Abstract
We investigated the influence of altered muscle duty cycle on the performance decrements and neuromuscular responses occurring during constant-load, fatiguing bouts of knee extension exercise. We experimentally altered the durations of the muscularly inactive portion of the limb movement cycle and hypothesized that greater relative durations of inactivity within the same movement task would 1) reduce the rates and extent of muscle performance loss and 2) increase the forces necessary to trigger muscle fatigue. In each condition (duty cycle = 0.6 and 0.3), male subjects [age = 25.9 ± 2.0 yr (SE); mass = 85.4 ± 2.6 kg], completed 9–11 exhaustive bouts of two-legged knee extension exercise, at force outputs that elicited failure between 4 and 290 s. The novel duty cycle manipulation produced two primary results; first, we observed twofold differences in both the extent of muscle performance lost (DC0.6 = 761 ± 35 N vs. DC0.3 = 366 ± 49 N) and the time course of performance loss. For example, exhaustive trials at the midpoint of these force ranges differed in duration by more than 30 s (t0.6 = 36 ± 2.6 vs. t0.3 = 67 ± 4.3 s). Second, both the minimum forces necessary to exceed the peak aerobic capacity and initiate a reliance on anaerobic metabolism, and the forces necessary to elicit compensatory increases in electromyogram activity were 300% greater in the lower vs. higher duty cycle condition. These results indicate that the fatigue-induced compensatory behavior to recruit additional motor units is triggered by a reliance on anaerobic metabolism for ATP resynthesis and is independent of the absolute level or fraction of the maximum force produced by the muscle.
Keywords: muscle fatigue, neuromuscular compensation, performance-duration relationship
it has long been recognized that there is a duration-dependent relationship between the levels of muscular force and mechanical power that can be maintained in exercise performed until failure. Between exhaustive efforts that elicit failure within seconds to several minutes, the losses in muscular force and power output are exponential; however, beyond this initial period of rapid decrements, similar levels of muscular performance can be sustained for hours (19, 27, 31, 40, 45). For example, the current human record running speed (29) is 185% faster than the speed that can be sustained for an 8-min run, yet extending the duration from several minutes to 2 h results in only an additional 8% loss in running speed (32). Thus, within the endurance portion of this relationship, performance levels are essentially sustainable, a recognition that has long been attributed to the reliance on aerobic metabolism for the ATP resynthesis necessary to support the muscular contraction. In contrast, comparatively little is known about the physiological mechanisms determining the much greater extent, and more rapid time course, of muscle performance loss that occurs during the nonsustainable, short-duration phase of this general relationship.
For whole body modes of exercise eliciting failure between 3 and 300 s, the relative rates and extent of performance loss are defined by highly accurate, individual performance-duration relationships, determined from three physiologically based components (13, 14). Two of these measures are subject-specific and define the upper and lower limits of each individual's short-duration performance range; these measures are the mode-specific musculoskeletal maximum performance (i.e., speed, power, force) sustained for 3 s or less and the upper level of performance that can be supported for roughly 5–8 min. Efforts of this duration rely primarily, but not exclusively, on aerobic metabolism for ATP resynthesis. Direct measures of cellular metabolism from exhaustive efforts of similar durations indicate that aerobic metabolism accounts for 91% of the total metabolic energy liberated during these trials (23). The third factor is an empirically derived exponential time constant describing the rates of muscle performance loss between the upper and lower end points of the nonsustainable performance range. The exponential time constant appears to be similar among different individuals (30, 51) but varies between modes of exercise (52). The between-mode, rather than between-individual, differences implicate the mechanics of the muscle contraction as an important factor dictating the time course of performance loss and the development of muscle fatigue during short-duration exhaustive exercise. However, the empirical evidence necessary to evaluate the influence of altered mechanical factors, such as relative durations of activity and inactivity, contraction frequency, and shortening velocity, on the extent of force loss and rates of performance decrements occurring with increments in the duration of muscular activity during exhaustive dynamic tasks is not yet available. These data have both experimental and applied relevance because the times to failure at relative levels of force application are a widely used measure in studies of muscle fatigue, and prolonging high force efforts is the objective of numerous competitive events and training methods used to induce neuromuscular adaptation.
Therefore, we developed an experimental design to manipulate the mechanics of the muscle contraction by altering the duration of the inactive period within the movement cycle (i.e., the duty cycle), while subjects performed constant-load, dynamic knee extension exercise. The goal was to alter the time course and extent of muscle performance loss occurring in exhaustive dynamic bouts of the same muscular task. We devised a custom knee extension ergometer (after Ref. 3) that permitted subjects to provide positive physical work (J) by applying force against the ergometer throughout the limb's extension. We anticipated that the mechanical power outputs sustained at a subject's peak rate of oxygen uptake would be similar between the experimental conditions, because mechanical power does not appear to vary substantially with limb cadence at this intensity (5, 49, 54). The physical consequence (Eq. 1) of similar mechanical power outputs with different periods of muscular inactivity provides an experimental opportunity to markedly alter the level of muscular force necessary to exceed the upper level of performance supported primarily by aerobic metabolism and elicit a transition to a net reliance on anaerobic metabolism for force production.
Thus, the experimental manipulation used here permits a test of the long-standing paradigm that the onset of muscle fatigue and the associated motor responses are directly linked to the level of muscle force applied (21). This expectation provides a primary rationale for the near-universal practice within the neuromuscular fatigue literature of referencing muscle performance to the force elicited during a maximum voluntary contraction (i.e., MVC). Here, we used surface electromyography (EMG) to identify the presence of impaired muscular force production (7, 20, 24, 37, 41) during both sustainable and nonsustainable knee extension exercise. We considered a progressive increase in EMG amplitude occurring while muscle force production remained constant as the indicator of a compensatory response by the nervous system to maintain force output in the presence of fatigue (1, 10, 17, 26). This response is essentially absent (11, 15, 48) during muscle contractions performed for long durations at relatively low levels of force production that are supported primarily by the body's aerobic system. Therefore, we hypothesized that muscle performance loss and the compensatory fatigue response occur independently of muscle force and are triggered when aerobic metabolism can no longer support the contraction and a net reliance on anaerobic metabolism is required. We specifically expected to observe 1) lower levels of force output necessary to elicit the onset of fatigue-induced compensatory neuromuscular activity, and 2) greater relative rates of performance loss between equivalent levels of muscle force production in the higher vs. lower duty cycle condition, despite performing the same motor task.
MATERIALS AND METHODS
Subjects.
Seven male subjects without a history of surgical invasion to the leg [age = 25.9 ± 2.0 yr (SE); mass = 85.4 ± 2.6 kg], volunteered and provided their written informed consent to participate in this study, which was preapproved by the local Institutional Review Board. Six of the seven subjects were actively engaged in either endurance or resistance training at the time of the study. A summary of their relevant physiological and performance characterstics appears in Table 1.
Table 1.
Physiological performance characteristics of the subjects
| Duty Cycle |
||
|---|---|---|
| Variable | 0.3 | 0.6 |
| Pmech max,W | 152.8 ± 12.9* | 409.9 ± 8.3 |
| Fmech max, N | 1102.2 ± 69.3 | 1007.3 ± 29.0 |
| Paer, W | 95.1 ± 5.3 | 89.6 ± 7.3 |
| Faer, N | 735.7 ± 37.0* | 246.3 ± 17.8 |
| V̇o2peak, l O2/min | 2.33 ± 0.16* | 2.10 ± 0.16 |
Values are expressed as means ± SE. The mechanical maximums (mech max), were measured from three successive contractions while subjects provided a maximum effort and adhered to the experimental cadences. The aerobic maximums (aer) were the minimum pedal forces and power outputs necessary to elicit the condition specific V̇o2peak.
Significant difference between the two duty cycles (P < 0.05).
Experimental protocol.
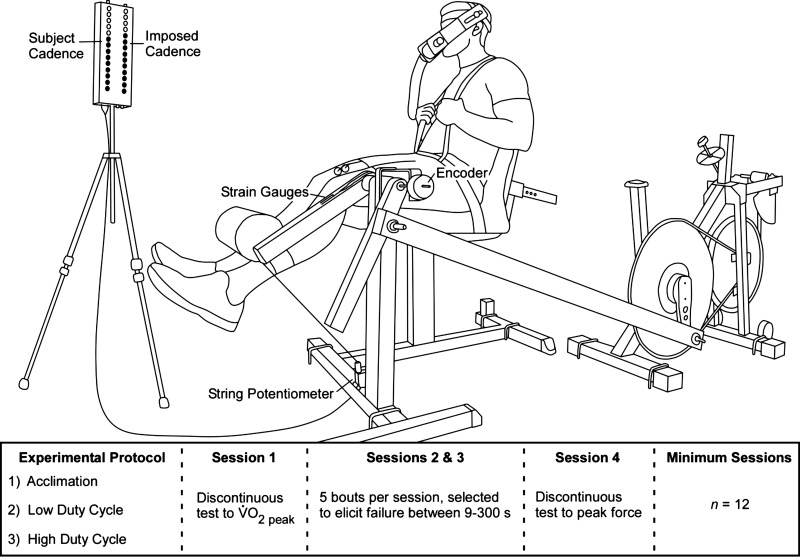
We employed a two-legged knee extension protocol performed at different duty cycles and force levels to test our hypotheses of altered time courses of performance loss and thresholds necessary to initiate the compensatory neuromuscular response. The variation in duty cycle—here the ratio of the durations of muscle force application to the entire movement cycle—was accomplished by using a visual feedback array (6, 25) consisting of two columns of light-emitting diodes (LED). One of the columns was driven by an adjustable timing circuit that could be altered to achieve the desired active vs. inactive durations. The second column was driven by a string potentiometer (SP1-50; Celesco, Chattsworth, CA) connected to the ergometer arm and provided feedback by illustrating the subject's actual displacement. Subjects were instructed to match the progress displayed on their array column to the imposed cadence of the other column (Fig. 1).
Fig. 1.
The custom ergometer assessed force application via strain gauges and limb excursions from an incremental encoder. The LED array provided visual feedback to maintain the imposed movement cadences. Subjects completed three experimental conditions of two-legged knee extension exercise; each condition required four laboratory visits as described in methods.
Subjects typically completed three different duty cycle conditions, which required a minimum of 12 laboratory visits. We administered the conditions as follows: subjects completed sessions at an intermediate cadence for acclimation (4) and subsequently completed a low and a high duty cycle manipulation. Two of the seven subjects completed the acclimation and a single duty cycle manipulation; as a result, data from six subjects are included in both the low and the high duty cycle data sets. Each duty cycle condition required a minimum of four laboratory visits (Fig. 1). During the first visit per condition, subjects completed a progressive, force-incremented, discontinuous test to determine the minimum force level necessary to elicit their V̇o2 peak. Sessions two and three, in each duty cycle condition, consisted of a standardized warm-up (172 ± 16 N), followed by five, randomly administered, constant-load exhaustive trials at a range of forces chosen to elicit failure between 4 and 300 s. During these sessions, subjects were allowed to take as much rest between the trials as they deemed necessary to be fully recovered but were required to wait at least 15 min. The last lab visit for each duty cycle condition consisted of a standardized warm-up, followed by a progressive discontinuous test to determine the subject's maximum musculoskeletal force output for three consecutive contractions while adhering to the specified cadence. Given the considerable number of laboratory visits required, we monitored the subjects for a training effect. A training effect would result in improved musculoskeletal or aerobic performance throughout the course of the subject's participation. This was not observed.
Custom knee extension ergometer.
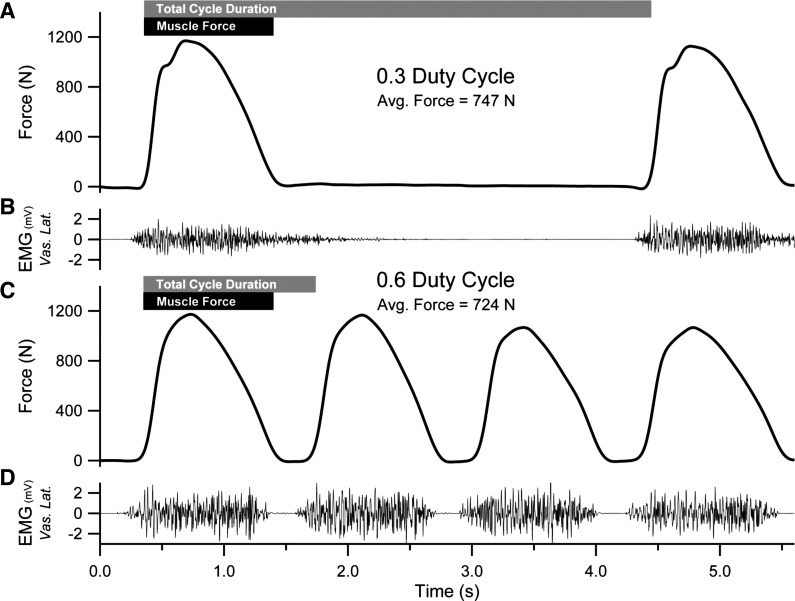
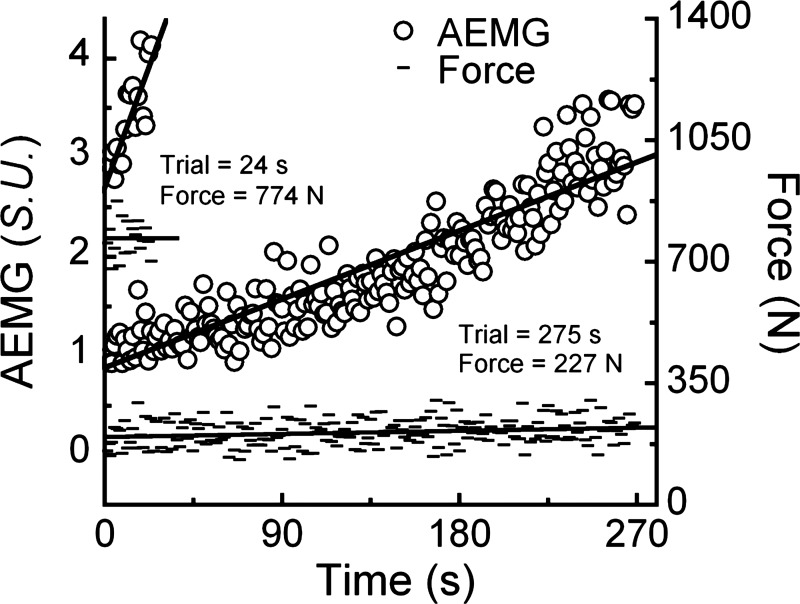
The custom knee extension ergometer consisted of an instrumented knee extension machine (GCEC340; Body-Solid, Forest Park, IL) connected to the pedal of a cycle ergometer (828E; Monark, Varberg, Sweden) by a hollow metal tube (3). The knee extension machine and cycle ergometer were secured to a stage to maintain proper alignment between the equipment. The connecting tube (l = 1.63 m) was attached to the ergometer's load arm with a distance of 0.108 m from the ergometer's axis of rotation. This ensured that a full knee excursion by the subject produced an angular displacement at the pedal that was small enough to achieve passive return. To decrease the reliance on the cycle ergometer's nylon brake, we augmented the standard 54:14 gear ratio with a 130-tooth sprocket attached to the pedal crank to create a gear ratio of 130:14. These modifications produced patterns of force application throughout the knee extension cycle that resembled those of typical whole body activities (Fig. 2) and fundamentally differ from previous studies using a similarly designed knee extension ergometer (e.g., Fig. 2 in Ref. 3).
Fig. 2.
Original recordings of the forces applied against the ergometer (A and C), and EMG (B and D) obtained during constant-load knee extension trials performed at the 0.6 and 0.3 duty cycle conditions. In these trials, the average forces were similar; however, the time to failure differed by nearly 4 min (20 vs. 253 s) in the 0.6 vs. 0.3 conditions, respectively. Gray (total movement cycle) and black (muscle activity) bars indicate the durations of muscle activity vs. inactivity created by the experimental manipulation.
The forces applied by the legs were measured from four single-element strain gauges (FLA-30-11; Tokyo Sokki Kenkyujo, Tokyo, Japan) arranged in a full wheatstone bridge configuration. The gauges were mounted on the tensile and compressive surfaces and aligned in parallel 0.051 m below the axis of rotation of the ergometer's extension arm. The relationship between force applied to the ergometer extension arm and the resulting resistance change within the strain gauge circuit was calibrated with multiple 11-point calibrations that were linear across the entire range of applied forces (R2> 0.99). To ensure the forces measured by the strain gauges were due to the action of the extensor muscles of the leg, subjects were informed that they could not grasp any part of the ergometer with their hands. We minimized possible movements of the upper body by securing subjects to the seat of the ergometer at the shoulders and hips with an adjustable four-point restraint system. In addition, the lower limbs were secured to the ergometer proximal to the ankle, which ensured repeatable placement of the shank against the ergometer during the knee extension phase.
Angular displacement was measured with an incremental rotary encoder (DFS60E-TBAM00360; Sick Stegmann, Dayton, OH) mounted to the axis of rotation of the ergometer arm; the encoder had a resolution of 360 counts per revolution. The analog signals from the encoder, strain gauges, and LED array were digitized at 3,003 Hz (Digidata 1440; Molecular Devices, Sunnyvale, CA) and recorded to a personal computer for subsequent signal conditioning and analysis (Igor 6.2; Wavemetrics; Portland, OR). The power output applied to the knee extension arm during each contraction was calculated from the measures of applied force and knee extension velocity in accordance with
| (1) |
where F is the force applied by the legs to the ergometer, r is the moment arm of the ergometer, θ is the angular displacement undergone during the extension, and s is the duration of the entire movement cycle.
Imposed cadence and trial durations.
We imposed the experimental cadences by altering the ratio of the knee extension duration to the limb replacement duration that was visible to the subjects on the previously described LED feedback array. Subjects were instructed to match their progress to the cadence displayed by the sequential illumination and darkening of the column driven by the adjustable timing circuit. The target duration of the knee extension phase was 1 s throughout this study. The limb replacement phase was programmed to have the following durations: 1 s for the acclimation condition (0.5 duty cycle), 0.33 s for the high duty cycle condition (0.75 duty cycle), and 3 s for the low duty cycle condition (0.25 duty cycle). Because of a slight mismatch between the imposed movement periods and the subject-achieved durations, likely due to the 6.2° interdiode resolution of the array, the mean performed duty cycles of the high and low conditions were 0.63 ± 0.01 (n = 61 trials) and 0.30 ± 0.01 (n = 62 trials), respectively. Hereafter, we refer to the high and low duty cycle conditions as the 0.6 and 0.3 conditions, respectively. Exhaustive trials were terminated when subjects could not match the cadence imposed by the LED feedback array for 5 s, despite receiving warnings of the mismatch and putting forth a maximum effort to regain the cadence.
Oxygen uptake.
Rates of oxygen uptake (V̇o2) were obtained from the subject's expired gas and analyzed with a computerized metabolic system (TrueOne 2400, ParvoMedics, Sandy, UT). During the initial session of each duty cycle condition, subjects completed a progressive, discontinuous, force incremented, knee extension test that consisted of 5-min bouts of knee extension, interspersed with rest periods of at least 3 min during the initial bouts, and until full recovery in the bouts approaching the aerobic limit. Peak rates of oxygen uptake for each duty cycle condition were the highest minute value obtained during either the final 5-min bout administered in the initial session of each condition or from an exhaustive effort eliciting failure in greater than 4 min, obtained in one of the subsequent sessions.
During the exhaustive knee extension trials, expired gases were collected in a series of Douglas bags. Aliquots from each bag were analyzed for O2 and CO2 fractions using the metabolic system's analyzers. Expired volumes were determined by using a dry gas meter (CD4, model 4015; Parkinson-Cowan, C. Poe, Houston, TX) with simultaneous measures of the gas temperature (no. 4015; Control Company, Friendswood, TX). Rates of oxygen uptake were determined in accordance with Consalazio et al. (18).
Power output and applied force.
The reported measures of force and power for each trial were the average of all contractions performed within the particular knee extension bout. Thus, the values of Paer and Faer were those measured during the same trial that elicited the peak rate of oxygen uptake as described above. Forces and power outputs greater than those eliciting the aerobic maximums require a net reliance on anaerobic metabolism for the additional force production. Existing evidence suggests the transition to a reliance on anaerobic metabolism occurs at force levels and at relative intensities less than the V̇o2 peak (34, 42). However, we use this conservative simplification because the anaerobic contribution to force production during in vivo muscle activity cannot be measured directly, whereas applied forces greater than those necessary to elicit the aerobic peak are measurable and unambiguously indicate an anaerobic contribution to force production (14).
Peak mechanical power and force (Pmech max and Fmech max).
The maximum muscular performances were determined from the discontinuous test to peak force and were the greatest measures of force and power achieved in three consecutive contractions while matching the imposed cadence for each duty cycle condition.
Electromyography.
Bipolar surface EMG electrodes were placed on the skin of the right and left legs overlying the muscle bellies of the distal portions of the vastus lateralis and the vastus medialis with an interelectrode distance of 0.025 m. The reference electrode was placed on the anterior aspect of the iliac crest. Repeatable electrode placement between data collection sessions was achieved by marking the location of each electrode with indelible ink during the subject's first and subsequent visits to the laboratory. Subjects were instructed to reapply the ink as necessary throughout their participation in the study. To reduce electrical impedance, the skin was shaved, lightly abraded, and cleaned with alcohol prior to electrode placement. If the interelectrode impedance exceeded 3,000 Ω, the electrodes were removed, and the skin was cleaned prior to reinstrumentation with new electrodes. The surface EMG signals were amplified (500×) and filtered (3-3,000 Hz half-amplitude band pass and 60-Hz notch filter) using four amplifiers (P511; Grass-Telefactor, Warwick, RI). The analog outputs from the amplifiers were digitized at 3,003 Hz and recorded throughout the duration of each knee extension trial by a computer using the previously described A/D converter. The recorded EMG data were rectified and averaged on a per contraction basis (AEMG). EMG records from the exhaustive trials (n = 484) were screened and excluded from further analysis if there were signs of flawed electrical connectivity (n = 10) or if the AEMG data decreased throughout the course of the trial (n = 19). This occurred for unknown reasons in 3.9% of the acquired records (i.e., number of trials × muscles interrogated).
Data analysis.
For the 0.6 and 0.3 duty cycle conditions, we calculated the exponential time constants describing the decrements in force (kF0.6, kF0.3) and power (kP0.6, kP0.3) that occurred with increments in effort duration from the 9–11 exhaustive trials administered to each subject. These calculations involved a residual minimizing, iterative procedure, to solve for the exponential time constants, k, based on the measured terms in the following equations:
| (2) |
| (3) |
where P(t) and F(t) are the measures of power and force for each of the 9–11 exhaustive trials of duration t, and Paer, Faer, Pmech max, and Fmech max are the levels of power and force output eliciting the V̇o2 peak and the mechanical burst maximums, respectively, from each subject in the two duty cycle conditions. The e−kt term describes the time course of relative performance loss.
To quantitatively evaluate whether the cumulative duration of muscle force production sets a common time course of muscle fatigue and performance loss, we adjusted the force-duration relationship to reflect only the time that the muscle was applying force against the ergometer. The cumulative duration of muscle force application was obtained from the product of the trial duration and the mean duty cycle achieved in each condition. The mean duty cycle was obtained from all of the exhaustive trials completed at either the low or high duty cycle manipulations, i.e., 0.63 (n = 61 trials) and 0.30 (n = 62 trials).
We calculated the durations that would elicit a level of muscular performance representing the 50% midpoint (i.e., the half-life) of the performance range that exists between the mechanical maximums (Pmech max and Fmech max) and the performance level supported at the aerobic peaks (Paer and Faer), in accordance with
| (4) |
where t1/2 is the duration of an exhaustive trial at the midpoint of the performance range, ln 0.5 is the natural log of 50%, and k is the relevant exponential time constant (e.g., kF0.6, kF0.3) describing the time course of the loss in muscle force with increments in the effort duration.
Statistics.
Data are reported throughout the article as means ± SE. Differences between the means for the two duty cycles were tested with a paired-samples t-test. The a priori level of significance for the statistical tests was set at P < 0.05.
RESULTS
Duration of muscle force application.
We administered a total of 508 bouts of knee extension exercise and analyzed 14,889 muscle contractions from the the 0.6 duty cycle condition and 5,991 muscle contractions from the 0.3 duty cycle condition. The greater number of contractions in the 0.6 duty cycle was due to the greater limb movement frequency of this condition, with subjects matching the frequencies imposed by the LED array in each condition, 0.76 Hz and 0.25 Hz (Fig. 2). However, the mean duration of the active phase of the cycle varied somewhat from the imposed cadences in both conditions. In the 0.6 duty cycle, the durations of force application were less than those indicated by the 1-s extension phase of the LED array (0.83 ± 0.015 s). In contrast, while completing the 0.3 duty cycle trials, subjects used contractile durations that were slightly longer than those of the imposed cadence (1.20 ± 0.039 s).
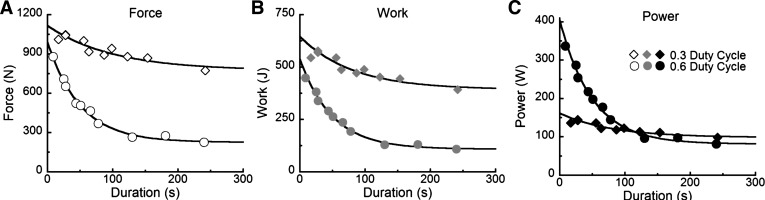
Subjects used similar knee extension excursions in the 0.6 and 0.3 duty cycle conditions; these were 76.3 ± 1.0° and 77.9 ± 0.5° (P = 0.49) for the exhaustive trials of similar force output, respectively. Therefore, the values of physical work (J) performed per contraction (35) parallel the forces applied during the exhaustive trials administered (Fig. 3). Thus, in accordance with our experimental design, the between-duty cycle differences in muscle force account for the altered levels of work performed rather than the more complicated scenario occuring if both force application and muscle length change were to vary.
Fig. 3.
Performance-duration relationships for the average per contraction levels of force (A), work (B), and mechanical power (C) in relation to the exhaustive trial durations performed at the 0.6 and 0.3 duty cycle conditions, for a representative subject. Curves were fit in accordance with Eqs. 2 and 3 using measured values of Faer,Waer, Paer, Fmech max, Wmech max and Pmech max for this subject. The performance decrements in force and work occurred with essentially identical time courses due to the experimental design that maintained angular displacement, thus requiring work to vary in response to force. R2 values for the curves illustrated were F0.3 = 0.87, F0.6 = 0.99, W0.3 = 0.85, W0.6 = 0.99, P0.3 = 0.85, and P0.6 = 0.99.
Power output and applied force.
The mechanical power outputs that elicited the peak rates of oxygen uptake (V̇o2 peak) were obtained from trials lasting at least 240 s (0.6 duty cycle = 258 ± 7 s; 0.3 duty cycle = 261 ± 9 s), and were 90 ± 7 W and 95 ± 5 W for the 0.6 and 0.3 duty cycle conditions, respectively. The similar power outputs at the aerobic peak (P = 0.57) were produced with applied forces that differed threefold, and were 246 ± 18 N and 736 ± 37 N (P < 0.001) for the 0.6 and 0.3 conditions (Figs. 3 and 4), respectively. The mechanical outputs provided by the working muscle elicited rates of oxygen uptake that differed by 10% between the conditions and were 2.10 ± 0.16 l O2/min for the 0.6 duty cycle and 2.33 ± 0.16 l O2/min for the 0.3 duty cycle (P = 0.01). When expressed mass specifically, the values were 25.3 ± 2.7 ml O2·kg−1·min−1 and 28.4 ± 2.4 ml O2·kg−1·min−1, respectively (P = 0.02).
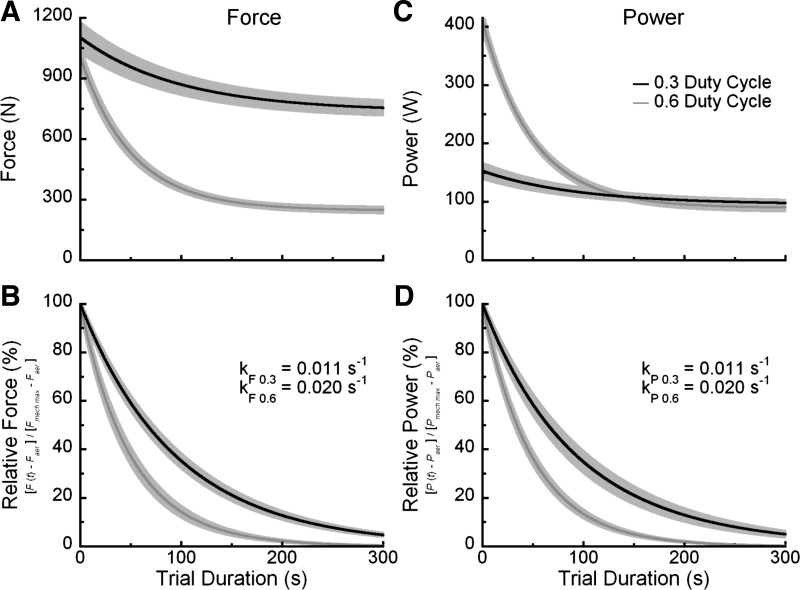
Fig. 4.
Study means for the decrements in absolute force (A) and power (C) that occur with increments in the duration of the exhaustive efforts. The time course of the relative decrements for force (B) and power (D) illustrate the fraction of the nonsustainable range supported for trial durations between 3 and 300 s; these data are equivalent to the exponential term (e−kt) in Eqs. 2 and 3. Shaded gray regions represent ± 1 SE.
Maximum mechanical power and force (Pmech max and Fmech max).
The mean values for peak power output were 410 ± 8 W and 153 ± 13 W (P < 0.001) and were achieved with similar maximum force applications of 1,007 ± 29 N and 1,102 ± 69 N (P = 0.15) for the 0.6 and 0.3 duty cycle conditions, respectively (Fig. 4). The lesser power outputs produced during the 0.3 duty cycle were primarily due to the greater durations of the inactive phase of the contraction cycle.
Exponential loss of muscular performance (k).
From each subject's duty cycle, specific measures of duration (t), power output [P(t)], and force application [F(t)] obtained during the exhaustive knee extension trials (Fig. 3) and from their respective measured mechanical (Pmech max and Fmech max) and aerobic (Paer and Faer) maximums, we determined the time course of power and force loss in accordance with Eqs. 2 and 3, by calculating the exponential time constants (kP and kF) that described these decrements (Fig. 4). For both the 0.6 and 0.3 duty cycle conditions, the measured force and power-duration relationships were well described by the calculated time constants. The mean R2 for the force exponents were 0.97 and 0.93, respectively, for the 0.6 and 0.3 conditions, whereas the power exponents described the data with a mean R2 of 0.97 and 0.86, for the 0.6 and 0.3 duty cycles, respectively.
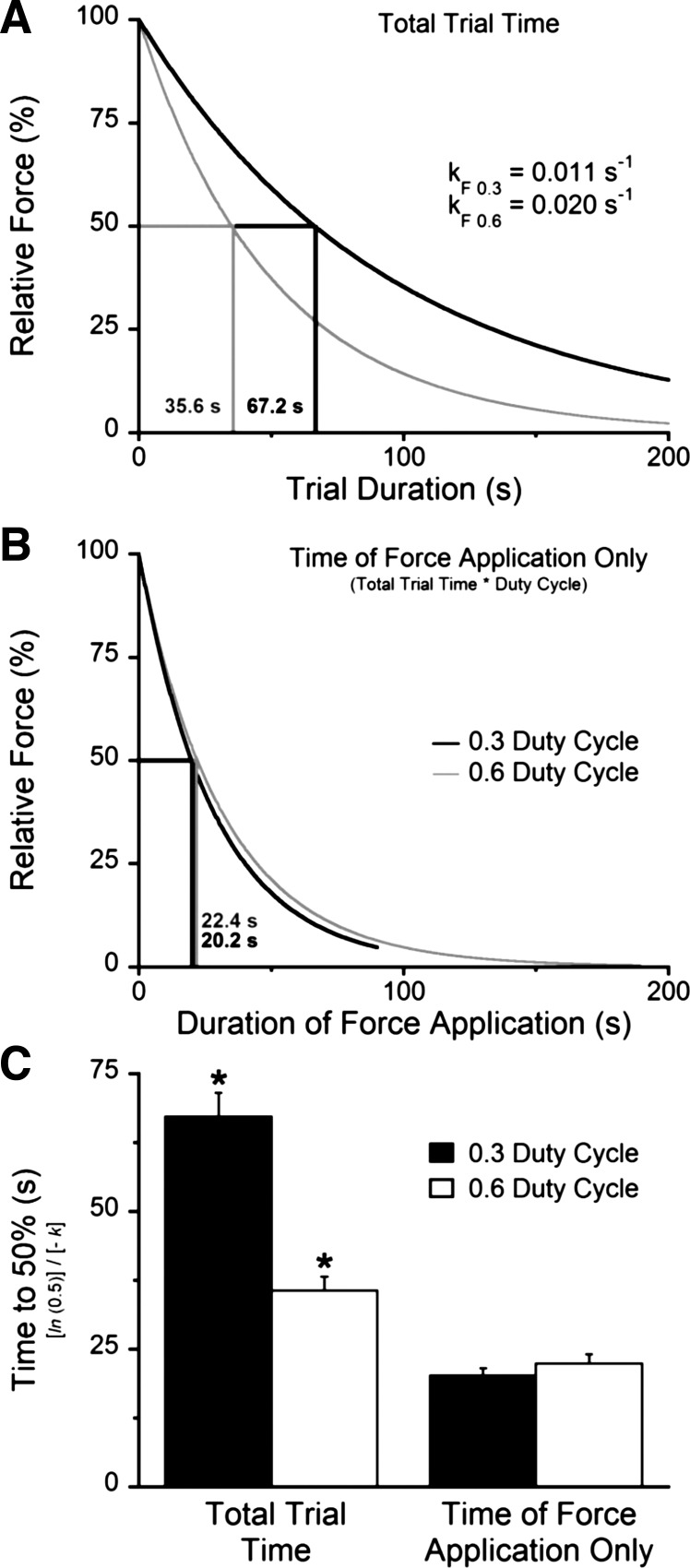
The study means for the calculated exponential constants based on the measured trial durations (Fig. 4) were greater at the 0.6 vs. 0.3 duty cycle condition for both force (kF0.6 = 0.020 ± 0.001 s−1 vs. kF0.3 = 0.011 ± 0.001 s−1; P < 0.001) and power (kP0.6 = 0.020 ± 0.001 s−1 vs. kP0.3 = 0.011 ± 0.001 s−1; P = 0.002). In contrast, the exponential time constants calculated from the cumulative duration of muscle force production (i.e., measured trial duration × duty cycle) were similar between the two duty cycle conditions (kcum0.6 = 0.032 ± 0.002 s−1 vs. kcum0.3 = 0.035 ± 0.003 s−1; P = 0.20). The exhaustive efforts representing the midpoint of the force decrements occurring in the nonsustainable performance range were 35.6 ± 2.6 s and 67.2 ± 4.3 s (P < 0.001) for the 0.6 and 0.3 duty cycle conditions, respectively (Fig. 5). When the force decrements were adjusted to the cumulative duration of force production, the midpoints were achieved in the similar time periods of 22.4 ± 1.6 s and 20.2 ± 1.3 s (P = 0.15), respectively.
Fig. 5.
The time course of force decrements (e−kt) were twice as rapid in the 0.6 vs. 0.3 duty cycle condition (A and C). In contrast, when trial durations were expressed as the cumulative duration of muscle force production only, these differences were eliminated (B). For instance, trials representing the midpoint of the nonsustainable performance range occurred for durations of 35.6 vs. 67.2 s, respectively (C; P < 0.001). However, the levels of force production eliciting this midpoint were reached with nearly the same cumulative durations of muscle activity [22.4 vs. 20.2 s, respectively (P = 0.30); C].
Electromyography.
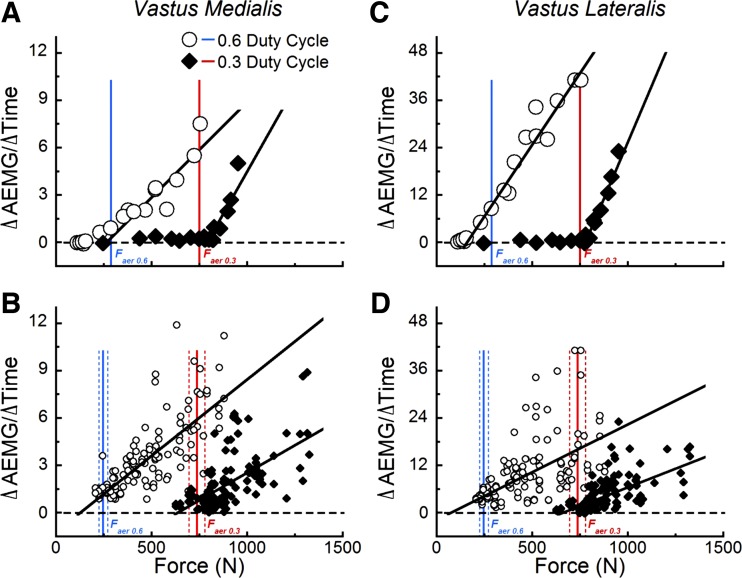
AEMG, measured on a per contraction basis, increased throughout the duration of the constant load, exhaustive knee extension trials (Fig. 6). Typically, we observed higher rates of AEMG increase (ΔAEMG/Δtime) during shorter compared with longer trials (Fig. 7). During low-force trials AEMG was essentially constant throughout the exercise (Fig. 8). We identified the minimum forces necessary to trigger a positive increase in the AEMG, occuring in at least three of the muscles evaluated, as the force threshold for the onset of neuromuscular compensation. This threshold occurred at forces in the 0.3 duty cycle condition that were 3.7 times greater than within the 0.6 condition (559 ± 37 N vs. 152 ± 17 N; P < 0.001), which corresponded to 76 ± 5% and 62 ± 4% of the force outputs that elicited the peak rates of oxygen uptake (Faer) for the 0.6 and 0.3 duty cycle conditions, respectively.
Fig. 6.
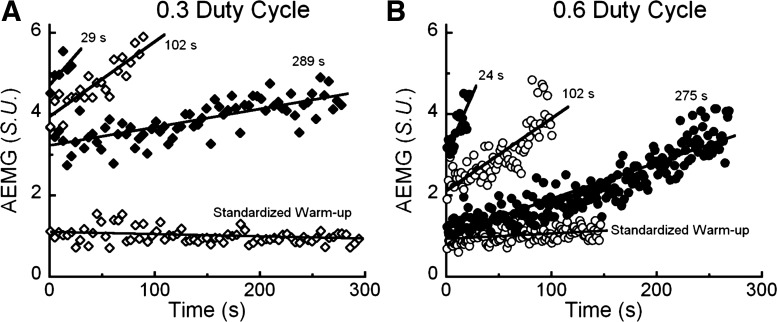
Representative, averaged contraction-by-contraction EMG (AEMG) values from the left vastus medialis during two constant-load, knee extension trials performed until failure at the 0.6 duty cycle condition. Force outputs during the 24 s and 275 s trials were nearly constant; however, EMG activity increased throughout each exhaustive trial and did so more rapidly in the shorter vs. longer trials. AEMG values are standardized (S.U.) to the first four contractions performed in the 275-s trial.
Fig. 7.
Contraction-by-contraction EMG (AEMG) values from the left vastus medialis during six exhaustive constant-load, knee extension trials with different muscle duty cycles. Here only, to compare the EMG data between laboratory visits, these data were expressed relative to the standardized warm-ups.
Fig. 8.
Levels of force application in excess of this representative subject's aerobic peak (A and C), triggered a reliance on anaerobic metabolism for force production, and as a result, rates of EMG activity increased (ΔAEMG/Δtime). At lesser levels of force application, these measures were essentially constant throughout the exercise bout. B and D: rates of AEMG increase from the 123 exhaustive bouts of knee extension administered in this study. Measured forces (A and C), and study means of the forces ± SE (B and D) eliciting the aerobic peaks are illustrated by the vertical lines in each panel (blue = 0.6 duty cycle, red = 0.3 duty cycle).
Hereafter, we consider the lower threefold difference between the forces eliciting the condition-specific areobic peaks (246 ± 18 N and 736 ± 37 N, for the 0.6 and 0.3 conditions, respectively) as the threshold for the onset of compensatory neuromuscular activity. We feel this is the most conservative treatment of the data, because trials at and above Faer elicited increases in the AEMG from all four muscles examined, and for these trials, the extent of the increase was both greater in magnitude and more consistent. Finally, the EMG technique when used on successive laboratory visits, as required here, should not be used for the identification of precise force thresholds because of inconsistencies inherent to signal normalization (22).
DISCUSSION
Our experimental manipulation targeted the inactive period of the muscle contraction cycle to achieve a twofold difference in the muscle duty cycles used during constant-load knee extension trials. This perturbation altered the relationship between the muscle's mechanical performance and the durations sustained until failure in the following ways; first, the difference in the time course of the relative performance decrements differed twofold (Figs. 3 and 4). Second, the minimum forces necessary to elicit the peak rates of oxygen uptake and initiate a net reliance on anaerobic metabolism were threefold greater in the lower vs. higher duty cycle condition (736 ± 37 N vs. 246 ± 18 N). Third, the minimum forces eliciting progressive increases in contraction-by-contraction EMG also differed by more than threefold (Fig. 8). The finding of similar forces eliciting the peak rates of oxygen uptake and the onset of the compensatory neuromuscular activity within each duty cycle condition, but with marked differences between conditions, implicates the reliance on anaerobic metabolism as the trigger that evokes the fatigue-induced compensatory response. More generally, these data indicate an important role for the mechanics of muscular contraction in setting the time course and extent of performance lost during short-duration exhaustive exercise. The data further support our hypothesis that the onset of muscle fatigue can occur at markedly different levels of force production, but at a similar metabolic transition, while completing the same task.
Contractile mechanics determine rates and extent of muscle performance loss.
Our findings extend the general quantitative description of short-duration nonsustainable muscular performance from the whole body level (14) to exercise modes activating a substantially lesser muscle volume (Table 1). In both duty cycle conditions, the performance duration relationships that we measured for knee extension exercise closely followed the general form previously established from the whole body modes of sprint running (13) and cycling (52). We found the time course of performance loss to be similar between subjects (30, 51), whereas the absolute levels of force output possible for a given subject or exhaustive duration were due to variations in either the burst maximum (Fmech max) or the sustainable performance level (Faer) present between the subjects. These results, and previous studies, suggest that the maximum burst performances are set by the contractile mechanics imposed upon the muscle by the mode of exercise (e.g., 38, 53). Here, the same mode of dynamic exercise resulted in peak forces applied for three consecutive contractions that were similar between the duty cycle conditions. In contrast, the lower limit of the nonsustainable performance range appears to be determined by the level of force production that is supported primarily by aerobic metabolism (33, 49). This metabolic transition occurs at slightly lesser performances than the peak rate of oxygen uptake (our Fig. 8; see also Refs. 33 and 42) simplification that we have used conservatively here and previously (12, 13, 51). Nonetheless, the data indicate that the level of force production responsible for the transition from primarily aerobic metabolism to a net reliance on anaerobic sources of ATP resynthesis can differ markedly while completing the same muscular task. For example, although the peak rates of oxygen uptake varied by only 10% between the duty cycle conditions (2.33 vs. 2.10 l O2/min), the forces applied to the ergometer at this common intensity differed by 300% (Fig. 4). We believe the greater levels of sustainable force production in the 0.3 duty cycle condition are attributed to the longer inactive period between contractions, which may provide a greater opportunity for blood flow (8, 16, 43), oxygen delivery for aerobic resynthesis of ATP (7, 46) and potentially the restoration of the chemical environment within the muscle cell following the contraction.
The duty cycle manipulation also resulted in a twofold difference in the relative rates of performance loss, i.e., the exponential time constants differed by a factor of 2 (Fig. 3). The magnitude of this effect is well illustrated by the 30-s greater trial durations representing the 50% midpoint of the nonsustainable performance range for the 0.3 vs. 0.6 conditions (t1/2 = 36 ± 2.6 vs. 67 ± 4.3 s; and Fig. 5A). However, when we accounted for the different periods of muscle inactivity imposed by the experimental manipulation, the exponential time constants describing the performance loss were essentially the same. For example, when the midpoint trial durations referenced above were adjusted by the measured duty cycles, these trials used 22 ± 1.6 and 20 ± 1.3 s of cumulative muscle activity in the 0.6 and 0.3 duty cycle conditions, respectively (Fig. 5). The similar time course of force loss, when expressed as if the muscles were in continuous use, implicates the muscle duty cycle and the duration of muscle activity as main factors determining the progression of performance loss and muscle fatigue in vivo. A potential explanation for this result is that the cumulative duration of muscle activity may influence the extent of metabolite concentration changes that are thought to influence the instant of task failure (28, 50). These data suggest that other mechanical parameters describing the frequency, velocity, and periods of muscle activity may also affect the time course of exhaustive performance loss in addition to the more commonly described suite of metabolically based biochemical factors (36).
Compensatory neuromuscular activity.
The EMG data provide several novel conclusions regarding the neural mechanisms used to maintain force application during dynamic, constant-load exhaustive efforts. In both duty cycle conditions, during trials with levels of force application that were essentially sustainable and supported primarily by aerobic metabolism, we found nearly no increase in EMG (Fig. 8). In contrast, in trials with levels of force production that exceeded those eliciting the aerobic peaks, we measured progressive increases in EMG activity, as others have (12, 15, 41). In these trials, which necessarily require a net reliance on anaerobic metabolism, the compensatory behavior began at the outset of each exhaustive trial and continued contraction-by-contraction until the point of failure (Figs. 6 and 7). The duty cycle manipulation used here resulted in forces at the thresholds for the onset of the fatigue-induced compensatory neuromuscular activity that were 246 ± 18 N and 736 ± 37 N for the 0.6 and 0.3 conditions, respectively, and corresponded to 25 ± 2.0 and 67 ± 2.9% of the respective burst force maximums. This is a substantial increase in the range of muscular forces that can be provided without the development of muscle force impairment or fatigue. The similar increases in both the forces, supported at the aerobic peaks, and the threshold for EMG increase (Fig. 8) support the hypothesis of a metabolic basis for the onset of the compensatory neuromuscular response to overcome impaired force production. This compensatory behavior is not likely attributed to a discrete response occurring within a single fiber type because of the marked difference in the forces necessary to elicit the fatigue response within the same musculature. This conclusion is unaffected by the small simplification that we use to quantify the force level necessary to elicit a reliance on anaerobic metabolism.
Finally, our results illustrate that to adequately compare short-duration fatiguing efforts between subjects, experimental conditions, or modes of exercise, the performances must be referenced to both the upper and lower physiologically based limits that define the short-duration nonsustainable range. This is not current practice. Although investigators with a different focus separately recognize the importance of the burst and sustainable levels of force production, studies aimed at identifying the mechanisms of fatigue or to provide physiological explanations of between-subject differences routinely reference measured data to either the upper burst maximum (e.g., top speed, peak power, maximum voluntary contraction) or lower, sustainable (e.g., V̇o2 peak, critical power) limit only. Our results illustrate the potential impact of the current approach. When the force thresholds for the onset of muscle fatigue observed in the duty cycle conditions are expressed relative to the burst maximums, which in dynamic contractions, are analogous to the maximum voluntary contraction (MVC), the onset of compensatory recruitment occurred at 25 and 67% of the maximums for the 0.3 and 0.6 conditions, respectively. Thus, the classical analysis would support a conclusion of different fatiguing mechanisms, despite the similar underlying metabolic transition occurring at very different levels of force production that we document here.
Perspectives and Significance
The experimental design used and data obtained here are focused on understanding fatigue-induced neuromuscular alterations that originate in the body's periphery and, thus, do not directly inform fatigue-based mechanisms occurring within the central nervous system. However, these results do provide a potential strategy for interventions aimed at improving muscle condition and whole body performance. First, in activities in which performance is determined primarily by force rather than power (9, 47), and duty cycle can be readily altered, considerably greater muscle forces can be sustained for longer durations at lower duty cycles. For example, in the trials illustrated in Fig. 2, the duty cycle manipulation resulted in durations to failure that were over 12 times greater and included over fourfold more contractions for the 0.3 vs. 0.6 condition despite the similar levels of force produced by the muscle. Second, greater forces that can be sustained for longer durations increase the mechanical stress experienced by the tissues, and may promote enhanced growth rates within the muscles (39) and bones (44). These are the desired effects for many interventions aimed at inducing training-related muscle hypertrophy or ameliorating age-related and spaceflight-induced atrophy of these tissues. Although resistance exercise training is contraindicated in some older adults, for others, the strategies identified by these results provide an opportunity to augment force output and repetition number in accordance with existing guidelines (2).
Concluding remarks.
Our manipulation of the muscle duty cycle, achieved by altering the rest period of the contraction cycle, resulted in a twofold difference in the rates of performance loss and a threefold difference in the force thresholds necessary to elicit the onset of fatigue and neuromuscular compensation. The consistency of the results among the different subjects, between experimental conditions, in static (15) and dynamic contractions, as well as compared with whole body sprinting (14) and cycling (12) exercise, strongly suggests there is a common physiological basis for the onset of force impairment and muscle fatigue observed during short-duration, exhaustive exercise. We conclude that a reliance on anaerobic metabolism for ATP resynthesis impairs muscle force production and triggers the nervous system to recruit additional motor units into the active volume of muscle fibers.
GRANTS
This work was supported by an award from the Institute for Translational Health Sciences (National Institutes of Health Grant UL1RR025014) to M. W. Bundle, and a graduate fellowship to C. W. Sundberg from the Montana Space Grant Consortium (NASA-NNX10AJ83H).
DISCLOSURES
No conflicts of interest, financial or otherwise, are declared by the authors.
AUTHOR CONTRIBUTIONS
Author contributions: C.W.S. and M.W.B. conception and design of research; C.W.S. and M.W.B. performed experiments; C.W.S. and M.W.B. analyzed data; C.W.S. and M.W.B. interpreted results of experiments; C.W.S. and M.W.B. prepared figures; C.W.S. and M.W.B. drafted manuscript; C.W.S. and M.W.B. edited and revised manuscript; C.W.S. and M.W.B. approved final version of manuscript.
ACKNOWLEDGMENTS
We are grateful to Peter Weyand and Steve Barrett for frequent discussions and intellectual contributions in the preliminary phase of this work. We thank Sandra Hunter for valuable comments on an earlier draft of this manuscript. Katie Burton and the Pedal House bike shop provided assistance replacing and redesigning broken ergometer parts. We also thank our subjects for their willingness to provide the rigorous efforts that have made this study possible.
REFERENCES
- 1.Adam A, De Luca CJ. Recruitment order of motor units in human vastus lateralis muscle is maintained during fatiguing contractions. J Neurophysiol 90: 2919–2927, 2003. [DOI] [PubMed] [Google Scholar]
- 2.American College of Sports Medicine, Chodzko-Zajko WJ, Proctor DN, Fiatarone Singh MA, Minson CT, Nigg CR, Salem GJ, Skinner JS. American College of Sports Medicine position stand. Exercise and physical activity for older adults. Med Sci Sports Exerc 41: 1510–1530, 2009. [DOI] [PubMed] [Google Scholar]
- 3.Andersen P, Adams RP, Sjøgaard G, Thorboe A, Saltin B. Dynamic knee extension as model for study of isolated exercising muscle in humans. J Appl Physiol 59: 1646–1653, 1985. [DOI] [PubMed] [Google Scholar]
- 4.Bangsbo J, Krustrup P, González-Alonso J, Saltin B. ATP production and efficiency of human skeletal muscle during intense exercise: effect of previous exercise. Am J Physiol Endocrinol Metab 280: E956–E964, 2001. [DOI] [PubMed] [Google Scholar]
- 5.Barker T, Poole DC, Noble ML, Barstow TJ. Human critical power-oxygen uptake relationship at different pedalling frequencies. Exp Physiol 91: 621–632, 2006. [DOI] [PubMed] [Google Scholar]
- 6.Barrett SF, Bundle MW. Instrumentation array for biomechanical reproducibility. Biomed Sci Instrum 46: 51–56, 2010. [PubMed] [Google Scholar]
- 7.Baudry S, Sarrazin S, Duchateau J. Effects of load magnitude on muscular activity and tissue oxygenation during repeated elbow flexions until failure. Eur J Appl Physiol 113: 1895–1904, 2013. [DOI] [PubMed] [Google Scholar]
- 8.Bellemare F, Wight D, Lavigne CM, Grassino A. Effect of tension and timing of contraction on the blood flow of the diaphragm. J Appl Physiol 54: 1597–1606, 1983. [DOI] [PubMed] [Google Scholar]
- 9.Bellizzi MJ, King KAD, Cushman SK, Weyand PG. Does the application of ground force set the energetic cost of cross-country skiing? J Appl Physiol 85: 1736–1743, 1998. [DOI] [PubMed] [Google Scholar]
- 10.Bigland-Ritchie B, Cafarelli E, Vøllestad NK. Fatigue of submaximal static contractions. Acta Physiol Scand (Suppl) 128: 127–148, 1986. [PubMed] [Google Scholar]
- 11.Bigland-Ritchie B, Woods JJ. Changes in muscle contractile properties and neural control during human muscular fatigue. Muscle Nerve 7: 691–699, 1984. [DOI] [PubMed] [Google Scholar]
- 12.Bundle MW, Ernst CL, Bellizzi MJ, Wright S, Weyand PG. A metabolic basis for impaired muscle force production and neuromuscular compensation during sprint cycling. Am J Physiol Regul Integr Comp Physiol 291: R1457–R1464, 2006. [DOI] [PubMed] [Google Scholar]
- 13.Bundle MW, Hoyt RW, Weyand PG. High-speed running performance: a new approach to assessment and prediction. J Appl Physiol 95: 1955–1962, 2003. [DOI] [PubMed] [Google Scholar]
- 14.Bundle MW, Weyand PG. Sprint exercise performance: does metabolic power matter? Exerc Sport Sci Rev 40: 174–182, 2012. [DOI] [PubMed] [Google Scholar]
- 15.Burnley M, Vanhatalo A, Jones AM. Distinct profiles of neuromuscular fatigue during muscle contractions below and above the critical torque in humans. J Appl Physiol 113: 215–223, 2012. [DOI] [PubMed] [Google Scholar]
- 16.Broxterman RM, Ade CJ, Wilcox SL, Schlup SJ, Craig JC, Barstow TJ. Influence of duty cycle on the power-duration relationship: Observations and potential mechanisms. Respir Physiol Neurobiol 192: 102–111, 2014. [DOI] [PubMed] [Google Scholar]
- 17.Carpentier A, Duchateau J, Hainaut K. Motor unit behavior and contractile changes during fatigue in the human first dorsal interosseus. J Physiol 534: 903–912, 2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Consolazio CF, Johnson RE, Pecora LJ. Physiological Measurements and Metabolic Functions in Man. New York: McGraw-Hill, 1963. [Google Scholar]
- 19.Dawson MJ, Wilkie DR. Theoretical and practical considerations in harnessing manpower. Proceedings of the J Roy Aeronaut Soc Symposium on Man-Powered Filght, 1977. [Google Scholar]
- 20.Edwards RG, Lippold OCJ. The relation between force and integrated electrical activity in fatigued muscle. J Physiol 132: 677–681, 1956. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Enoka RM, Stuart DG. Neurobiology of muscle fatigue. J Appl Physiol 72: 1631–1648, 1992. [DOI] [PubMed] [Google Scholar]
- 22.Farina D, Merletti R, Enoka RM. The extraction of neural strategies from the surface EMG. J Appl Physiol 96: 1486–1495, 2004. [DOI] [PubMed] [Google Scholar]
- 23.Ferguson RA, Krustrup P, Kjær M, Mohr M, Ball D, Bangsbo J. Effect of temperature on skeletal muscle energy turnover during dynamic knee-extensor exercise in humans. J Appl Physiol 101: 47–52, 2006. [DOI] [PubMed] [Google Scholar]
- 24.Fuglevand AJ, Zackowski KM, Huey KA, Enoka RM. Impairment of neuromuscular propagation during human fatiguing contractions at submaximal forces. J Physiol 460: 549–572, 1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Fulco CS, Lewis SF, Frykman PN, Boushel R, Smith S, Everett HA, Cymerman A, Pandolf KB. Quantitation of progressive muscle fatigue during dynamic leg exercise in humans. J Appl Physiol 79: 2154–2162, 1995. [DOI] [PubMed] [Google Scholar]
- 26.Garland SJ, Enoka RM, Serrano LP, Robinson GA. Behavior of motor units in human biceps brachii during a submaximal fatiguing contraction. J Appl Physiol 76: 2411–2419, 1994. [DOI] [PubMed] [Google Scholar]
- 27.Hill AV. The physiological basis of athletic records. Nature 116: 544–548, 1925. [Google Scholar]
- 28.Hogan MC, Richardson RS, Haseler LJ. Human muscle performance and PCr hydrolysis with varied inspired oxygen fractions: a 31P-MRS study. J Appl Physiol 86: 1367–1373, 1999. [DOI] [PubMed] [Google Scholar]
- 29.Hommel H. Biomechanical analyses of selected events at the 12th IAAF World Championships in Athletics, International Association of Athletics Federations, www.iaaf.org, 2009. [Google Scholar]
- 30.Hopkins WG, Edmond IM, Hamilton BH, Macfarlane DJ, Ross BH. Relation between power and endurance for treadmill running of short duration. Ergonomics 32: 1565–1571, 1989. [DOI] [PubMed] [Google Scholar]
- 31.Hunter SK, Enoka RM. Sex differences in the fatigability of arm muscles depends on absolute force during isometric contractions. J Appl Physiol 91: 2686–2694, 2001. [DOI] [PubMed] [Google Scholar]
- 32.International Association of Athletics Federations. World records, and athlete best performances. http://www.iaaf.org/ (Accessed August 20, 2014). [Google Scholar]
- 33.Jones AM, Wilkerson DP, DiMenna F, Fulford J, Poole DC. Muscle metabolic responses to exercise above and below the “critical power” assessed using 31P-MRS. Am J Physiol Regul Integr Comp Physiol 294: R585–R593, 2008. [DOI] [PubMed] [Google Scholar]
- 34.Jones AM, Vanhatalo A, Burnley M, Morton RH, Poole DC. Critical power: implications for determination of V̇o2 max and exercise tolerance. Med Sci Sports Exerc 42: 1876–1890, 2010. [DOI] [PubMed] [Google Scholar]
- 35.Josephson RK. Mechanical power output from striated muscle during cyclic contractions. J Exp Biol 114: 493–512, 1985. [Google Scholar]
- 36.Kayser B, Narici M, Binzoni T, Grassi B, Cerretelli P. Fatigue and exhaustion in chronic hypobaric hypoxia: influence of exercising muscle mass. J Appl Physiol 76: 634–640, 1994. [DOI] [PubMed] [Google Scholar]
- 37.Lind AR, Petrofsky JS. Amplitude of the surface electromyograms during fatiguing isometric contractions. Muscle Nerve 2: 257–264, 1979. [DOI] [PubMed] [Google Scholar]
- 38.Martin JC, Lamb SM, Brown NAT. Pedal trajectory alters maximal single-leg cycling power. Med Sci Sports Exerc 34: 1332–1336, 2002. [DOI] [PubMed] [Google Scholar]
- 39.Miyazaki M, Esser KA. Cellular mechanisms regulating protein synthesis and skeletal muscle hypertrophy in animals. J Appl Physiol 106: 1367–1373, 2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Moritani T, Nagata A, DeVries HA, Muro M. Critical power as a measure of physical work capacity and anaerobic threshold. Ergonomics 24: 339–350, 1981. [DOI] [PubMed] [Google Scholar]
- 41.Moritani T, Takaishi T, Matsumoto T. Determination of maximal power output at neuromuscular fatigue threshold. J Appl Physiol 74: 1729–1734, 1993. [DOI] [PubMed] [Google Scholar]
- 42.Poole DC, Ward SA, Gardner GW, Whipp BJ. Metabolic and respiratory profile of the upper limit for prolonged exercise in man. Ergonomics 31: 1265–1279, 1988. [DOI] [PubMed] [Google Scholar]
- 43.Rådegran G, Saltin B. Muscle blood flow at onset of dynamic exercise in humans. Am J Physiol Heart Circ Physiol 274: H314–H322, 1998. [DOI] [PubMed] [Google Scholar]
- 44.Raggatt LJ, Partridge NC. Cellular and molecular mechanisms of bone remodeling. J Biol Chem 285: 25,103–25,108, 2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Rohmert W. Ermittlung von erholungspausen für statische arbeit des menschen. Int Z Angew Physiol Einschl Arbeitphysiol 18: 123–164, 1960. [PubMed] [Google Scholar]
- 46.Taelman J, Vanderhaegen J, Robijns M, Naulaers G, Spaepen A, Van Huffel A. Estimation of muscle fatigue using surface electromyography and near-infrared spectroscopy. In: Oxygen Transport to Tissue XXXII, Advances in Experimental Medicine and Biology, edited by LaManna JC, Puchowicz MA, Xu K, Harrison DK, Bruley DF. New York: Springer, vol. 701, 2011, p. 353–359. [DOI] [PubMed] [Google Scholar]
- 47.Taylor CR. Relating mechanics and energetics during exercise. Adv Vet Sci Comp Med. 38A:181–215, 1994. [PubMed] [Google Scholar]
- 48.Taylor JL, Gandevia SC. A comparison of central aspects of fatigue in submaximal and maximal voluntary contractions. J Appl Physiol 104: 542–550, 2008. [DOI] [PubMed] [Google Scholar]
- 49.Vanhatalo A, Doust JH, Burnley M. Determination of critical power using a 3-min all-out cycling test. Med Sci Sports Exerc 39: 548–555, 2007. [DOI] [PubMed] [Google Scholar]
- 50.Vanhatalo A, Fulford J, DiMenna FJ, Jones AM. Influence of hyperoxia on muscle metabolic responses and the power-duration relationship during severe-intensity exercise in humans: a 31P magnetic resonance spectroscopy study. Exp Physiol 95: 528–540, 2010. [DOI] [PubMed] [Google Scholar]
- 51.Weyand PG, Bundle MW. Energetics of high-speed running: integrating classical theory and contemporary observations. Am J Physiol Regul Integr Comp Physiol 288: R956–R965, 2005. [DOI] [PubMed] [Google Scholar]
- 52.Weyand PG, Lin JE, Bundle MW. Sprint performance-duration relationships are set by the fractional duration of external force application. Am J Physiol Regul Integr Comp Physiol 290: R758–R765, 2006. [DOI] [PubMed] [Google Scholar]
- 53.Weyand PG, Sandell RF, Prime DNL, Bundle MW. The biological limits to running speed are imposed from the ground up. J Appl Physiol 108: 950–961, 2010. [DOI] [PubMed] [Google Scholar]
- 54.Zoladz JA, Rademaker ACH, Sargeant AJ. Human muscle power generating capability during cycling at different pedalling rates. Exp Physiol 85: 117–124, 2000. [PubMed] [Google Scholar]