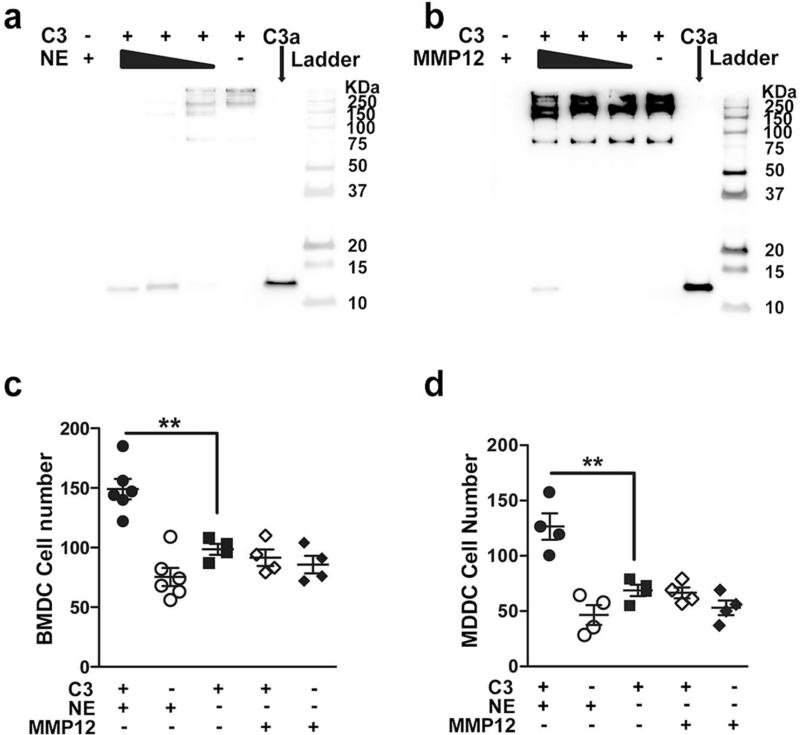
Figure 2. Neutrophil elastase (NE) and MMP12 cleave and activate complement C3.
Purified human C3 (100μg/ml) was cleaved with different concentrations of human (a) NE (10μg/ml, 1μg/ml, 0.1μg/ml) and (b) MMP12 (10μg/ml, 1μg/ml, 0.1μg/ml) for 4 hours at 37°C. Cleavage products were separated using 10% non-reducing Tricine gels, and detected by Western blot using anti-C3a antibody; purified C3a, NE and MMP12 were loaded as controls. (c) Bone marrow-derived dendritic cells (BMDCs; 5×104) and (d) myeloid-derived dendritic cells (MDDCs; 5×104) were suspended in media (RPMI-1640) and were placed on 48-well chemotaxis chambers for 1hr in the presence of intact or MMP12, NE cleaved C3 protein; control conditions included NE and MMP12. Transmigrating cells were detected in stained membranes visualized under microscope (20x) and reported as the average number of cells/field (n=4-6). **P<0.01, as determined by one-way ANOVA with Bonferroni's multiple comparison. Results are represented as mean±s.e.m, from 3 independent experiments.