-based metabolomics data. Anal Chem. (2009);81:8538–8544. doi: 10.1021/ac9014947. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Vinayavekhin N, Homan EA, Saghatelian A. Exploring disease through metabolomics. ACS Chem Biol. (2009);5:91–103. doi: 10.1021/cb900271r. [DOI] [PubMed] [Google Scholar]
- 47.Kim OY, Lee JH, Sweeney G. Metabolomic profiling as a useful tool for diagnosis and treatment of chronic disease: focus on obesity, diabetes and cardiovascular diseases. Expert Rev Cardiovasc Ther. (2013);11:61–68. doi: 10.1586/erc.12.121. [DOI] [PubMed] [Google Scholar]
- 48.Li ZY, Zheng XY, Gao XX, et al. Study of plasma metabolic profiling and biomarkers of chronic unpredictable mild stress rats based on gas chromatography/mass spectrometry. Rapid Commun Mass Spectrom. (2010);24:3539–3546. doi: 10.1002/rcm.4809. [DOI] [PubMed] [Google Scholar]
- 49.Wang X, Zhao T, Qiu Y, et al. Metabonomics approach to understanding acute and chronic stress in rat models. J Proteome Res. (2009);8:2511–2518. doi: 10.1021/pr801086k. [DOI] [PubMed] [Google Scholar]
- 50.Ni Y, Su M, Lin J, et al. Metabolic profiling reveals disorder of amino acid metabolism in four brain regions from a rat model of chronic unpredictable mild stress. FEBS Lett. (2008);582:2627–2636. doi: 10.1016/j.febslet.2008.06.040. [DOI] [PubMed] [Google Scholar]
- 51.Broadhurst DI, Kell DB. Statistical strategies for avoiding false discoveries in metabolomics and related experiments. Metabolomics. (2006);2:171–196. doi: 10.1007/s11306-006-0037-z. [DOI] [Google Scholar]
- 52.Xia J, Wishart DS. MetPA: a web-based metabolomics tool for pathway analysis and visualization. Bioinformatics. (2010);26:2342–2344. doi: 10.1093/bioinformatics/btq418. [DOI] [PubMed] [Google Scholar]
- 53.Xia J, Mandal R, Sinelnikov IV, Broadhurst D, Wishart DS. MetaboAnalyst 2.0-a comprehensive server for metabolomic data analysis. Nucleic Acids Res. (2012);40:W127–W133. doi: 10.1093/nar/gks374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Lee do Y, Park JJ, Barupal DK, Fiehn O. System response of metabolic networks in Chlamydomonas reinhardtii to total available ammonium. Mol Cell Proteomics. (2012);11:973–988. doi: 10.1074/mcp.M111.016733. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Wienkoop S, Morgenthal K, Wolschin F, Scholz M, Selbig J, Weckwerth W. Integration of metabolomic and proteomic phenotypes analysis of data covariance dissects starch and RFO metabolism from low and high temperature compensation response in arabidopsis thaliana. Mol Cell Proteomics. (2008);7:1725–1736. doi: 10.1074/mcp.M700273-MCP200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Meyer RC, Steinfath M, Lisec J, et al. The metabolic signature related to high plant growth rate in Arabidopsis thaliana. Proc Natl Acad Sci U S A. (2007);104:4759–4764. doi: 10.1073/pnas.0609709104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Jozefczuk S, Klie S, Catchpole G, et al. Metabolomic and transcriptomic stress response of Escherichia coli. Mol Syst Biol. (2010);6:364. doi: 10.1038/msb.2010.18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.González I, Déjean S, Martin PG, Gonçalves O, Besse P, Baccini A. Highlighting relationships between heterogeneous biological data through graphical displays based on regularized canonical correlation analysis. J Biol Syst. (2009);17:173–199. doi: 10.1142/S0218339009002831. [DOI] [Google Scholar]
- 59.Song IS, Lee do Y, Shin MH, et al. Pharmacogenetics meets metabolomics: discovery of tryptophan as a new endogenous oct2 substrate related to metformin disposition. PLoS One. (2012);7:e36637. doi: 10.1371/journal.pone.0036637. [DOI] [PMC free article] [PubMed] [Google Scholar]


