Abstract
Sarcopenia, the loss of skeletal muscle mass and function that occurs with aging, is associated with increased risk for several adverse health outcomes, including frailty, disability, falls, loss of independent living, and mortality. At present, no pharmacological treatment exists that is able to definitely halt the progression of sarcopenia. Likewise, no pharmacological remedies are yet available to prevent the onset of age-related muscle wasting. In this scenario, the combination of nutritional interventions and physical exercise appears to be the most effective strategy presently available for the management of sarcopenia. The purposes of this review are to summarize the current knowledge on the role of nutrition as a countermeasure for sarcopenia, illustrate the mechanisms of action of relevant dietary agents on the aging muscle, and introduce novel nutritional strategies that may help preserve muscle mass and function into old age. Issues related to the identification of the optimal timing of nutritional interventions in the context of primary and secondary prevention are also discussed. Finally, the prospect of elaborating personalized dietary and physical exercise recommendations through the implementation of integrated, high-throughput analytic approaches is illustrated.
Keywords: aging, protein, vitamin D, creatine, omics, personalized diet, Pachinko model
Introduction
Advancing age is associated with profound changes in body composition, the most notable of which, sarcopenia, is a major determinant of frailty, disability and mortality (1). After the age of 35 years, a healthy person loses muscle mass at a rate of 1–2% per year in conjunction with a 1.5% annual decline in strength, which accelerates to approximately 3% per year after the age of 60 (2). As a result, the muscle cross-sectional area of the thigh decreases by about 40% between 20 and 60 years of age. The magnitude of decline in fat-free mass is twice as great in men than in women, and is amplified in sedentary individuals relative to physically active peers (2). Beside losing muscle mass, an average adult can expect to gain approximately 0.45 kg (1 lb) of fat yearly between the ages of 30 and 60 (3). This shift in body composition is often masked by stable body weight and can result in a condition known as sarcopenic obesity, which further increases the risk of disability, morbidity and mortality (4).
Although the decline in muscle mass and strength with age has been known for many years, the study of cellular mechanisms responsible for such a decay is still in its infancy. Nevertheless, several processes and mechanisms have been identified that are likely to contribute to the development of sarcopenia, including altered hormonal status (e.g., decline of growth hormone, estrogens and testosterone levels), increased production of pro-inflammatory cytokines, loss of α-motor neurons, muscular mitochondrial dysfunction, altered autophagy, accelerated myonuclear apoptosis, and impaired satellite cell (Sc) function (5). All these factors eventually lead to an imbalance between anabolic and catabolic processes, which results in muscle protein breakdown, loss of myocytes, insufficient Sc replenishment, and ultimately declines in muscle mass and function (5). Given the complex and intertwining relationship among these putative etiological factors and the incomplete knowledge on the cellular processes underlying the pathogenesis of sarcopenia, the quest for effective treatments to manage this condition has been an intriguing, yet unresolved issue for geriatricians. The scenario is further complicated by the fact that operational definitions of sarcopenia and criteria to select the most appropriate target population for clinical trials on this syndrome have been released only recently (6–10). Moreover, indications about the biomarkers to be used to detect sarcopenia and track its progression over time have just been published (11). However, the scientific community is still debating on how to identify clinically meaningful thresholds that distinguish normal from abnormal values of muscle mass and strength (12).
Given these limitations, it is not surprising that no pharmacological treatment is currently available to impede the progression of sarcopenia. Similarly, no pharmacological remedies exist to prevent the onset of age-related muscle loss. In this context, an adequate protein and energy intake together with physical exercise (resistance, aerobic or their combination) has been indicated as the most effective strategy currently available to manage sarcopenia (13, 14).
The purposes of this review are to present the most recent findings pertaining the role of nutrition in the management of sarcopenia, illustrate the mechanisms of action of relevant dietary agents on the aging muscle, and introduce novel nutritional strategies that may help combat sarcopenia. The paper is divided into two main sections. The first summarizes the current nutritional recommendations against sarcopenia, with a particular emphasis on those released by the Society for Sarcopenia, Cachexia, and Wasting Disease (15). This section also illustrates issues related to the identification of the optimal timing of nutritional interventions in the context of primary and secondary prevention strategies. The second part of the article introduces novel nutritional candidates and dietary strategies that may be harnessed to preserve muscle mass and function in old age. Special attention is paid to the prospect of developing personalized dietary and exercise training recommendations via the implementation of cutting edge, high-throughput analytic approaches.
Current nutritional recommendations to manage sarcopenia
On quality and quantity
Food intake declines gradually throughout adulthood. Between 20 and 80 years, the mean daily energy intake drops by approximately 1,300 kcal and 600 kcal in men and women, respectively (16), which often results in a dietary energy supply below the commonly accepted reference values (17). Several age-related pathologic conditions, including masticatory disability, and medications contribute to reducing food consumption in late life (18). Furthermore, advancing age per se is associated with a physiologic reduction in appetite (“anorexia of aging”) that can eventually evolve into pathologic anorexia and malnutrition (19). Older persons also develop changes in eating habits, with predilection for energy-dilute foods such as grains, vegetables and fruits, in place of energy-dense sweets and protein-rich nutrients (20). As a consequence of these factors, the prevalence of malnutrition ranges from 5 to 20% in community-dwelling older adults and exceeds 60% in institutionalized elderly (21).
These observations have led to the proposition that nutritional interventions based on the provision of an adequate energy supply (i.e., 24–36 kcal·kg−1·day−1) and on the supplementation of specific nutrients could be effective in preventing and/or reversing sarcopenia and physical frailty, especially when combined with physical exercise (13, 22). However, although numerous studies in older persons with overt malnutrition or specific disease conditions have shown overall positive effects of nutritional supplementation, attempts to specifically improve muscle mass and function through dietary interventions in non-malnourished sarcopenic elderly have yielded mixed results (23). It should be considered that a true nutritional supplementation is difficult to achieve in older individuals. Indeed, most elderly undergoing dietary interventions decrease their dietary intake proportionally, with the consequence that the total daily energy intake remains substantially unchanged in spite of the supplementation (23). More importantly, the composition of supplements and the duration of the intervention that have been tested so far could be inadequate to meet the actual nutritional needs of sarcopenic persons (23).
For a nutritional intervention to be effective against sarcopenia, it should: a) provide an adequate caloric intake; b) ensure the provision of appropriate nutrients, taking into account age, sex, metabolic profile, health status, physical activity level, and concomitant therapies; c) provide the adequate quality and quantity of nutrients at the right time, that is, when there is a physiological need; d) be protracted for a time sufficient to impact muscle health. Based on this premises, in 2010, the Society for Sarcopenia, Cachexia, and Wasting Disease convened an expert panel to develop nutritional recommendations for the prevention and management of sarcopenia (15). The panel, besides acknowledging the central role of physical exercise, highlighted the importance of an adequate intake of calories and several nutrients, including proteins and amino acids, vitamin D, and creatine (15). The evidence in support to these and other dietary agents as well as their mechanisms of action in the context of sarcopenia are presented in the following paragraphs.
Proteins and amino acids
Skeletal muscle mass is regulated by the complex interplay among a host of factors; however, it is undoubted that the balance between protein synthesis and breakdown plays a pivotal role in the process (24). Optimal muscle protein metabolism, in turn, is highly dependent upon an adequate intake of dietary-derived proteins and amino acids (25). A report from the Health, Aging, and Body Composition Study has recently highlighted the importance of protein intake for the preservation of lean body mass in old age (26). The association between dietary protein supply and changes in appendicular lean mass was explored in over 2,000 community-dwelling men and women aged 70–79 years during a 3-year period. After adjustment for potential confounders, individuals in the highest quintile of protein consumption lost nearly 40% less appendicular lean mass than did those in the lowest quintile (26).
Epidemiological data indicate that older persons are at high risk for inadequate protein intake. It is reported that 32–41% of women and 22–38% of men older than 50 years consume less than the recommended dietary allowance (RDA) for protein (0.8 g·kg−1·day−1), and virtually no older adult introduces the highest acceptable macronutrient distribution range (AMDR) for protein (35% of total energy intake; 27). In addition, the extraction of dietary amino acids by the splanchnic bed is altered in advanced age, which can translate into lower peripheral amino acid concentrations (28). Finally, the aged muscle possesses a reduced ability to up-regulate protein synthesis in response to anabolic stimuli, such as protein intake and physical exercise (29). Nevertheless, muscles from older individuals retain the capacity of mounting a robust anabolic response following the ingestion of protein-rich meals (30). Whether this adaption is maintained in the oldest olds is presently unknown. It is therefore very likely that the current RDA for protein does not fulfill the criterion of the “the average daily dietary nutrient intake level sufficient to meet the nutrient requirement of nearly all (97 to 98%) healthy individuals in a particular life stage and gender group” (17), since it may not adequately protect older adults from developing sarcopenia (31). It also needs to be considered that the current protein RDA was established on the basis of studies employing nitrogen balance techniques to assess protein requirements. However, such methodology does not take into account clinically relevant outcomes, such as the maintenance of muscle mass and function over the life course.
Given these considerations, an increase in protein intake above 0.8 g·kg−1·day−1 is advocated as a possible countermeasure for age-related muscle loss (32). Specifically, a daily protein intake of 1.0 g·kg−1 has been identified as the minimum amount required to maintain muscle mass in old age, at least in women (33). The feeding pattern is also important to optimize muscle protein metabolism (Figure 1). For instance, Arnal et al. (33) showed that, in older women, a 2-week protein pulse-feeding pattern, in which 80% of the daily protein intake (~1.0 g·kg−1) was provided in one meal, was more efficient in improving whole-body protein retention than the same amount of protein distributed evenly across four meals. Nevertheless, the ingestion of large quantities of proteins in a single meal may be difficult to maintain over the long term, especially in old age. Furthermore, most researchers agree that the amount of proteins introduced should be spread equally throughout the day in order to ensure a greater 24-hour anabolic response (15, 34, 35; Figure 1). Thus, according to the available evidence, older persons should be encouraged to ingest between 1.0 and 1.5 g·kg−1·day−1 of protein through the consumption of a moderate serving (25–30 g) of high-quality protein sources at each meal (15, 34, 35).
Figure 1. Proposed effects of protein intake and feeding pattern on muscle protein synthesis.
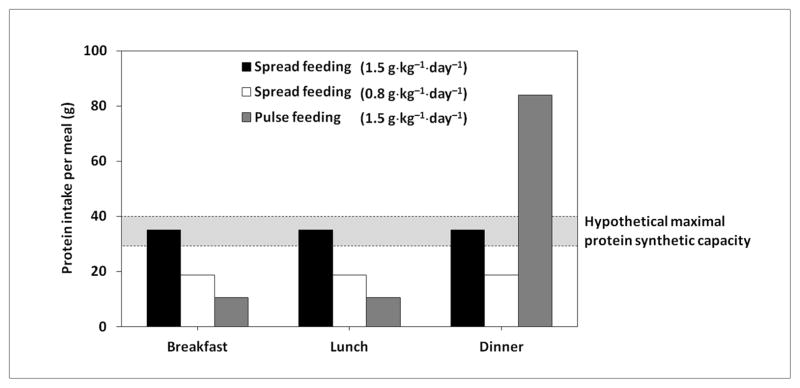
A protein intake of 0.8 g·kg−1·day−1 (56 g for a person weighing 70 kg) may not be sufficient to maximally stimulate muscle protein synthesis. The ingestion of 1.5 g·kg−1·day−1 of protein (105 g for a person weighing 70 kg) may differentially impact muscle protein synthesis depending on the feeding pattern. A spread feeding pattern, in which an equal amount of proteins is ingested at each meal, ensures a 24-h optimal stimulation of protein synthesis. A pulse feeding pattern, in which 80% of daily protein proteins is consumed in one meal and the remaining 20% is ingested in the other two meals, may not optimize the protein synthetic capacity. Adapted from Paddon-Jones D & Rasmussen BB, Curr Opin Clin Nutr Metab Care 2009 (34).
The absorption kinetics and the amino acid composition of dietary proteins are also important factors to be taken into account. Indeed, the speed of absorption of dietary amino acids by the gut affects postprandial protein synthesis, breakdown, and deposition (36). This observations led to the development of the fast vs. slow protein concept (36), which may have important implications for the management of sarcopenia. In young adults, slowly digested proteins (e.g., casein) induce greater protein retention than those that are rapidly digested (e.g., whey; 37). An opposite pattern has been detected in elderly individuals (38). Accordingly, Pennings et al. (39) have recently shown that the ingestion of whey proteins stimulates postprandial muscle protein accretion in older men more efficiently than casein or casein hydrolysate.
The amino acid composition of dietary proteins has a great impact on their muscle anabolic potency (40). Indeed, essential amino acids (EAAs) are the primary stimulus for protein synthesis (Figure 2). In particular, leucine (abundant in whey) is recognized as the master dietary regulator of muscle protein turnover (41), due to its ability to activate the mammalian target of rapamycin (mTOR) pathway (42) and inhibiting the proteasome (43). Although the aging muscle shows a reduced anabolic response to low doses (e.g., 7 g) of EAAs (44), higher doses (e.g., 10–15 g, with at least 3 g of leucine) are capable of overcoming such anabolic resistance and induce a protein synthetic response similar to that observed in young adults (45). Older persons should therefore be recommended to consume protein sources containing a relatively high proportion of EAAs (i.e., high-quality proteins), including lean meat-based and dairy products, beef meat, and leucine-rich foods (e.g., soybeans, whey, cowpea, lentils; 15, 34). Older adults practicing resistance exercise may also be advised to ingest supplements of fast proteins (e.g., whey) or amino acids (e.g., leucine-enriched balanced EAA mix) immediately before or 2–3 hours after the training session to enhance exercise-induced muscle hypertrophy (46–48).
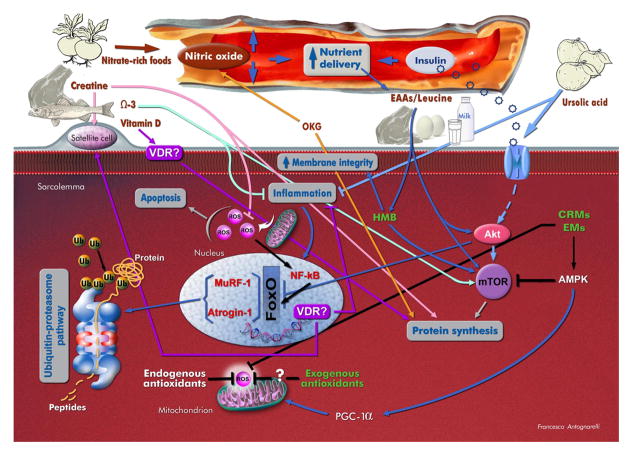
Figure 2. Overview of putative mechanisms of action of relevant dietary agents on the sarcopenic muscle.
Abbreviations: AMPK: AMP-activated protein kinase; CRMs: caloric restriction mimetics; EAAs: essential amino acids; EMs: exercise mimetics; FoxO: forkhead box protein O; HMB: β-hydroxy β-methylbutyrate; mTOR: mammalian target of rapamycin; MuRF-1: muscle-specific RING finger-1; OKG: ornithine α-ketoglutarate; PGC-1α: peroxisome proliferator-activated receptor-γ coactivator-1α; ROS: reactive oxygen species; Ub: ubiquitin; VDR: vitamin D receptor; Ω3: omega-3 fatty acids.
Common concerns related to high protein intake in advanced age (e.g., possible detrimental effects on bone, renal, neurological and cardiovascular health) are not substantiated by most studies. Nevertheless, elevated plasma concentrations of five branched-chain and aromatic amino acids (leucine, isoleucine, valine, tyrosine, and phenylalanine) have been associated with increased risk of developing diabetes in a population of 2,422 young adult and middle-aged individuals over a 12-year follow-up (49). This finding raises the concern that long-term supplementations with proteins and/or EAAs might be detrimental for muscle health, due to the association of insulin resistance with impaired protein metabolism and muscle wasting (50).
In summary, while a general agreement exists about the need for increasing the daily protein intake in advanced age, additional trials are necessary to establish the optimal amount, type and timing of protein and amino acid supplementation, and to determine possible long-term adverse effects of such nutritional regimens.
Vitamin D
Vitamin D is a secosteroid prohormone that is generated through photolysis of 7-dehydrocholesterol in the skin by UV light or obtained from the diet (51). Vitamin D from both exogenous and endogenous sources is subsequently activated by two sequential hydroxylation reactions. The first takes place largely in the liver to produce 25-hydroxy vitamin D (calcidiol). This molecule circulates bound to a specific transport protein and is indicative of vitamin D status. The second hydroxylation takes place mainly in the kidney to generate 1α,25-dihydroxy vitamin D (calcitriol), the hormonally active metabolite of vitamin D. Calcitriol is also produced by several other tissues with autocrine/paracrine pattern and local effects (51).
Serum levels of vitamin D decline steadily with aging, due to inadequate dietary intake, reduced sunshine exposure, impaired capacity of the skin to synthesize vitamin D under the influence of UV light, and diminished renal conversion of 25-hydroxy vitamin D to its active form (52). It is estimated that over 1 billion people worldwide have vitamin D deficiency or insufficiency (i.e., serum 25-hydroxy vitamin D levels < 30 ng·mL−1 or < 75 nmol·L−1; 53). Remarkably, 40 to 100% of US and European community-dwelling older adults show vitamin D deficiency (53), and the situation is even more dramatic in institutionalized elderly, as a consequence of poorer dietary intake, decreased sunshine exposure, and higher multimorbidity rates (54).
Low vitamin D levels seem to be associated with a multitude of adverse health outcomes, including osteoporosis, diabetes mellitus, rheumatoid arthritis, several cancers, cardiovascular disease, cognitive decline, multiple sclerosis, and infectious diseases (53). In addition, in older adults, low serum concentrations of vitamin D and high serum levels of parathyroid hormone (PTH) have been associated with reduced muscle mass and strength (55). In a prospective study in free-living elderly, serum levels of vitamin D were found to be strong, independent predictors of changes in muscle mass and strength over 2.6 years of follow-up (56).
Although it is undoubted that vitamin D influences muscle metabolism and trophism, the mechanisms whereby this hormone acts on skeletal myofibers are still under debate. According to classical view, actions of vitamin D are performed through binding to specific receptors (vitamin D receptors, VDRs) located on the cell surface and in the nucleus (57; Figure 2). Specifically, the activation of nuclear VDR initiates a genomic pathway that modulates the transcription of target genes involved in calcium uptake, phosphate transport across the cell membrane, phospholipid metabolism, production of inflammatory cytokines, and Sc proliferation and terminal differentiation (57). Binding of vitamin D to membrane-bound VDR triggers a non-genomic pathway that regulates the release of calcium into the cytosol, necessary for muscle contraction, and stimulates protein synthesis (58). A recent study by Wang and DeLuca (59), using a highly sensitive immunohistochemical assay, has questioned the existence of VDRs either nuclear or membrane-bound in muscle tissue. Actions of vitamin D on muscle could therefore be mediated by systemic changes involving calcium, phosphate and PTH levels in the circulation (59).
Whatever the exact mechanism of action, vitamin D supplementation to older persons has shown to improve muscle function, reduce the incidence of falls, and positively impact muscle fiber composition and morphology (60–63). Based on these findings, it is currently recommended to measure serum levels of 25-hydroxy vitamin D in all sarcopenic patients and to prescribe vitamin D supplements [800 IU (20 μg) ·day−1] to those with values lower than 100 nmol·L−1 (40 ng·mL−1; 15).
The consumption of foods rich in vitamin D should also be encouraged. The best dietary sources of vitamin D3 (cholecalciferol) include oily fish (e.g., salmon, mackerel and herring) and liver oils from cod, tuna and shark (53). Sun-dried mushrooms contain variable amounts of vitamin D2 (ergocalciferol). Foods fortified with vitamin D (both D2 and D3) are also available, such as milk, breads, some yogurts and cheeses in the US, and some margarines, cereals, and milk in Europe. Nevertheless, in the absence of adequate sunshine exposure, the RDA for vitamin D is rarely ensured by common diets and supplementation is usually necessary. Vitamin D supplementation is relatively safe; however, the risk of adverse drug reactions, in particular hypercalcemia and its consequences (e.g., nephrolithiasis, gastrointestinal abnormalities, hypertension, arterial stiffness, cognitive impairment, electrocardiographic modifications) needs to be taken into account, especially in persons with impaired renal function and/or treated with thiazide diuretics (64).
Creatine monohydrate
Creatine (Cr) is a guanidino compound that is produced endogenously in the liver and kidney from reactions involving the amino acids glycine, arginine and methionine. Cr is also introduced with foods (e.g., lean red meat, tuna, and salmon). De novo synthesized and dietary-derived Cr is actively taken up by muscle cells and neurons and converted into the high-energy metabolite phosphocreatine (PCr) in a reaction catalyzed by the enzyme creatine-kinase. During the initial phase of an intense muscular effort, PCr donates anaerobically a phosphate group to ADP to form ATP (65). At rest or during periods of low activity, the excess ATP is used to regenerate PCr from Cr.
Due to its function as a temporal and spatial energy buffer, Cr represents a valid ergogenic aid (66). In addition, studies have shown that Cr administration may promote muscle anabolism by modulating muscle protein kinetics, Sc activity and content, and anabolic hormone secretion (67; Figure 2). For instance, short-term supplementation with Cr monohydrate to young healthy subjects was found to increase the transcription of genes involved in muscle protein synthesis, cell repair and survival pathways, and Sc activation (68). The protein anabolic response induced by Cr administration is probably linked to the direct activation of the mTOR pathway (68). In addition, supplementation with Cr monohydrate, in combination with resistance training, has shown to increase muscular levels of insulin-like growth factor 1 (IGF-1) in young adult subjects (69). IGF-1, in turn, stimulates muscle protein synthesis through mTOR-dependent and independent mechanisms (70). A further means whereby Cr supplementation promotes muscle health is through the inhibition of protein breakdown. Notably, down-regulation of muscle protein catabolism following Cr administration appears to be maintained in advanced age (71). Studies have also shown that Cr supplementation attenuates ROS generation and protects against nucleic acid oxidative damage (72). However, given the complex roles of ROS in muscle physiology (vide infra), it is unclear if the anti-oxidant effect of Cr translates into muscle protection in late life.
In advanced age, a decrease in intramuscular Cr levels has been documented (73), which together with the favorable actions of Cr on muscle trophism and bioenergetics, provides a good rationale for its supplementation as a countermeasure for sarcopenia. Indeed, administration of Cr monohydrate to older adults engaged in strength training programs has shown to increase intramuscular total Cr concentration (74), augment muscle mass (71, 74, 75), and improve muscle strength (71, 74–76). Interestingly, older persons supplemented with Cr may experience long-term residual beneficial effects after the end of the intervention. Indeed, in elderly men, 12 weeks of Cr supplementation combined with resistance exercise produced significant gains in muscle mass and strength, which were retained after 12 weeks of withdrawal from Cr (77).
The combination of Cr and whey protein may potentiate the muscle anabolic effects of the two nutrients during resistance exercise. In this regard, Candow et al. (71) showed that older men supplemented with Cr monohydrate (0.1 g·kg−1·day−1) and whey protein (0.3 g·kg−1·day−1) during a 10-week whole-body resistance training experienced a greater increase in fat-free mass and muscle strength compared with those who consumed Cr alone or an isocaloric carbohydrate placebo. This finding suggests a synergistic stimulation of muscle protein anabolism by Cr and high-quality protein sources.
In summary, Cr monohydrate has emerged as an inexpensive and efficient nutritional supplement able to improve muscle mass and performance in older adults when combined with resistance exercise. Hence, short-term Cr supplementation (e.g., 5–20 g·day−1 of Cr monohydrate for 2 weeks) may be advisable in older persons engaged in strength training programs (15). Nevertheless, further studies are required to establish the optimal dosing and timing of Cr supplementation in sarcopenic elderly and to determine the risk of possible adverse events associated with this intervention (e.g., renal damage).
Antioxidants
According to the well-known free radical theory of aging, the accumulation of oxidative damage to cellular macromolecules over the life course is the central mechanism driving the aging process (78). Based on this theory, the appearance of all age-related phenotypes, including sarcopenia, should therefore be primarily ascribed to the detrimental effects of reactive oxygen species (ROS). Accordingly, in older persons, elevated levels of oxidative damage have been linked with reduced muscle mass and strength (79, 80), and mobility disability (81). Conversely, positive associations have been determined between higher antioxidant status and measures of physical function in elderly populations. For instance, in the InCHIANTI study, a population-based study of older persons living in the Chianti geographic area (Tuscany, Italy), higher plasma levels of carotenoids were associated with a lower risk of developing mobility disability (82) and declining muscle strength (83). Similar associations have been found for selenium (84), vitamin E (85), and vitamin C (86). These observations would theoretically provide a good rationale for the administration of antioxidants as a countermeasure for sarcopenia. Unfortunately, redox physiology is not as straightforward as it appears. Indeed, while excessive ROS generation is associated with the pathogenesis of several disease conditions and, perhaps, contributes to aging and sarcopenia, oxidant production within a hormetic window plays fundamental roles in cell signaling and adaptation to various stressors (87; Figure 3). For instance, during moderate intensity exercise, ROS promote force production, stimulate mitochondrial biogenesis, and improve cellular antioxidant and repair capacity (88, 89). This hormetic response is preserved in older individuals (90). Consistently, some very recent studies suggest that mild pro-oxidants might even possess anti-aging properties (91). On the other hand, the indiscriminate use of antioxidant supplements could abrogate the physiological actions of ROS, cause reductive stress and blunt the beneficial effects of non-exhaustive physical exercise (92). The scenario is further complicated by the fact that commonly prescribed antioxidants (e.g., selenium, vitamin A, vitamin C, vitamin E, and β-carotene) may paradoxically behave as potent pro-oxidants under some circumstances, due to their chemical and physical properties (93). Meta-analyses and systematic reviews have clearly shown an increased mortality risk associated with antioxidant supplementation in both healthy individuals and patients with various diseases, which should warn against the widespread use of uncontrolled supplementations (94–96).
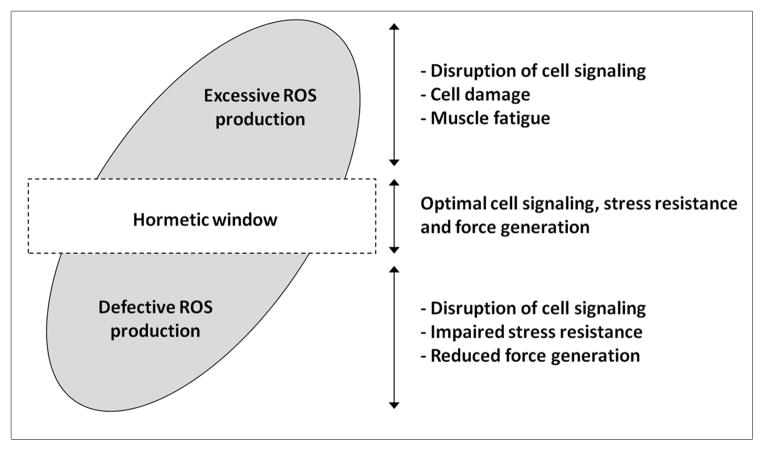
Figure 3. Proposed effects of varying levels of ROS on skeletal myocyte homeostasis.
ROS generation within the hormetic window optimizes cell signaling, stress resistance and force production. Both excessive and defective ROS generation impairs muscle tissue homeostasis by disrupting cell signaling, stress resistance and cellular integrity.
In conclusion, given the incomplete knowledge about redox physiology and the possible harms associated with antioxidant supplementation, there is currently no scientific rationale that justifies the implementation of this intervention to prevent or treat sarcopenia (97). Indeed, the recommendations by the Society for Sarcopenia, Cachexia, and Wasting Disease do not even mention antioxidants as an approach against sarcopenia (15). Hence, unless an overt deficiency is documented, antioxidant supplements are not to be prescribed to sarcopenic individuals, especially to those engaged in physical exercise programs. Nevertheless, older persons should still be advised to consume foods rich in antioxidants, such as fruits and vegetables, because they also contain a vast array of healthy nutrients (e.g., vitamins, minerals and fibers).
Type and timing of nutritional interventions for sarcopenia
The available evidence provides clear indications about the type of nutritional supplement(s) to be prescribed to sarcopenic elderly (15, 34). The choice of specific nutritional supplementations should also be based on the patient’s preferences and the eventual presence of comorbid conditions that may benefit from or be exacerbated by certain nutrients. For instance, in the presence of renal failure, protein and Cr supplements may not be advisable, whilst the administration of omega-3 fatty acids (98; vide infra) is likely to be beneficial. One important caveat of current nutritional recommendations for sarcopenia is that no clear indication is provided about the duration of the intervention that maximizes the benefits and reduces the risk for adverse effects. Indeed, while six months have been indicated as the minimum timeframe to expect sizable changes in muscle mass (11), it is unclear whether nutritional supplementations induce linear gains in muscle mass and strength over time or if a ceiling effect occurs before substantial improvements in muscle health have been attained. The evaluation of the effectiveness of nutritional interventions for sarcopenia is complicated by the multifaceted nature of the syndrome, which involves various combinations of declines in muscle mass and strength as well as in physical performance (6). It is possible that distinct nutritional interventions may differentially impact the various clinical features of sarcopenia. This implies that specific combinations of nutrients may be necessary to optimize treatment in subsets of sarcopenic patients and this needs to be addressed by future clinical trials. Finally, the safety of long-term nutritional interventions in sarcopenic elderly has yet to be established.
Another major point to be addressed concerns the optimal timing of nutritional interventions. Clearly, nutritional supplementation and possibly physical exercise need to be prescribed to all individuals diagnosed with sarcopenia. But, is it possible to identify subjects who are at risk to become sarcopenic and in whom supplementation could be started earlier in life? In this regard, it has been shown that lifestyle habits and physical health during adulthood could determine the rate of muscle strength decline and the development of functional limitations in advanced age (99, 100). For instance, Stenholm et al. (100) found that midlife physically strenuous work, excess body weight, smoking, cardiovascular disease, hypertension, diabetes mellitus, and asthma predicted muscle strength decline over 22 years of follow-up. In addition, significant weight loss, becoming physically sedentary, persistent smoking, incident coronary heart disease, diabetes mellitus, chronic bronchitis, chronic low-back pain, long-lasting cardiovascular disease, hypertension, and asthma were associated with accelerated declines in handgrip strength (100). Other common conditions associated with early onset and accelerated progression of sarcopenia include cancer, peripheral artery disease, chronic obstructive pulmonary disease, congestive heart failure, rheumatoid arthritis, kidney failure, chronic liver diseases, HIV infection, and long-term use of medications such as steroids and certain nonsteroidal anti-inflammatory drugs (50). Interestingly, birth weight and prepubertal and pubertal growth may affect muscle mass and strength as well as physical performance in late life (101–103). In further support to the existence of a developmental component in the sarcopenia syndrome, Robinson and colleagues (104) found that a greater exposure to breast milk in infancy was associated with greater grip strength in older community-dwelling men.
Taken together, these findings suggest that individuals at high risk for sarcopenia could be identified well before the decline in muscle mass and strength reaches a critical threshold. Information pertaining to physiological, pathological, functional, and pharmacological parameters could be utilized to compute a “sarcopenia risk score” or a “sarcopenia risk chart”, similar to what is routinely done for other medical conditions (e.g., cardiovascular disease and osteoporosis). Such an approach could allow the timely implementation of primary preventive strategies, including nutritional interventions. As discussed further on, the development of integrated “omics” profiling techniques will likely assist in the early identification of subjects at risk to become sarcopenic as well as in the definition of the nutritional intervention that more appropriately matches the patient’s specific needs.
Novel dietary candidates and nutrition strategies against sarcopenia
Amino acid metabolites and precursors: β-hydroxy β-methylbutyrate and ornithine α-ketoglutarate
β-hydroxy β-methylbutyrate (HMB) is a metabolite of leucine that is receiving increasing attention as a potential nutritional aid against sarcopenia (105). Following its absorption, dietary leucine is transaminated to α-ketoisocaproate (KIC), which is further metabolized into either isovaleryl-CoA or HMB (106). Under normal conditions, the majority of KIC is converted into isovaleryl-CoA, while only approximately 5% of leucine is metabolized to HMB (106). This implies that, in order to reach pharmacological levels of HMB, this compound needs to be administered directly, rather than via increasing the ingestion of leucine.
HMB has long been used as an ergogenic aid, due to its anti-catabolic, anabolic and lipolytic effects. In the skeletal muscle, HMB inhibits protein breakdown by down-regulating proteolytic pathways, stimulates protein synthesis through the activation of the mTOR pathway, improves sarcolemmal integrity via its conversion into hydroxymethylglutaryl-CoA, and increases fatty acid oxidative capacity (105; Figure 2). The effects of HMB on muscle metabolism and body composition make this compound a good candidate for nutritional interventions against sarcopenia. Indeed, studies have shown that HMB supplementation is able to reverse deficits in net anabolism and improve muscle strength in sarcopenic older adults, especially when combined with resistance exercise. For instance, Vukovich and colleagues (107) found that, compared with placebo, 8-week supplementation with HMB (3 g·day−1) elicited greater increases in muscle strength and improvements in body composition in healthy older persons participating in a strength training program. Furthermore, the daily administration of a nutritional mixture containing HMB (2 g), arginine (5 g) and lysine (1.5 g) for 12 weeks improved measurements of physical performance, muscle strength, fat-free mass and protein synthesis in sedentary older women (108). Similar results were reported in a cohort of sedentary elderly men and women, who underwent a yearlong daily supplementation with a nutrition cocktail of HMB (2–3 g), arginine (5–7.5 g), and lysine (1.5–2.25 g; 109). A post hoc analysis based on the participant’s vitamin D status revealed that the nutrient cocktail was effective in increasing muscle mass regardless of vitamin D levels (110). In contrast, muscle strength gains were observed only in participants with serum concentrations of 25-hydroxy vitamin D3 ≥ 30 ng·mL−1, indicating a synergistic effect of HMB/arginine/lysine and vitamin D.
In summary, HMB has emerged as an interesting candidate for nutritional interventions against sarcopenia. Further investigations are necessary to determine the optimal dosage, timing and duration of supplementation with HMB, as well as the effects of the combination of this compound with other nutrients (e.g., creatine, vitamin D, proteins, amino acids or associated metabolites) on muscle health in advanced age.
Ornithine α-ketoglutarate (OKG), an ionic salt formed by two molecules of ornithine and one molecule of α-ketoglutarate, is an intriguing compound with a multitude of potential favorable actions on muscle protein metabolism (Figure 2). OKG is the precursor of several amino acids, such as glutamate, glutamine, arginine and proline, and of other bioactive compounds, including polyamines, citrulline, α-ketoisocaproate and nitric oxide (NO), that are important modulators of protein metabolism and hemodynamics in skeletal muscle (111). OKG is also a potent secretagogue of anabolic hormones, such as insulin and growth hormone (111). Hence, through direct and indirect actions, OKG could intervene in several processes involved in age-related muscle loss (112).
Stimulation of protein anabolism by oral or intravenous OKG administration has been demonstrated in several pathological conditions associated with hypercatabolism and muscle wasting (e.g., malnutrition, cancer cachexia, burn injury, and surgery) in both experimental animals and humans (113–116). Importantly, administration of OKG (10 g·day−1 for 2 months) has shown to improve nutritional status, ameliorate quality of life, and reduce healthcare costs in ambulatory elderly patients (113). Although OKG may be a valuable nutritional aid against sarcopenia, the effects of its supplementation on muscle mass and function in non-malnourished older persons have yet to be investigated.
Omega-3 fatty acids
Omega-3 fatty acids (also referred to as n-3 fatty acids) are polyunsaturated fatty acids (PUFAs) with many potential health benefits (117). These nutrients, in particular eicosapentaenoic acid (EPA; C20:5 n-3), docosahexaenoic acid (DHA; C22:6 n-3) and α-linolenic acid (αLNA; 18:3 n-3), are also receiving attention as possible nutritional countermeasures for sarcopenia. Indeed, a positive association has been found between the consumption of fatty fish (the best dietary source of omega-3 fatty acids) and grip strength in a cohort of community-dwelling older persons (118). Specifically, an increase in grip strength of 0.43 kg and 0.48 kg in community-dwelling men and women, respectively, was documented for each additional portion of fatty fish consumed per week (118). Emerging evidence also suggests that supplementation with omega-3 fatty acids could improve muscle protein anabolism and reduce levels of pro-inflammatory cytokines in older adults (Figure 2). In a recent study, Smith and coworkers (119) showed that the administration of a commercially available formulation of EPA and DHA for 8 weeks increased the rate of muscle protein synthesis during hyperaminoacidemic-hyperinsulinemic clamping in healthy older persons. This effect was accompanied by increased activation of the mTOR pathway, suggesting that omega-3 fatty acids might directly stimulate protein synthesis. On the other hand, Cornish and Chilibeck (120) found that 12-week supplementation with αLNA (~14 g·day−1) combined with resistance training reduced circulating levels of interleukin 6, although the effects on muscle mass and strength were marginal.
These preliminary data suggest that an adequate intake of omega-3 fatty acids could represent an effective nutritional remedy for sarcopenia. Although there is no established dietary reference intake (DRI) for these nutrients, the adequate intake for EPA is set at 1.6 and 1.1 g·day−1 for men and women, respectively (121). Such an intake is ensured by the consumption of at least two servings of fish a week. Fatty fish like salmon, mackerel, herring, lake trout, sardines and albacore tuna and their oils are the best food sources of omega-3 fatty acids (and interestingly of vitamin D). However, it is not known if the current adequate intake for omega-3 fatty acids is sufficient to preserve muscle mass and function during aging. Investigations concerning the long-term effects of omega-3 supplementation on muscle health in old age are also warranted. Finally, the risk for adverse events associated with long-term omega-3 supplementation (e.g., prolongation of bleeding time, atrial fibrillation and flutter, and liver toxicity) in sarcopenic elderly needs to be addressed
Ursolic acid
Ursolic acid is a lipophilic pentacyclic triterpenoid abundant in apple peels, but also present in other fruits (e.g., plum, cranberry, and blueberry) and many herbs (e.g., rosemary, hawthorn, thyme, basil, oregano, and peppermint; 122). The compound is attributed anti-inflammatory and antihyperlipidemic properties, and is a major component of traditional Chinese medicinal herbs used in antidiabetic preparations (122). Recently, ursolic acid has been identified as a possible remedy for muscle atrophy in a screen for small molecule inhibitors of skeletal muscle wasting (123). In mice subjected to fasting or spinal cord injury, acute administration of ursolic acid attenuated the severity of muscle loss through the repression of atrophy-associated genes (atrogin-1 and MuRF1), up-regulation of trophic genes (PKB/Akt and S6 kinase), and stimulation of insulin/IGF-1 signaling (123; Figure 2). Chronic administration of ursolic acid to unstressed mice induced muscle hypertrophy with similar mechanisms (123). In a later study from the same group, supplementation with ursolic acid to mice consuming a high-fat diet increased muscle mass and strength, improved glucose tolerance, promoted brown adipose tissue deposition, and decreased white adipose tissue mass and the severity of hepatic steatosis (124).
As a whole, these preclinical findings suggest that ursolic acid might represent an ideal nutritional aid to counteract age-related changes in body composition and to improve metabolic homeostasis. However, the effects of supplementation with ursolic acid in sarcopenic elderly have yet to be explored.
Nitrate and nitrate-rich foods
For long time, nitrate (NO3−) and nitrite (NO2−) have been considered inert end products of NO metabolism and potentially toxic food additives. However, recent evidence indicates that NO3− and NO2− from both endogenous and dietary sources are metabolized in blood and tissues to bioactive nitric oxides, including NO (125). The latter is a pleiotropic cell signaling molecule involved in the regulation of blood flow, blood pressure, glucose homeostasis, mitochondrial bioenergetics and biogenesis, inflammation, and tissue response to low oxygen levels (125).
The diet is a major source of NO3−, the content of which is especially high in certain vegetables (e.g., celery, cress, chervil, lettuce, red beetroot, spinach, and rocket). Food-derived NO3− is actively taken up by the salivary glands, excreted in saliva, and reduced to NO2− by commensal bacteria of the oral cavity (126). Several mechanisms, including non-enzymatic processes mediated by vitamin C and polyphenols, are then responsible for the reduction of NO2− to NO (126).
The effects of dietary NO3− on the aging muscle have not yet been investigated; however some of the processes involved in the pathogenesis of sarcopenia could be targeted by this nutritional factor (Figure 2). For example, dietary NO3− increases gastric NO levels, which could relieve the earlier satiety feeling typical of old age, therefore eliminating one of the components of “anorexia of aging” (19). Moreover, insulin resistance, a highly common condition in late life, is mechanistically associated with endothelial dysfunction as well as with reduced muscle perfusion and anabolic signaling (50). Dietary NO3−, by increasing circulating NO levels, could improve endothelial function and nutrient supply to muscles, therefore restoring a normal protein anabolic response to insulin. Furthermore, supplementation with dietary inorganic NO3− has recently been shown to reduce the metabolic cost of exercise and to improve muscular mitochondrial bioenergetics in young healthy subjects (127, 128). This effect may have potential implications for the management of sarcopenia, given the central role postulated for mitochondrial dysfunction in the pathogenesis of age-related muscle degeneration (129). Finally, our group has recently reported that the administration of the angiotensin-converting enzyme inhibitor enalapril to aged rats ameliorated sarcopenia indices partly via improvements in NO signaling (130, 131). It is possible that similar adaptations might be elicited by supplementation with NO3−.
Studies are required to establish whether supplementation with inorganic NO3− can offer a nutritional aid for the prevention and treatment of sarcopenia. Investigations are also needed to address the traditional, but still controversial safety issues regarding the consumption of NO2− and NO3− for their supposed association with gastric cancer and increased generation of reactive nitrogen species.
Caloric restriction mimetics, exercise mimetics and gymnomimetics
A large body of evidence indicates that physiological stressors such as mild to moderate caloric restriction (CR) and regular physical exercise exert converging beneficial effects on overall health and muscle homeostasis in advanced age through their actions on muscular mitochondrial function, protein metabolism, and cell death/survival pathways (132). Nevertheless, it is likely that most people will not be able to maintain substantial food restrictions for the long term. Furthermore, weight loss may be harmful in nonobese older persons, as it can accelerate muscle loss and increase the risk of disability and mortality (133). Similarly, the implementation of exercise training interventions is hampered by the lack of motivation of most persons. In addition, many older adults may be unable to engage in regular physical exercise due to concomitant disabling conditions.
Intriguingly, recent studies suggest that a broad range of bioactive substances from plant sources, including fruits, vegetables, grains, herbs and spices, could mimic the signaling events that underpin some of the effects of CR and physical exercise, thus partially reproducing their health benefits (134). These dietary-derived “CR mimetics” (CRMs) and “exercise mimetics” (EMs) are phytochemicals with well-recognized antioxidant properties, but their bioactivity is likely promoted by a multitude of other mechanisms (Figure 2). Examples of such “mimetic” compounds include resveratrol (found in grapes and red wine), quercetin (found in apples, onions and berries), epigallocatechin-3-gallate (found in green tea), and nootkatone (found in grapefruit). Studies in vitro and in rodent models have shown that these phytochemicals are able to activate several pathways stimulated by both CR and exercise, including peroxisome proliferator-activated receptor-γ coactivator-1α (PGC-1α), sirtuin 1 (SIRT1), and AMP-activated protein kinase (AMPK; 134). In addition, preclinical investigations found that the administration of specific CRMs or EMs to rodents was able to promote skeletal muscle anabolism (135, 136). However, the effects of these mimetic agents on human muscle aging have not yet been investigated.
Although no single compound will probably ever reproduce the broad range of health benefits brought about by CR or exercise, the prospect of mimicking, even if partly, these physiological stressors is especially appealing for geriatric medicine. The development of effective CRMs and EMs requires a comprehensive scientific approach to untangle the complexity of stress responses to CR and exercise of the organism as a whole as well as tissue, organ and systems adaptations to acute and chronic perturbations (137). A recent work by Lewis and colleagues (138) has epitomized this type of approach through the characterization of changes in plasma levels of a wide range of metabolites in response to aerobic exercise. Exposure of cultured myotubes to a combination of metabolites, the levels of which were increased in the circulation in response to exercise (glycerol, niacinamide, glucose-6-phosphate, pantothenate, and succinate), resulted in up-regulation of nur77 (138). The latter is an orphan nuclear receptor that is induced in skeletal muscle after exercise as well as a transcriptional regulator of genes involved in glucose and lipid metabolism. Interestingly, the “gymnomimetic” effect was not observed when the metabolites were used separately.
Despite these provocative findings, the field of CRMs, EMs and gymnomimetics is still in its infancy. Much more research is needed to obtain a better characterization of the bioactive properties of these compounds and a deeper understanding of the stressor-activated metabolic pathways they are supposed to mimic. Finally, studies are required to determine if supplementation with CRMs, EMs and gymnomimetics may be harnessed as a safe therapeutic approach against sarcopenia.
Manipulation of the gut microbiota
Humans are complex biological “superorganisms”, in which vast, diverse and dynamic microbial ecosystems have coevolved (139). Symbiontic microbial inhabitants are found in several discrete host niches, such as the skin, the oral cavity and the respiratory, gastrointestinal (GI) and urogenital tracts. The interaction between the human host and its microbial inhabitants plays an essential role in the definition of the host’s health status. This is not surprising given the fact that the microbial genetic repertoire (microbiome) is approximately 100-fold more abundant than that of the human host. The GI tract, in particular, houses over 1,000 distinct bacterial species, for a total of about 1014 microorganisms (gut microbiota), with important implications for nutrient bioavailability, glucose and lipid metabolism, immune system conditioning and response, protection against pathogens, and drug metabolism and toxicity (140). Notably, dysregulated host-microbial interactions have been directly implicated in the development of a number of pathological conditions such as obesity, type I and type II diabetes mellitus, cardiovascular disease, asthma, eczema, inflammatory bowel diseases, colorectal cancer, hepatic steatosis, multiple sclerosis, and autism (141, 142).
Substantial inter-individual variability occurs within the gut microbiota, and numerous human factors, including the host’s genome, diet, age and eventual drug therapies, exert positive or negative selective pressure upon the microbiota (140). The adult-like structure of the gut microbiota becomes established approximately after the first year of life, and remains relatively stable through healthy adulthood (143). In advanced age, changes in the GI tract function and other age-related events (e.g., modifications of diet and lifestyle habits, and alterations of the immune system) deeply affect the structure and function of the gut microbiota as well as its homeostasis with the host’s immune system (144, 145). The altered human-microbial relationship can eventually lead to low-grade chronic inflammation, greater susceptibility to systemic infections, malnutrition, side effects of medications, and possibly accelerated progression of chronic diseases, frailty, and sarcopenia (144, 145). These observations suggest that appropriate manipulations of the gut microbiota might be harnessed to obtain therapeutic gains in old age. In this context, comprehensive approaches based on the combination of different “omic” sciences [e.g., (meta)genomics, microbiomics, trascriptomics, proteomics, and metabolomics] are receiving considerable interest, as they could shed light on the reciprocal influence between age-related changes in the gut microbiota and the physiology of older individuals. Such methodologies could eventually identify possible targets for pharmaco-nutritional interventions aiming at improving the wellness of elderly people, for example through the administration of functional foods targeting an individual’s specific needs.
As for sarcopenia, a better understanding of the symbiotic relationship between the aging human host and the gut microbiota is of utmost importance to develop interventions aimed at restoring an optimal human-microbial relationship. Indeed, alterations in the gut microbiota could contribute to etiopathogenesis of sarcopenia, since this microbial ecosystem is involved in the regulation of inflammatory and redox status, splanchnic extraction of nutrients, fat mass deposition, and insulin sensitivity. In addition, the gut microbiota may influence (and be influenced by) the bioavailability and bioactivity of most nutritional factors proposed as remedies against sarcopenia. For instance, the colonic microbiota could modulate the metabolic fate of candidate CRMs and EMs (e.g., resveratrol, quercetin, epigallocatechin-3-gallate), by converting these compounds into bioactive substances (146). In turn, these phytochemicals could modify the gut microbial composition and/or activity (146).
Besides the administration of antibiotics, three major approaches are currently available to manipulate the composition and function of the gut microbiota: probiotics, prebiotics and synbiotics. Probiotics are defined as “live microorganisms which when administered in adequate amounts confer a health benefit on the host” (147). Lactic acid bacteria, bifidobacteria and certain yeasts and bacilli are the most common microbes used as probiotics. Prebiotics consists in “nondigestible food ingredients that beneficially affect the host by selectively stimulating the growth and/or activity of one or a limited number of bacterial species already resident in the colon” (148). Examples include galacto-oligosaccharides, fructo-oligosaccharides, inulin, and lactitol. Finally, synbiotics are nutritional supplements combining probiotics and prebiotics, the administration of which produces health benefits resulting from the synergistic actions of the two components. Studies have shown that supplementation with probiotics, prebiotics and synbiotics can improve bowel function, modulate immune system function, and improve nutrient bioavailability (149). However, no studies have yet specifically investigated the effects of such interventions on muscle aging. Given the importance of the gut microbiota in the regulation of human physiology, further research is warranted to explore the potential application of its manipulation for the management of sarcopenia.
Concluding remarks and future perspectives
Sarcopenia is one of the most burdensome geriatric syndromes. The complexity of its pathogenesis and the heterogeneity of clinical correlates render the development of preventive and therapeutic interventions a challenging task. Nevertheless, specific nutritional interventions have shown to produce significant improvements in muscle mass and function in older adults, especially when combined with physical exercise. Furthermore, novel nutritional factors and dietary intervention strategies are coming on the scene.
Future developments in the field will require a shift of paradigm, moving from a disease-centered concept toward a holistic, network-based perspective of sarcopenia (150). Sophisticated high-throughput analytic platforms have recently been developed to characterize the phenotype of a person from his/her genetic background to the nearly complete repertoire of molecules representing crucial metabolic processes of the human-microbe hybrid (151–153). The resulting integrated personal “omics” profile (iPOP) may allow to monitor an individual’s health status, predict the development of specific disease conditions, including sarcopenia, and plan preventive interventions (152, 154). In sarcopenic individuals, the adoption of an integrated “multiomic” approach, combined with appropriate functional and imaging assessments, could help develop a full and personalized characterization of the patient, identify the system(s) compromised, and implement targeted treatments (Figure 4). Such an approach would also provide a fundamentally different perspective on diet management. For instance, nutrigenomics and metabolomics, through the characterization of an individual’s nutritional and metabolic status, may provide critical information on the way nutrients are harnessed by the body and on the effects of nutrient consumption on the organism physiology (153). This knowledge is crucial to design effective dietary interventions that match the actual requirements of the sarcopenic patient and, more in general, drive an individual’s metabolic phenotype to a healthier direction. The development of effective nutritional intervention to manage chronic conditions, including sarcopenia, also relies on the appreciation of the functional effects of the diet as a whole, moving from traditional studies on single dietary agents to investigations on the global diet effect on the individual’s physiology (153). The implementation of such approach will likely open the way to personalized diets and nutritional forecasting, therefore allowing to tailor dietary recommendations to the person’s needs in both health and disease.
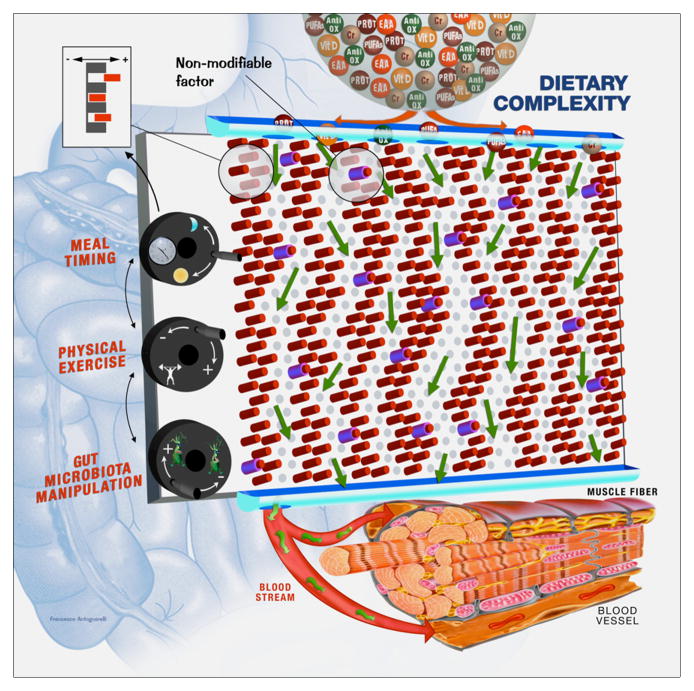
Figure 4. Dynamic “Pachinko model” of nutritional regulation of muscle physiology.
The complex mechanisms whereby diet influences muscle physiology could be simplistically represented by the operation of a Pachinko machine (Japanese pinball). In this model, proposed for the first time by Nicholson & Wilson in 2003 (155) to describe xenobiotic metabolism, nutrition may be considered as a flow of balls (dietary constituents) through the “human system”. The metabolic fate of nutrients and their effects on muscle physiology are not determined absolutely, but are probabilistic and influenced by several factors (depicted as pins). Two types of pins are distinguished. One set corresponds to non-modifiable factors, such as age, sex, genetic background, etc. The other set of pins identifies modifiable factors, such as epigenetic and/or transcriptional regulation of genes, post-translational modifications of enzymes, physiochemical conditions, hemodynamics, etc., which could be tuned (figure inset) through hypothetical “control knobs”. “Control knobs” represent external factors (e.g., meal timing, type and amount of physical activity, and manipulation of gut microbiota composition and function) that can be exploited to optimize the effects of nutrients on muscle physiology. According to the proposed model, muscle health can be impacted by manipulating the diet composition and/or by acting on “control knobs”. This dynamic Pachinko model epitomizes the complexity that needs to be deciphered to develop a personalized multimodal intervention against sarcopenia. The implementation of integrated “multiomic” approaches may enable the identification of the outputs (exit holes) of the “metabolic pin-ball”. This information, combined with appropriate functional and imaging assessments, could allow to develop a full and personalized characterization of the sarcopenic patient, identify the system(s) compromised, and design targeted treatments. Abbreviations: Anti-Ox: antioxidants; Cr: creatine; EAA: essential amino acids; Prot: proteins; PUFAs: polyunsaturated fatty acids; Vit D: vitamin D.
In conclusion, the combination of well-established imaging and functional assessments with innovative high-throughput methodologies may help decipher the complexity of sarcopenia and provide a whole new avenue for the prevention and treatment of this syndrome (Figure 4).
Acknowledgments
This work was partly supported by funds from the Italian Ministry of Economy and Finance to the CNR for the Project FaReBio di Qualità, AICR No 11-0075 (R.C.), NIA RO1-AG21042, NIDDK RO1-DK090115-01A1 and the University of Florida’s Institute on Aging and Claude D. Pepper Older Americans Independence Center (NIA 1P30AG028740, C.L.), and by the Centro Studi Achille e Linda Lorenzon (R.B. and E.M.). The authors thank Mr. Francesco Antognarelli for his invaluable assistance with illustrations.
Footnotes
Conflict of interest: C.C.S. has served as consultant for Abbott, Baxter, Nestlé and Nutricia, and received research funding from Nestlé and Nutricia; M.C. has served as consultant for Sanofi France and received research funding from Pfizer.; A.M., C.L., E.M., F.L., M.B., R.B., and R.C. have no conflict of interest to disclose.
References
- 1.Roseberg IH. Summary comments. Am J Clin Nutr. 1989;50:1231–3. [Google Scholar]
- 2.Hughes VA, Frontera WR, Roubenoff R, Evans WJ, Singh MA. Longitudinal changes in body composition in older men and women: role of body weight change and physical activity. Am J Clin Nutr. 2002;76:473–81. doi: 10.1093/ajcn/76.2.473. [DOI] [PubMed] [Google Scholar]
- 3.Forbes GB. Longitudinal changes in adult fat-free mass: influence of body weight. Am J Clin Nutr. 1999;70:1025–31. doi: 10.1093/ajcn/70.6.1025. [DOI] [PubMed] [Google Scholar]
- 4.Waters DL, Baumgartner RN, Garry PJ, Vellas B. Advantages of dietary, exercise-related, and therapeutic interventions to prevent and treat sarcopenia in adult patients: an update. Clin Interv Aging. 2010;5:259–70. doi: 10.2147/cia.s6920. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Marzetti E, Lees HA, Wohlgemuth SE, Leeuwenburgh C. Sarcopenia of aging: Underlying cellular mechanisms and protection by calorie restriction. BioFactors. 2009;35:28–35. doi: 10.1002/biof.5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Cruz-Jentoft AJ, Baeyens JP, Bauer JM, Boirie Y, Cederholm T, Landi F, et al. Sarcopenia: European consensus on definition and diagnosis: Report of the European Working Group on Sarcopenia in Older People. Age Ageing. 2010;39:412–23. doi: 10.1093/ageing/afq034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Muscaritoli M, Anker SD, Argiles J, Aversa Z, Bauer JM, Biolo G, et al. Consensus definition of sarcopenia, cachexia and pre-cachexia: joint document elaborated by Special Interest Groups (SIG) “cachexia-anorexia in chronic wasting diseases” and “nutrition in geriatrics”. Clin Nutr. 2010;29:154–9. doi: 10.1016/j.clnu.2009.12.004. [DOI] [PubMed] [Google Scholar]
- 8.Fielding RA, Vellas B, Evans WJ, Bhasin S, Morley JE, Newman AB, et al. Sarcopenia: an undiagnosed condition in older adults. Current consensus definition: prevalence, etiology, and consequences. International working group on sarcopenia. J Am Med Dir Assoc. 2011;12:249–56. doi: 10.1016/j.jamda.2011.01.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Morley JE, Abbatecola AM, Argiles JM, Baracos V, Bauer J, Bhasin S, et al. Sarcopenia with limited mobility: an international consensus. J Am Med Dir Assoc. 2011;12:403–9. doi: 10.1016/j.jamda.2011.04.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Pahor M, Cesari M. Designing Phase II B trials in sarcopenia: the best target population. J Nutr Health Aging. 2011;15:725–30. doi: 10.1007/s12603-011-0058-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Cesari M, Fielding RA, Pahor M, Goodpaster B, Hellerstein M, Abellan van Kan G, et al. Biomarkers of sarcopenia in clinical trials. Recommendations from the International Working Group on sarcopenia. J Frailty Aging. 2012;1:102–10. doi: 10.1007/s13539-012-0078-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Marzetti E. Imaging, functional and biological markers for sarcopenia: the pursuit of the golden ratio. J Frailty Aging. 2012;1:97–8. doi: 10.14283/jfa.2012.15. [DOI] [PubMed] [Google Scholar]
- 13.Rolland Y, Dupuy C, Abellan van Kan G, Gillette S, Vellas B. Treatment strategies for sarcopenia and frailty. Med Clin North Am. 2011;95:427–38. ix. doi: 10.1016/j.mcna.2011.02.008. [DOI] [PubMed] [Google Scholar]
- 14.Malafarina V, Uriz-Otano F, Iniesta R, Gil-Guerrero L. Effectiveness of nutritional supplementation on muscle mass in treatment of sarcopenia in old age: a systematic review. J Am Med Dir Assoc. 2012 doi: 10.1016/j.jamda.2012.08.001. In press. [DOI] [PubMed] [Google Scholar]
- 15.Morley JE, Argiles JM, Evans WJ, Bhasin S, Cella D, Deutz NE, et al. Nutritional recommendations for the management of sarcopenia. J Am Med Dir Assoc. 2010;11:391–6. doi: 10.1016/j.jamda.2010.04.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Briefel RR, McDowell MA, Alaimo K, Caughman CR, Bischof AL, Carroll MD, et al. Total energy intake of the US population: the third National Health and Nutrition Examination Survey, 1988–1991. Am J Clin Nutr. 1995;62:1072S–80S. doi: 10.1093/ajcn/62.5.1072S. [DOI] [PubMed] [Google Scholar]
- 17.Trumbo P, Schlicker S, Yates AA, Poos M. Dietary reference intakes for energy, carbohydrate, fiber, fat, fatty acids, cholesterol, protein and amino acids. J Am Diet Assoc. 2002;102:1621–30. doi: 10.1016/s0002-8223(02)90346-9. [DOI] [PubMed] [Google Scholar]
- 18.Morley JE. Undernutrition in older adults. Fam Pract. 2012;29 (Suppl 1):i89–i93. doi: 10.1093/fampra/cmr054. [DOI] [PubMed] [Google Scholar]
- 19.Morley JE. Anorexia of aging: a true geriatric syndrome. J Nutr Health Aging. 2012;16:422–5. doi: 10.1007/s12603-012-0061-9. [DOI] [PubMed] [Google Scholar]
- 20.Drewnowski A, Shultz JM. Impact of aging on eating behaviors, food choices, nutrition, and health status. J Nutr Health Aging. 2001;5:75–9. [PubMed] [Google Scholar]
- 21.Sieber CC. Nutritional screening tools--How does the MNA compare? Proceedings of the session held in Chicago May 2–3, 2006 (15 Years of Mini Nutritional Assessment) J Nutr Health Aging. 2006;10:488–92. [PubMed] [Google Scholar]
- 22.Volkert D, Sieber CC. Protein requirements in the elderly. Int J Vitam Nutr Res. 2011;81:109–19. doi: 10.1024/0300-9831/a000061. [DOI] [PubMed] [Google Scholar]
- 23.Milne AC, Avenell A, Potter J. Meta-analysis: protein and energy supplementation in older people. Ann Intern Med. 2006;144:37–48. doi: 10.7326/0003-4819-144-1-200601030-00008. [DOI] [PubMed] [Google Scholar]
- 24.Paddon-Jones D, Short KR, Campbell WW, Volpi E, Wolfe RR. Role of dietary protein in the sarcopenia of aging. Am J Clin Nutr. 2008;87:1562S–6S. doi: 10.1093/ajcn/87.5.1562S. [DOI] [PubMed] [Google Scholar]
- 25.Wolfe RR. Regulation of muscle protein by amino acids. J Nutr. 2002;132:3219S–24S. doi: 10.1093/jn/131.10.3219S. [DOI] [PubMed] [Google Scholar]
- 26.Houston DK, Nicklas BJ, Ding J, Harris TB, Tylavsky FA, Newman AB, et al. Dietary protein intake is associated with lean mass change in older, community-dwelling adults: the Health, Aging, and Body Composition (Health ABC) Study. Am J Clin Nutr. 2008;87:150–5. doi: 10.1093/ajcn/87.1.150. [DOI] [PubMed] [Google Scholar]
- 27.Kerstetter JE, O’Brien KO, Insogna KL. Low protein intake: the impact on calcium and bone homeostasis in humans. J Nutr. 2003;133:855S–61S. doi: 10.1093/jn/133.3.855S. [DOI] [PubMed] [Google Scholar]
- 28.Boirie Y, Gachon P, Beaufrere B. Splanchnic and whole-body leucine kinetics in young and elderly men. Am J Clin Nutr. 1997;65:489–95. doi: 10.1093/ajcn/65.2.489. [DOI] [PubMed] [Google Scholar]
- 29.Koopman R. Dietary protein and exercise training in ageing. Proc Nutr Soc. 2011;70:104–13. doi: 10.1017/S0029665110003927. [DOI] [PubMed] [Google Scholar]
- 30.Symons TB, Schutzler SE, Cocke TL, Chinkes DL, Wolfe RR, Paddon-Jones D. Aging does not impair the anabolic response to a protein-rich meal. Am J Clin Nutr. 2007;86:451–6. doi: 10.1093/ajcn/86.2.451. [DOI] [PubMed] [Google Scholar]
- 31.Campbell WW, Trappe TA, Wolfe RR, Evans WJ. The recommended dietary allowance for protein may not be adequate for older people to maintain skeletal muscle. J Gerontol A Biol Sci Med Sci. 2001;56:M373–M380. doi: 10.1093/gerona/56.6.m373. [DOI] [PubMed] [Google Scholar]
- 32.Gaffney-Stomberg E, Insogna KL, Rodriguez NR, Kerstetter JE. Increasing dietary protein requirements in elderly people for optimal muscle and bone health. J Am Geriatr Soc. 2009;57:1073–9. doi: 10.1111/j.1532-5415.2009.02285.x. [DOI] [PubMed] [Google Scholar]
- 33.Arnal MA, Mosoni L, Boirie Y, Houlier ML, Morin L, Verdier E, et al. Protein pulse feeding improves protein retention in elderly women. Am J Clin Nutr. 1999;69:1202–8. doi: 10.1093/ajcn/69.6.1202. [DOI] [PubMed] [Google Scholar]
- 34.Paddon-Jones D, Rasmussen BB. Dietary protein recommendations and the prevention of sarcopenia. Curr Opin Clin Nutr Metab Care. 2009;12:86–90. doi: 10.1097/MCO.0b013e32831cef8b. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Symons TB, Sheffield-Moore M, Wolfe RR, Paddon-Jones D. A moderate serving of high-quality protein maximally stimulates skeletal muscle protein synthesis in young and elderly subjects. J Am Diet Assoc. 2009;109:1582–6. doi: 10.1016/j.jada.2009.06.369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Boirie Y, Dangin M, Gachon P, Vasson MP, Maubois JL, Beaufrere B. Slow and fast dietary proteins differently modulate postprandial protein accretion. Proc Natl Acad Sci U S A. 1997;94:14930–5. doi: 10.1073/pnas.94.26.14930. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Dangin M, Boirie Y, Garcia-Rodenas C, Gachon P, Fauquant J, Callier P, et al. The digestion rate of protein is an independent regulating factor of postprandial protein retention. Am J Physiol Endocrinol Metab. 2001;280:E340–E348. doi: 10.1152/ajpendo.2001.280.2.E340. [DOI] [PubMed] [Google Scholar]
- 38.Dangin M, Guillet C, Garcia-Rodenas C, Gachon P, Bouteloup-Demange C, Reiffers-Magnani K, et al. The rate of protein digestion affects protein gain differently during aging in humans. J Physiol. 2003;549:635–44. doi: 10.1113/jphysiol.2002.036897. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Pennings B, Boirie Y, Senden JM, Gijsen AP, Kuipers H, Van Loon LJ. Whey protein stimulates postprandial muscle protein accretion more effectively than do casein and casein hydrolysate in older men. Am J Clin Nutr. 2011;93:997–1005. doi: 10.3945/ajcn.110.008102. [DOI] [PubMed] [Google Scholar]
- 40.Tang JE, Phillips SM. Maximizing muscle protein anabolism: the role of protein quality. Curr Opin Clin Nutr Metab Care. 2009;12:66–71. doi: 10.1097/MCO.0b013e32831cef75. [DOI] [PubMed] [Google Scholar]
- 41.Anthony JC, Anthony TG, Kimball SR, Jefferson LS. Signaling pathways involved in translational control of protein synthesis in skeletal muscle by leucine. J Nutr. 2001;131:856S–60S. doi: 10.1093/jn/131.3.856S. [DOI] [PubMed] [Google Scholar]
- 42.Anthony JC, Yoshizawa F, Anthony TG, Vary TC, Jefferson LS, Kimball SR. Leucine stimulates translation initiation in skeletal muscle of postabsorptive rats via a rapamycin-sensitive pathway. J Nutr. 2000;130:2413–9. doi: 10.1093/jn/130.10.2413. [DOI] [PubMed] [Google Scholar]
- 43.Nakashima K, Ishida A, Yamazaki M, Abe H. Leucine suppresses myofibrillar proteolysis by down-regulating ubiquitin-proteasome pathway in chick skeletal muscles. Biochem Biophys Res Commun. 2005;336:660–6. doi: 10.1016/j.bbrc.2005.08.138. [DOI] [PubMed] [Google Scholar]
- 44.Katsanos CS, Kobayashi H, Sheffield-Moore M, Aarsland A, Wolfe RR. Aging is associated with diminished accretion of muscle proteins after the ingestion of a small bolus of essential amino acids. Am J Clin Nutr. 2005;82:1065–73. doi: 10.1093/ajcn/82.5.1065. [DOI] [PubMed] [Google Scholar]
- 45.Katsanos CS, Kobayashi H, Sheffield-Moore M, Aarsland A, Wolfe RR. A high proportion of leucine is required for optimal stimulation of the rate of muscle protein synthesis by essential amino acids in the elderly. Am J Physiol Endocrinol Metab. 2006;291:E381–E387. doi: 10.1152/ajpendo.00488.2005. [DOI] [PubMed] [Google Scholar]
- 46.Tipton KD, Rasmussen BB, Miller SL, Wolf SE, Owens-Stovall SK, Petrini BE, et al. Timing of amino acid-carbohydrate ingestion alters anabolic response of muscle to resistance exercise. Am J Physiol Endocrinol Metab. 2001;281:E197–E206. doi: 10.1152/ajpendo.2001.281.2.E197. [DOI] [PubMed] [Google Scholar]
- 47.Pennings B, Koopman R, Beelen M, Senden JM, Saris WH, Van Loon LJ. Exercising before protein intake allows for greater use of dietary protein-derived amino acids for de novo muscle protein synthesis in both young and elderly men. Am J Clin Nutr. 2011;93:322–31. doi: 10.3945/ajcn.2010.29649. [DOI] [PubMed] [Google Scholar]
- 48.Pasiakos SM, McClung HL, McClung JP, Margolis LM, Andersen NE, Cloutier GJ, et al. Leucine-enriched essential amino acid supplementation during moderate steady state exercise enhances postexercise muscle protein synthesis. Am J Clin Nutr. 2011;94:809–18. doi: 10.3945/ajcn.111.017061. [DOI] [PubMed] [Google Scholar]
- 49.Wang TJ, Larson MG, Vasan RS, Cheng S, Rhee EP, McCabe E, et al. Metabolite profiles and the risk of developing diabetes. Nat Med. 2011;17:448–53. doi: 10.1038/nm.2307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Buford TW, Anton SD, Judge AR, Marzetti E, Wohlgemuth SE, Carter CS, et al. Models of accelerated sarcopenia: critical pieces for solving the puzzle of age-related muscle atrophy. Ageing Res Rev. 2010;9:369–83. doi: 10.1016/j.arr.2010.04.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Cesari M, Incalzi RA, Zamboni V, Pahor M. Vitamin D hormone: a multitude of actions potentially influencing the physical function decline in older persons. Geriatr Gerontol Int. 2011;11:133–42. doi: 10.1111/j.1447-0594.2010.00668.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Johnson MA, Kimlin MG. Vitamin D, aging, and the 2005 Dietary Guidelines for Americans. Nutr Rev. 2006;64:410–21. doi: 10.1111/j.1753-4887.2006.tb00226.x. [DOI] [PubMed] [Google Scholar]
- 53.Holick MF. Vitamin D deficiency. N Engl J Med. 2007;357:266–81. doi: 10.1056/NEJMra070553. [DOI] [PubMed] [Google Scholar]
- 54.Braddy KK, Imam SN, Palla KR, Lee TA. Vitamin D deficiency/insufficiency practice patterns in a veterans health administration long-term care population: a retrospective analysis. J Am Med Dir Assoc. 2009;10:653–7. doi: 10.1016/j.jamda.2009.08.010. [DOI] [PubMed] [Google Scholar]
- 55.Visser M, Deeg DJ, Lips P. Low vitamin D and high parathyroid hormone levels as determinants of loss of muscle strength and muscle mass (sarcopenia): the Longitudinal Aging Study Amsterdam. J Clin Endocrinol Metab. 2003;88:5766–72. doi: 10.1210/jc.2003-030604. [DOI] [PubMed] [Google Scholar]
- 56.Scott D, Blizzard L, Fell J, Ding C, Winzenberg T, Jones G. A prospective study of the associations between 25-hydroxy-vitamin D, sarcopenia progression and physical activity in older adults. Clin Endocrinol (Oxf) 2010;73:581–7. doi: 10.1111/j.1365-2265.2010.03858.x. [DOI] [PubMed] [Google Scholar]
- 57.Ceglia L. Vitamin D and skeletal muscle tissue and function. Mol Aspects Med. 2008;29:407–14. doi: 10.1016/j.mam.2008.07.002. [DOI] [PubMed] [Google Scholar]
- 58.Montero-Odasso M, Duque G. Vitamin D in the aging musculoskeletal system: an authentic strength preserving hormone. Mol Aspects Med. 2005;26:203–19. doi: 10.1016/j.mam.2005.01.005. [DOI] [PubMed] [Google Scholar]
- 59.Wang Y, DeLuca HF. Is the vitamin D receptor found in muscle? Endocrinology. 2011;152:354–63. doi: 10.1210/en.2010-1109. [DOI] [PubMed] [Google Scholar]
- 60.Bischoff HA, Stahelin HB, Dick W, Akos R, Knecht M, Salis C, et al. Effects of vitamin D and calcium supplementation on falls: a randomized controlled trial. J Bone Miner Res. 2003;18:343–51. doi: 10.1359/jbmr.2003.18.2.343. [DOI] [PubMed] [Google Scholar]
- 61.Pfeifer M, Begerow B, Minne HW, Suppan K, Fahrleitner-Pammer A, Dobnig H. Effects of a long-term vitamin D and calcium supplementation on falls and parameters of muscle function in community-dwelling older individuals. Osteoporos Int. 2009;20:315–22. doi: 10.1007/s00198-008-0662-7. [DOI] [PubMed] [Google Scholar]
- 62.Sato Y, Iwamoto J, Kanoko T, Satoh K. Low-dose vitamin D prevents muscular atrophy and reduces falls and hip fractures in women after stroke: a randomized controlled trial. Cerebrovasc Dis. 2005;20:187–92. doi: 10.1159/000087203. [DOI] [PubMed] [Google Scholar]
- 63.Yamada M, Arai H, Yoshimura K, Kajiwara Y, Sonoda T, Nishiguchi S, et al. Nutritional supplementation during resistance training improved skeletal muscle mass in community-dwelling frail older adults. J Frailty Aging. 2012;1:64–70. doi: 10.14283/jfa.2012.12. [DOI] [PubMed] [Google Scholar]
- 64.Hathcock JN, Shao A, Vieth R, Heaney R. Risk assessment for vitamin D. Am J Clin Nutr. 2007;85:6–18. doi: 10.1093/ajcn/85.1.6. [DOI] [PubMed] [Google Scholar]
- 65.Kurosawa Y, Hamaoka T, Katsumura T, Kuwamori M, Kimura N, Sako T, et al. Creatine supplementation enhances anaerobic ATP synthesis during a single 10 sec maximal handgrip exercise. Mol Cell Biochem. 2003;244:105–12. [PubMed] [Google Scholar]
- 66.Bemben MG, Lamont HS. Creatine supplementation and exercise performance: recent findings. Sports Med. 2005;35:107–25. doi: 10.2165/00007256-200535020-00002. [DOI] [PubMed] [Google Scholar]
- 67.Candow DG, Forbes SC, Little JP, Cornish SM, Pinkoski C, Chilibeck PD. Effect of nutritional interventions and resistance exercise on aging muscle mass and strength. Biogerontology. 2012;13:345–58. doi: 10.1007/s10522-012-9385-4. [DOI] [PubMed] [Google Scholar]
- 68.Safdar A, Yardley NJ, Snow R, Melov S, Tarnopolsky MA. Global and targeted gene expression and protein content in skeletal muscle of young men following short-term creatine monohydrate supplementation. Physiol Genomics. 2008;32:219–28. doi: 10.1152/physiolgenomics.00157.2007. [DOI] [PubMed] [Google Scholar]
- 69.Burke DG, Candow DG, Chilibeck PD, MacNeil LG, Roy BD, Tarnopolsky MA, et al. Effect of creatine supplementation and resistance-exercise training on muscle insulin-like growth factor in young adults. Int J Sport Nutr Exerc Metab. 2008;18:389–98. doi: 10.1123/ijsnem.18.4.389. [DOI] [PubMed] [Google Scholar]
- 70.Schiaffino S, Mammucari C. Regulation of skeletal muscle growth by the IGF1-Akt/PKB pathway: insights from genetic models. Skelet Muscle. 2011;1:4. doi: 10.1186/2044-5040-1-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Candow DG, Little JP, Chilibeck PD, Abeysekara S, Zello GA, Kazachkov M, et al. Low-dose creatine combined with protein during resistance training in older men. Med Sci Sports Exerc. 2008;40:1645–52. doi: 10.1249/MSS.0b013e318176b310. [DOI] [PubMed] [Google Scholar]
- 72.Sestili P, Martinelli C, Colombo E, Barbieri E, Potenza L, Sartini S, et al. Creatine as an antioxidant. Amino Acids. 2011;40:1385–96. doi: 10.1007/s00726-011-0875-5. [DOI] [PubMed] [Google Scholar]
- 73.Moller P, Bergstrom J, Furst P, Hellstrom K. Effect of aging on energy-rich phosphagens in human skeletal muscles. Clin Sci (Lond) 1980;58:553–5. doi: 10.1042/cs0580553. [DOI] [PubMed] [Google Scholar]
- 74.Brose A, Parise G, Tarnopolsky MA. Creatine supplementation enhances isometric strength and body composition improvements following strength exercise training in older adults. J Gerontol A Biol Sci Med Sci. 2003;58:11–9. doi: 10.1093/gerona/58.1.b11. [DOI] [PubMed] [Google Scholar]
- 75.Tarnopolsky M, Zimmer A, Paikin J, Safdar A, Aboud A, Pearce E, et al. Creatine monohydrate and conjugated linoleic acid improve strength and body composition following resistance exercise in older adults. PLoS ONE. 2007;2:e991. doi: 10.1371/journal.pone.0000991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Gotshalk LA, Volek JS, Staron RS, Denegar CR, Hagerman FC, Kraemer WJ. Creatine supplementation improves muscular performance in older men. Med Sci Sports Exerc. 2002;34:537–43. doi: 10.1097/00005768-200203000-00023. [DOI] [PubMed] [Google Scholar]
- 77.Candow DG, Chilibeck PD, Chad KE, Chrusch MJ, Davison KS, Burke DG. Effect of ceasing creatine supplementation while maintaining resistance training in older men. J Aging Phys Act. 2004;12:219–31. doi: 10.1123/japa.12.3.219. [DOI] [PubMed] [Google Scholar]
- 78.Harman D. Aging: a theory based on free radical and radiation chemistry. J Gerontol. 1956;11:298–300. doi: 10.1093/geronj/11.3.298. [DOI] [PubMed] [Google Scholar]
- 79.Chaput JP, Lord C, Cloutier M, Aubertin LM, Goulet ED, Rousseau S, et al. Relationship between antioxidant intakes and class I sarcopenia in elderly men and women. J Nutr Health Aging. 2007;11:363–9. [PubMed] [Google Scholar]
- 80.Howard C, Ferrucci L, Sun K, Fried LP, Walston J, Varadhan R, et al. Oxidative protein damage is associated with poor grip strength among older women living in the community. J Appl Physiol. 2007;103:17–20. doi: 10.1152/japplphysiol.00133.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Cesari M, Kritchevsky SB, Leeuwenburgh C, Pahor M. Oxidative damage and platelet activation as new predictors of mobility disability and mortality in elders. Antioxid Redox Signal. 2006;8:609–19. doi: 10.1089/ars.2006.8.609. [DOI] [PubMed] [Google Scholar]
- 82.Lauretani F, Semba RD, Bandinelli S, Dayhoff-Brannigan M, Lauretani F, Corsi AM, et al. Carotenoids as protection against disability in older persons. Rejuvenation Res. 2008;11:557–63. doi: 10.1089/rej.2007.0581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Lauretani F, Semba RD, Bandinelli S, Dayhoff-Brannigan M, Giacomini V, Corsi AM, et al. Low plasma carotenoids and skeletal muscle strength decline over 6 years. J Gerontol A Biol Sci Med Sci. 2008;63:376–83. doi: 10.1093/gerona/63.4.376. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Lauretani F, Semba RD, Bandinelli S, Ray AL, Guralnik JM, Ferrucci L. Association of low plasma selenium concentrations with poor muscle strength in older community-dwelling adults: the InCHIANTI Study. Am J Clin Nutr. 2007;86:347–52. doi: 10.1093/ajcn/86.2.347. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Semba RD, Blaum C, Guralnik JM, Moncrief DT, Ricks MO, Fried LP. Carotenoid and vitamin E status are associated with indicators of sarcopenia among older women living in the community. Aging Clin Exp Res. 2003;15:482–7. doi: 10.1007/BF03327377. [DOI] [PubMed] [Google Scholar]
- 86.Cesari M, Pahor M, Bartali B, Cherubini A, Penninx BW, Williams GR, et al. Antioxidants and physical performance in elderly persons: the Invecchiare in Chianti (InCHIANTI) study. Am J Clin Nutr. 2004;79:289–94. doi: 10.1093/ajcn/79.2.289. [DOI] [PubMed] [Google Scholar]
- 87.Handy DE, Loscalzo J. Redox regulation of mitochondrial function. Antioxid Redox Signal. 2012;16:1323–67. doi: 10.1089/ars.2011.4123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Ji LL. Antioxidant signaling in skeletal muscle: a brief review. Exp Gerontol. 2007;42:582–93. doi: 10.1016/j.exger.2007.03.002. [DOI] [PubMed] [Google Scholar]
- 89.Musaro A, Fulle S, Fano G. Oxidative stress and muscle homeostasis. Curr Opin Clin Nutr Metab Care. 2010;13:236–42. doi: 10.1097/MCO.0b013e3283368188. [DOI] [PubMed] [Google Scholar]
- 90.Safdar A, Hamadeh MJ, Kaczor JJ, Raha S, Debeer J, Tarnopolsky MA. Aberrant mitochondrial homeostasis in the skeletal muscle of sedentary older adults. PLoS ONE. 2010;5:e10778. doi: 10.1371/journal.pone.0010778. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Halliwell B. Free radicals and antioxidants - quo vadis? Trends Pharmacol Sci. 2011;32:125–30. doi: 10.1016/j.tips.2010.12.002. [DOI] [PubMed] [Google Scholar]
- 92.Ristow M, Zarse K, Oberbach A, Kloting N, Birringer M, Kiehntopf M, et al. Antioxidants prevent health-promoting effects of physical exercise in humans. Proc Natl Acad Sci U S A. 2009;106:8665–70. doi: 10.1073/pnas.0903485106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Gutteridge JM, Halliwell B. Antioxidants: Molecules, medicines, and myths. Biochem Biophys Res Commun. 2010;393:561–4. doi: 10.1016/j.bbrc.2010.02.071. [DOI] [PubMed] [Google Scholar]
- 94.Miller ER, III, Pastor-Barriuso R, Dalal D, Riemersma RA, Appel LJ, Guallar E. Meta-analysis: high-dosage vitamin E supplementation may increase all-cause mortality. Ann Intern Med. 2005;142:37–46. doi: 10.7326/0003-4819-142-1-200501040-00110. [DOI] [PubMed] [Google Scholar]
- 95.Bjelakovic G, Nikolova D, Gluud LL, Simonetti RG, Gluud C. Mortality in randomized trials of antioxidant supplements for primary and secondary prevention: systematic review and meta-analysis. JAMA. 2007;297:842–57. doi: 10.1001/jama.297.8.842. [DOI] [PubMed] [Google Scholar]
- 96.Bjelakovic G, Nikolova D, Gluud LL, Simonetti RG, Gluud C. Antioxidant supplements for prevention of mortality in healthy participants and patients with various diseases. Cochrane Database Syst Rev. 2012;3:CD007176. doi: 10.1002/14651858.CD007176.pub2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Fusco D, Colloca G, Lo Monaco MR, Cesari M. Effects of antioxidant supplementation on the aging process. Clin Interv Aging. 2007;2:377–87. [PMC free article] [PubMed] [Google Scholar]
- 98.Lauretani F, Maggio M, Pizzarelli F, Michelassi S, Ruggiero C, Ceda GP, et al. Omega-3 and renal function in older adults. Curr Pharm Des. 2009;15:4149–56. doi: 10.2174/138161209789909719. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Rantanen T, Guralnik JM, Foley D, Masaki K, Leveille S, Curb JD, et al. Midlife hand grip strength as a predictor of old age disability. JAMA. 1999;281:558–60. doi: 10.1001/jama.281.6.558. [DOI] [PubMed] [Google Scholar]
- 100.Stenholm S, Tiainen K, Rantanen T, Sainio P, Heliovaara M, Impivaara O, et al. Long-term determinants of muscle strength decline: prospective evidence from the 22-year mini-Finland follow-up survey. J Am Geriatr Soc. 2012;60:77–85. doi: 10.1111/j.1532-5415.2011.03779.x. [DOI] [PubMed] [Google Scholar]
- 101.Sayer AA, Syddall HE, Gilbody HJ, Dennison EM, Cooper C. Does sarcopenia originate in early life? Findings from the Hertfordshire cohort study. J Gerontol A Biol Sci Med Sci. 2004;59:M930–M934. doi: 10.1093/gerona/59.9.m930. [DOI] [PubMed] [Google Scholar]
- 102.Kuh D, Hardy R, Butterworth S, Okell L, Richards M, Wadsworth M, et al. Developmental origins of midlife physical performance: evidence from a British birth cohort. Am J Epidemiol. 2006;164:110–21. doi: 10.1093/aje/kwj193. [DOI] [PubMed] [Google Scholar]
- 103.Kuh D, Hardy R, Butterworth S, Okell L, Wadsworth M, Cooper C, et al. Developmental origins of midlife grip strength: findings from a birth cohort study. J Gerontol A Biol Sci Med Sci. 2006;61:702–6. doi: 10.1093/gerona/61.7.702. [DOI] [PubMed] [Google Scholar]
- 104.Robinson SM, Simmonds SJ, Jameson KA, Syddall HE, Dennison EM, Cooper C, et al. Muscle strength in older community-dwelling men is related to type of milk feeding in infancy. J Gerontol A Biol Sci Med Sci. 2012 doi: 10.1093/gerona/gls061. [DOI] [PubMed] [Google Scholar]
- 105.Wilson GJ, Wilson JM, Manninen AH. Effects of beta-hydroxy-beta-methylbutyrate (HMB) on exercise performance and body composition across varying levels of age, sex, and training experience: A review. Nutr Metab (Lond) 2008;5:1. doi: 10.1186/1743-7075-5-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Van Koevering M, Nissen S. Oxidation of leucine and alpha-ketoisocaproate to beta-hydroxy-beta-methylbutyrate in vivo. Am J Physiol. 1992;262:E27–E31. doi: 10.1152/ajpendo.1992.262.1.E27. [DOI] [PubMed] [Google Scholar]
- 107.Vukovich MD, Stubbs NB, Bohlken RM. Body composition in 70-year-old adults responds to dietary beta-hydroxy-beta-methylbutyrate similarly to that of young adults. J Nutr. 2001;131:2049–52. doi: 10.1093/jn/131.7.2049. [DOI] [PubMed] [Google Scholar]
- 108.Flakoll P, Sharp R, Baier S, Levenhagen D, Carr C, Nissen S. Effect of beta-hydroxy-beta-methylbutyrate, arginine, and lysine supplementation on strength, functionality, body composition, and protein metabolism in elderly women. Nutrition. 2004;20:445–51. doi: 10.1016/j.nut.2004.01.009. [DOI] [PubMed] [Google Scholar]
- 109.Baier S, Johannsen D, Abumrad N, Rathmacher JA, Nissen S, Flakoll P. Year-long changes in protein metabolism in elderly men and women supplemented with a nutrition cocktail of beta-hydroxy-beta-methylbutyrate (HMB), L-arginine, and L-lysine. JPEN J Parenter Enteral Nutr. 2009;33:71–82. doi: 10.1177/0148607108322403. [DOI] [PubMed] [Google Scholar]
- 110.Fuller JC, Jr, Baier S, Flakoll P, Nissen SL, Abumrad NN, Rathmacher JA. Vitamin D status affects strength gains in older adults supplemented with a combination of beta-hydroxy-beta-methylbutyrate, arginine, and lysine: a cohort study. JPEN J Parenter Enteral Nutr. 2011;35:757–62. doi: 10.1177/0148607111413903. [DOI] [PubMed] [Google Scholar]
- 111.Cynober L. Ornithine alpha-ketoglutarate as a potent precursor of arginine and nitric oxide: a new job for an old friend. J Nutr. 2004;134:2858S–62S. doi: 10.1093/jn/134.10.2858s. [DOI] [PubMed] [Google Scholar]
- 112.Walrand S. Ornithine alpha-ketoglutarate: could it be a new therapeutic option for sarcopenia? J Nutr Health Aging. 2010;14:570–7. doi: 10.1007/s12603-010-0109-7. [DOI] [PubMed] [Google Scholar]
- 113.Brocker P, Vellas B, Albarede JL, Poynard T. A two-centre, randomized, double-blind trial of ornithine oxoglutarate in 194 elderly, ambulatory, convalescent subjects. Age Ageing. 1994;23:303–6. doi: 10.1093/ageing/23.4.303. [DOI] [PubMed] [Google Scholar]
- 114.Segaud F, Combaret L, Neveux N, Attaix D, Cynober L, Moinard C. Effects of ornithine alpha-ketoglutarate on protein metabolism in Yoshida sarcoma-bearing rats. Clin Nutr. 2007;26:624–30. doi: 10.1016/j.clnu.2007.05.001. [DOI] [PubMed] [Google Scholar]
- 115.Donati L, Ziegler F, Pongelli G, Signorini MS. Nutritional and clinical efficacy of ornithine alpha-ketoglutarate in severe burn patients. Clin Nutr. 1999;18:307–11. doi: 10.1054/clnu.1999.0040. [DOI] [PubMed] [Google Scholar]
- 116.Wernerman J, Hammarqvist F, von der DA, Vinnars E. Ornithine-alpha-ketoglutarate improves skeletal muscle protein synthesis as assessed by ribosome analysis and nitrogen use after surgery. Ann Surg. 1987;206:674–8. doi: 10.1097/00000658-198711000-00020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Fetterman JW, Jr, Zdanowicz MM. Therapeutic potential of n-3 polyunsaturated fatty acids in disease. Am J Health Syst Pharm. 2009;66:1169–79. doi: 10.2146/ajhp080411. [DOI] [PubMed] [Google Scholar]
- 118.Robinson SM, Jameson KA, Batelaan SF, Martin HJ, Syddall HE, Dennison EM, et al. Diet and its relationship with grip strength in community-dwelling older men and women: the Hertfordshire cohort study. J Am Geriatr Soc. 2008;56:84–90. doi: 10.1111/j.1532-5415.2007.01478.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Smith GI, Atherton P, Reeds DN, Mohammed BS, Rankin D, Rennie MJ, et al. Omega-3 polyunsaturated fatty acids augment the muscle protein anabolic response to hyperinsulinaemia-hyperaminoacidaemia in healthy young and middle-aged men and women. Clin Sci (Lond) 2011;121:267–78. doi: 10.1042/CS20100597. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Cornish SM, Chilibeck PD. Alpha-linolenic acid supplementation and resistance training in older adults. Appl Physiol Nutr Metab. 2009;34:49–59. doi: 10.1139/H08-136. [DOI] [PubMed] [Google Scholar]
- 121.Food and Nutrition Board. Dietary Reference Intakes (DRIs): Recommended Intakes for Individuals. Institute of Medicine, National Academies; 2004. [Accessed 28 September 2012]. http://www.iom.edu/Activities/Nutrition/SummaryDRIs/~/media/Files/Activity%20Files/Nutrition/DRIs/5_Summary%20Table%20Tables%201-4.pdf. [Google Scholar]
- 122.Jager S, Trojan H, Kopp T, Laszczyk MN, Scheffler A. Pentacyclic triterpene distribution in various plants - rich sources for a new group of multi-potent plant extracts. Molecules. 2009;14:2016–31. doi: 10.3390/molecules14062016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Kunkel SD, Suneja M, Ebert SM, Bongers KS, Fox DK, Malmberg SE, et al. mRNA expression signatures of human skeletal muscle atrophy identify a natural compound that increases muscle mass. Cell Metab. 2011;13:627–38. doi: 10.1016/j.cmet.2011.03.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Kunkel SD, Elmore CJ, Bongers KS, Ebert SM, Fox DK, Dyle MC, et al. Ursolic acid increases skeletal muscle and brown fat and decreases diet-induced obesity, glucose intolerance and fatty liver disease. PLoS ONE. 2012;7:e39332. doi: 10.1371/journal.pone.0039332. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Lundberg JO, Gladwin MT, Ahluwalia A, Benjamin N, Bryan NS, Butler A, et al. Nitrate and nitrite in biology, nutrition and therapeutics. Nat Chem Biol. 2009;5:865–9. doi: 10.1038/nchembio.260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Lundberg JO. Nitrate transport in salivary glands with implications for NO homeostasis. Proc Natl Acad Sci U S A. 2012;109:13144–5. doi: 10.1073/pnas.1210412109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Larsen FJ, Weitzberg E, Lundberg JO, Ekblom B. Dietary nitrate reduces maximal oxygen consumption while maintaining work performance in maximal exercise. Free Radic Biol Med. 2010;48:342–7. doi: 10.1016/j.freeradbiomed.2009.11.006. [DOI] [PubMed] [Google Scholar]
- 128.Larsen FJ, Schiffer TA, Borniquel S, Sahlin K, Ekblom B, Lundberg JO, et al. Dietary inorganic nitrate improves mitochondrial efficiency in humans. Cell Metab. 2011;13:149–59. doi: 10.1016/j.cmet.2011.01.004. [DOI] [PubMed] [Google Scholar]
- 129.Marzetti E, Hwang JC, Lees HA, Wohlgemuth SE, Dupont-Versteegden EE, Carter CS, et al. Mitochondrial death effectors: relevance to sarcopenia and disuse muscle atrophy. Biochim Biophys Acta. 2010;1800:235–44. doi: 10.1016/j.bbagen.2009.05.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Carter CS, Giovannini S, Seo DO, Dupree J, Morgan D, Chung HY, et al. Differential effects of enalapril and losartan on body composition and indices of muscle quality in aged male Fischer 344 x Brown Norway rats. Age (Dordr) 2011;33:167–83. doi: 10.1007/s11357-010-9196-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Marzetti E, Calvani R, Dupree J, Lees HA, Giovannini S, Seo DO, et al. Late-life enalapril administration induces nitric oxide-dependent and independent metabolic adaptations in the rat skeletal muscle. Age (Dordr) 2012 doi: 10.1007/s11357-012-9428-4. In press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Marzetti E, Lawler JM, Hiona A, Manini T, Seo AY, Leeuwenburgh C. Modulation of age-induced apoptotic signaling and cellular remodeling by exercise and calorie restriction in skeletal muscle. Free Radic Biol Med. 2008;44:160–8. doi: 10.1016/j.freeradbiomed.2007.05.028. [DOI] [PubMed] [Google Scholar]
- 133.Landi F, Zuccala G, Gambassi G, Incalzi RA, Manigrasso L, Pagano F, et al. Body mass index and mortality among older people living in the community. J Am Geriatr Soc. 1999;47:1072–6. doi: 10.1111/j.1532-5415.1999.tb05229.x. [DOI] [PubMed] [Google Scholar]
- 134.Mercken EM, Carboneau BA, Krzysik-Walker SM, de CR. Of mice and men: the benefits of caloric restriction, exercise, and mimetics. Ageing Res Rev. 2012;11:390–8. doi: 10.1016/j.arr.2011.11.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Narkar VA, Downes M, Yu RT, Embler E, Wang YX, Banayo E, et al. AMPK and PPARdelta agonists are exercise mimetics. Cell. 2008;134:405–15. doi: 10.1016/j.cell.2008.06.051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Momken I, Stevens L, Bergouignan A, Desplanches D, Rudwill F, Chery I, et al. Resveratrol prevents the wasting disorders of mechanical unloading by acting as a physical exercise mimetic in the rat. FASEB J. 2011;25:3646–60. doi: 10.1096/fj.10-177295. [DOI] [PubMed] [Google Scholar]
- 137.Booth FW, Laye MJ. Lack of adequate appreciation of physical exercise’s complexities can pre-empt appropriate design and interpretation in scientific discovery. J Physiol. 2009;587:5527–39. doi: 10.1113/jphysiol.2009.179507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Lewis GD, Farrell L, Wood MJ, Martinovic M, Arany Z, Rowe GC, et al. Metabolic signatures of exercise in human plasma. Sci Transl Med. 2010;2:33ra37. doi: 10.1126/scitranslmed.3001006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Fujimura KE, Slusher NA, Cabana MD, Lynch SV. Role of the gut microbiota in defining human health. Expert Rev Anti Infect Ther. 2010;8:435–54. doi: 10.1586/eri.10.14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Human Microbiome Project Consortium. Structure, function and diversity of the healthy human microbiome. Nature. 2012;486:207–14. doi: 10.1038/nature11234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Kinross JM, Darzi AW, Nicholson JK. Gut microbiome-host interactions in health and disease. Genome Med. 2011;3:14. doi: 10.1186/gm228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Calvani R, Miccheli A, Capuani G, Tomassini MA, Puccetti C, Delfini M, et al. Gut microbiome-derived metabolites characterize a peculiar obese urinary metabotype. Int J Obes (Lond) 2010;34:1095–8. doi: 10.1038/ijo.2010.44. [DOI] [PubMed] [Google Scholar]
- 143.Vanhoutte T, Huys G, Brandt E, Swings J. Temporal stability analysis of the microbiota in human feces by denaturing gradient gel electrophoresis using universal and group-specific 16S rRNA gene primers. FEMS Microbiol Ecol. 2004;48:437–46. doi: 10.1016/j.femsec.2004.03.001. [DOI] [PubMed] [Google Scholar]
- 144.Biagi E, Candela M, Fairweather-Tait S, Franceschi C, Brigidi P. Aging of the human metaorganism: the microbial counterpart. Age (Dordr) 2012;34:247–67. doi: 10.1007/s11357-011-9217-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Claesson MJ, Jeffery IB, Conde S, Power SE, O’Connor EM, Cusack S, et al. Gut microbiota composition correlates with diet and health in the elderly. Nature. 2012;488:178–84. doi: 10.1038/nature11319. [DOI] [PubMed] [Google Scholar]
- 146.van Duynhoven J, Vaughan EE, Jacobs DM, Kemperman RA, van Velzen EJ, Gross G, et al. Metabolic fate of polyphenols in the human superorganism. Proc Natl Acad Sci U S A. 2011;108 (Suppl 1):4531–8. doi: 10.1073/pnas.1000098107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.FAO/WHO. Health and nutritional properties of probiotics in food including powder milk with live lactic acid bacteria: report of a joint FAO/WHO expert consultation. Córdoba, Argentina: Oct, 2001. [Accessed 28 September 2012]. http://www.who.int/foodsafety/publications/fs_management/en/probiotics.pdf. [Google Scholar]
- 148.Gibson GR, Roberfroid MB. Dietary modulation of the human colonic microbiota: introducing the concept of prebiotics. J Nutr. 1995;125:1401–12. doi: 10.1093/jn/125.6.1401. [DOI] [PubMed] [Google Scholar]
- 149.Tiihonen K, Ouwehand AC, Rautonen N. Human intestinal microbiota and healthy ageing. Ageing Res Rev. 2010;9:107–16. doi: 10.1016/j.arr.2009.10.004. [DOI] [PubMed] [Google Scholar]
- 150.Chan SY, Loscalzo J. The emerging paradigm of network medicine in the study of human disease. Circ Res. 2012;111:359–74. doi: 10.1161/CIRCRESAHA.111.258541. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Nicholson JK, Holmes E, Wilson ID. Gut microorganisms, mammalian metabolism and personalized health care. Nat Rev Microbiol. 2005;3:431–8. doi: 10.1038/nrmicro1152. [DOI] [PubMed] [Google Scholar]
- 152.Chen R, Mias GI, Li-Pook-Than J, Jiang L, Lam HY, Chen R, et al. Personal omics profiling reveals dynamic molecular and medical phenotypes. Cell. 2012;148:1293–307. doi: 10.1016/j.cell.2012.02.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Jones DP, Park Y, Ziegler TR. Nutritional metabolomics: progress in addressing complexity in diet and health. Annu Rev Nutr. 2012;32:183–202. doi: 10.1146/annurev-nutr-072610-145159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Ferguson JF. Omics gets personal: integrative profiling of health and disease. Circ Cardiovasc Genet. 2012;5:381–2. doi: 10.1161/CIRCGENETICS.112.963801. [DOI] [PubMed] [Google Scholar]
- 155.Nicholson JK, Wilson ID. Opinion: understanding ‘global’ systems biology: metabonomics and the continuum of metabolism. Nat Rev Drug Discov. 2003;2:668–76. doi: 10.1038/nrd1157. [DOI] [PubMed] [Google Scholar]