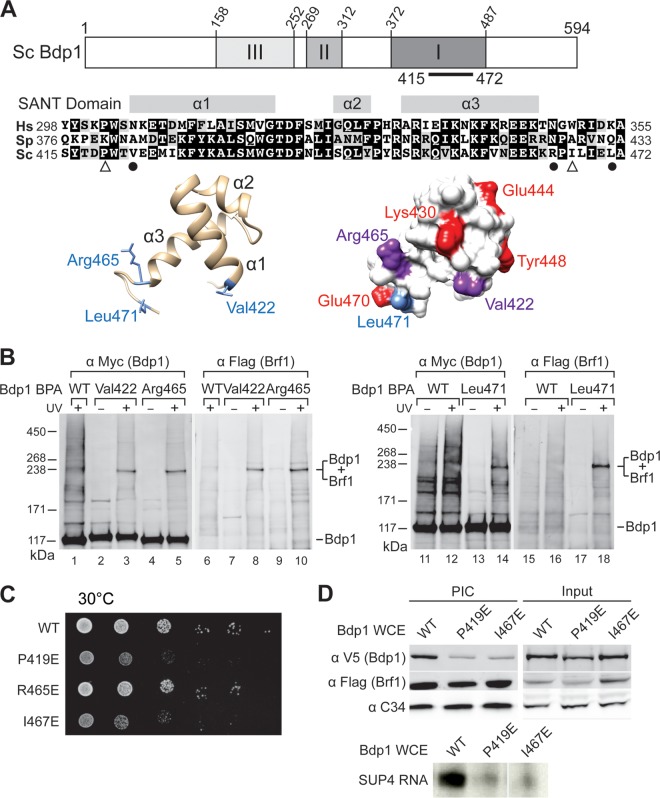
FIG 1.
The Bdp1 SANT domain cross-links to Brf1 and functions in PIC formation. (A) (Top) Essential regions I, II, and III in Saccharomyces cerevisiae (Sc) Bdp1 are shaded in the schematic. The black bar indicates the SANT domain. (Middle) Sequence alignment of the SANT domain from Bdp1 homologues. Hs, Homo sapiens (UniProt [http://www.uniprot.org/] accession number A6H8Y1; Sp, Schizosaccharomyces pombe, accession number O94481; Sc, Saccharomyces cerevisiae, UniProt accession number P46678. The three α helices in the SANT domain are indicated. Identical and similar residues are shaded in black and gray, respectively. Symbols below the sequence alignment: dots, residues with BPA cross-linking to Brf1; triangles, point mutations resulting in slow cell growth and defective transcription activity. (Bottom) Structural model of the SANT domain in a backbone trace (left) and the molecular surface (right). The model was generated by the Modeler program with the sequence with PDB accession number 2CU7 as the template. The blue side chains in the backbone trace model indicate residues involved in cross-linking with Brf1. In the molecular surface model, red residues are proposed to directly interact with Brf1, on the basis of NMR analysis. The residues interacting with Brf1, derived from both NMR analysis and BPA cross-linking, are in purple. Leu471 is proposed to interact only with Brf1 on the basis of BPA cross-linking. (B) Western analysis of photo-cross-linking. The BPA-substituted residues in the SANT domain are indicated above the lanes. Anti-Myc antibody revealed Myc epitope-tagged Bdp1 and fusion protein (lanes 1 to 5 and lanes 11 to 14). Anti-Flag antibody revealed the Flag epitope-tagged Brf1 in the fusion protein (lanes 6 to 10 and lanes 15 to 18). UV + and UV −, UV irradiation and no UV irradiation, respectively; WT, wild-type Bdp1 without BPA. (C) Cell growth analysis of Bdp1 SANT domain mutants by serial dilution spot assay. Representative slow cell growth at 30°C is shown. (D) Bdp1 SANT domain mutations affect PIC formation. (Top) Western blot analysis of proteins isolated from PIC formation. As indicated, Bdp1 mutant or WT whole-cell extracts were used in the IMT assay to isolate PICs on DNA containing the SUP4 gene. Individual proteins were probed with the antibodies indicated on the left. (Bottom) Autoradiogram of the synthesized SUP4 preliminary tRNA from PICs of the IMT assay.