FIG 5.
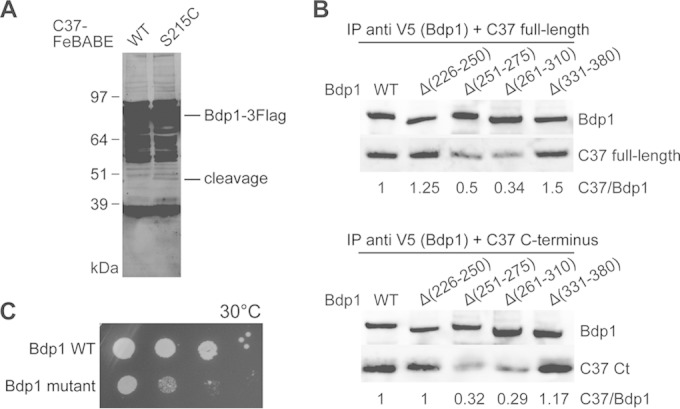
The C37 C terminus interacts with Bdp1 region II. (A) Western blot analysis of hydroxyl radical cleavage of Bdp1 by FeBABE-conjugated C37. As indicated, either the wild type or the C37 mutant with a single cysteine mutation at Ser215 was used in the FeBABE conjugation and subsequent hydroxyl radical protein cleavage assay. C-terminally Flag-tagged full-length Bdp1 and the cleaved peptide fragments were visualized by probing with an anti-Flag antibody. The corresponding cleavage site for the Bdp1 peptide fragment is aa 264. (B) Western blot analysis of coimmunoprecipitation of Bdp1 and C37. Anti-V5 antibody beads were used to precipitate the C-terminally V5 epitope-tagged Bdp1 and internal deletion mutants. Either the C37 full-length protein (top) or the C37 C-terminal (Ct) peptide fragment from aa 181 to 281 (bottom) was used in the coimmunoprecipitation assay. Bdp1 and C37 were probed with anti-V5 and anti-His antibodies, respectively. The relative intensity of C37 immunostaining is normalized to the intensity of Bdp1 immunostaining and is listed below each lane. (C) The cell growth phenotype was analyzed by a serial dilution spot assay. As indicated, Bdp1 quadruple point mutant R260E/D261K/E263K/K266E and WT strains were grown at 30°C.
