Abstract
Diffuse alveolar septal involvement is a rare form of pulmonary amyloidosis. Ante-mortem diagnosis is unusual, and most of the reported cases were diagnosed at autopsy. It has recently been reported that transbronchial lung biopsy via a flexible fiberoptic bronchoscope was a relatively safe method to confirm diffuse alveolar septal amyloidosis.
We report a case of pulmonary diffuse alveolar septal amyloidosis confirmed by transbronchial lung biopsy. The patient’s chief complaints were dyspnea on exertion and epigastric pain aggravated over a one-year period, while a chest roentgenogram showed bilateral diffuse interstitial infiltration. This case also showed nephrotic syndrome, cardiac arrhythmia, congestive heart failure, a tingling sensation in both hands and multiple nodules in the gastrointestinal tracts, suggesting involvement of the kidney, heart, peripheral nerves and gastrointestinal tracts.
We propose that when diffuse interstitial lung disease is present with systemic signs such as nephrotic syndrome or cardiac arrhythmia, amyloidosis should be considered as a possible diagnosis. Also, transbronchial lung biopsy may be a useful confirmative diagnostic tool.
Keywords: Pulmonary amyloidosis, Diffuse alveolar septal, Transbronchial biopsy
INTRODUCTION
Amyloidosis is a multisystem disease of diverse causes characterized by the presence of extracellular deposits of an insoluble fibrillar protein, amyloid1,2). Amyloid deposits are commonly found in the kidneys, nerves, vessels, liver, spleen, gastrointestinal tracts, heart and respiratory tracts3).
The pulmonary involvement of amyloidosis is usually classified as being tracheobronchial, parenchymal nodular and diffuse alveolar septal types4). Among these, the diffuse alveolar septal type is by far the rarest, and ante-mortem diagnosis is very unusual5). The diagnosis of diffuse alveolar septal amyloidosis is usually by post-mortem examination or open lung biopsy. Only a small number of cases have been confirmed by transbronchial lung biopsy using a flexible fiberoptic bronchoscope.
In Korean literature, there have been many reports of primary amyloidosis but no documented case of pulmonary amyloidosis. We describe a case of pulmonary diffuse alveolar septal amyloidosis confirmed by transbronchial lung biopsy, which also involved the kidneys, heart, nerves and gastrointestinal tracts, manifested by diffuse infiltration of the lung, nephrotic syndrome, cardiac arrhythmia and multiple nodules in the gastrointestinal tracts.
CASE REPORT
A 60-year-old woman presented with complaints of progressive dyspnea on exertion, epigastric pain and low anterior chest pain. She was well until one year ago, when epigastric pain developed. At that time, an abnormality in urinalysis was detected at a local clinic. One month later she underwent an appendectomy with an impression of acute appendicitis. At that time, a chest roentgenogram showed increased interstitial markings in both lower lung fields. Eight months ago, dyspnea on exertion and intermittent edema on her face and dorsum of the hands developed and progressed. For the last three months she was almost in a bed-ridden state due to dyspnea on exertion and weakness. She also felt postural dizziness and a tingling sensation in both hands. So, she was admitted to this hospital.
On physical examination, the blood pressure was 90/60 mmHg, the heart rate 60 beats/minute and respiration rate 24/min. Jugular venous engorgement was noted, and bilateral basal crackles were heard on the chest. The heartbeat was irregular, but a murmur was not heard. Abdominal examination revealed no organomegaly, but epigastric tenderness was noted. Pitting edema was present on both dorsum of the feet, presacral and pretibial areas.
A chest roentgenogram revealed a bilateral diffuse ground-glass density, especially in the lower lung fields, with pleural effusion (Fig. 1). Blood gas analysis showed pH 7.44, PaCO2 33 mmHg, PaO2 67 mmHg, HCO3− 22 mEq/L. The hemoglobin concentration was 11.8 g/dl, leukocyte count 6,300/mm3, platelet count 410,000/mm3 and erythrocyte sedimentation rate was 32 mm/hr. The coagulation profile was normal. The serum calcium level was 7.9 mg/dl, phosphorus 4.5 mg/dl, blood urea nitrogen 19 mg/dl and serum creatinine level 0.9 mg/dl. The serum total protein concentration was 5.0 g/dl, albumin 2.6 g/dl and cholesterol level 388 mg/dl. Urinalysis showed specific gravity 1.025, pH 6.0, albumin 4+, RBC 2–3/high power field (HPF), WBC 7–10/HPF, and 24-hour urine protein was 5,240 mg. Creatinine clearance was 66 ml/minute and the selectivity index of proteinuria was 0.12. In serum protein electrophoresis, albumin was 49.3%, alpha-1 globulin 4.0%, alpha-2 globulin 18.1%, beta globulin 17.5% and gamma globulin 11.3%. Urine protein electrophoresis showed albumin 66.2%, alpha-1 globulin 4.5%, alpha-2 globulin 5.7%, beta globulin 20.6% and gamma globulin 3.0%. In both electrophoresis, there was no M-peak, and urine Bence-Jones protein was negative.
Fig. 1.
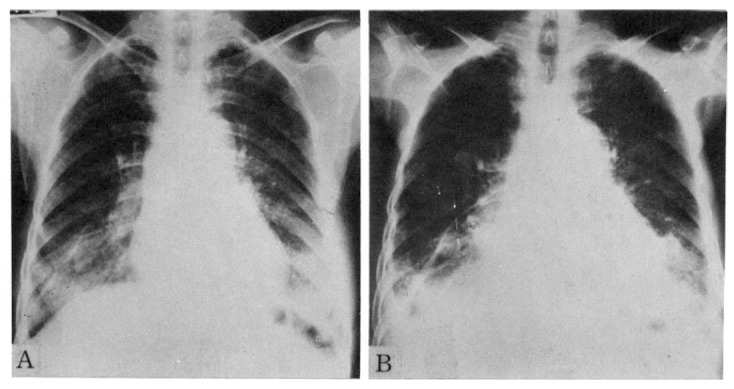
Serial chest radiographs of 60-year-old female patient.
A. The Initial radiograph shows bilateral diffuse ground glass appearing density, especially in the lower lung fields.
B. Radiograph obtained one year later A shows both aggravated lower lung field lesions. Note bilateral pleural effusion and increased heart size as compared with radiograph A.
The ECG finding was sinus bradyarrhythmia with sinus pause, junctional escape beats and low voltage QRS complex in the limb leads. A Holter ECG recording showed frequent sinus bradyarrhythmia with junctional escape rhythm, a few episodes of paroxysmal supraventricular tachycardia (sick sinus syndrome) and multiform ventricular premature beats (VPBs) (Fig. 2). In the echocardiogram, the left ventricular posterior wall, interventricular and interatrial septum were thickened, and the myocardium showed a hyperrefractile “granular sparkling” appearance (Fig. 3). A 99m Tc-pyrophosphate myocardial scan revealed no hot uptake.
Fig. 2.
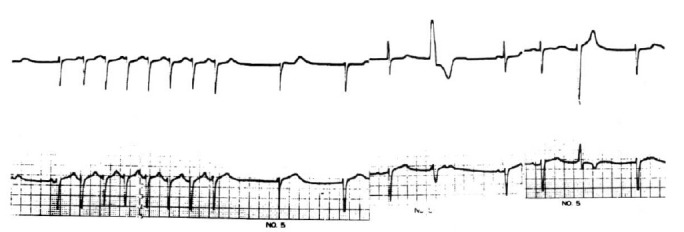
A Holter ECG recording shows sinus bradyarrhythmia with junctional escape rhythm, episode of paroxysmal supraventricular tachycardia (PSVT) and multiform ventricular premature beats (VPBs).
Fig. 3.
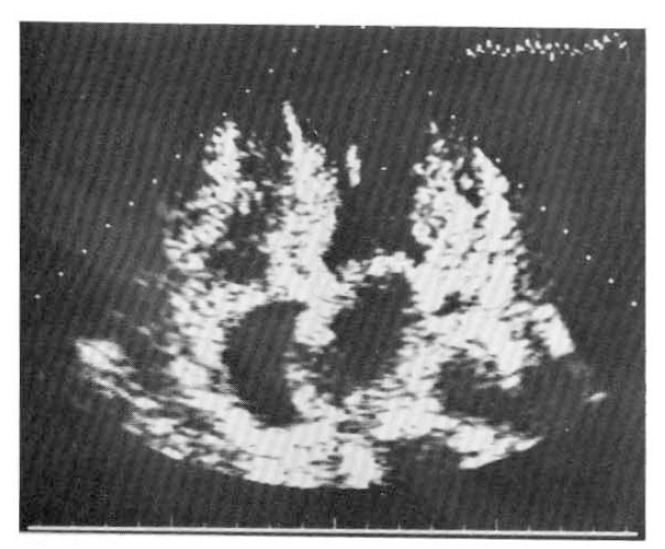
A two-dimensional echocardiogram shows a thickened left ventricular posterior wall, interatrial and interventricular septum. The myocardium shows an increased refractile pattern, a so-called “granular sparkling” appearance.
In an upper gastrointestinal series and colon study, multiple polypoid lesions were suspected in the stomach, ascending colon, splenic flexure of the transverse colon and sigmoid colon.
In a pulmonary function test, the forced vital capacity was 1.54L (68.1% of predicted value), and the forced expiratory volume in one second was 1. 48 L/sec (85.5% of predicted value), showing mainly a restrictive pattern. The carbon monoxide diffusing capacity corrected by alveolar volume (DLCO/VA) was 72.1% of the predicted value. High resolution computed tomography (HRCT) of the lung showed a diffuse thickening of the interlobular septal lines and prominence of bronchovascular core structure, suggesting a diffuse interstitial infiltrative process. Bilateral pleural effusion with mild pleural thickening was observed (Fig. 4).
Fig. 4.
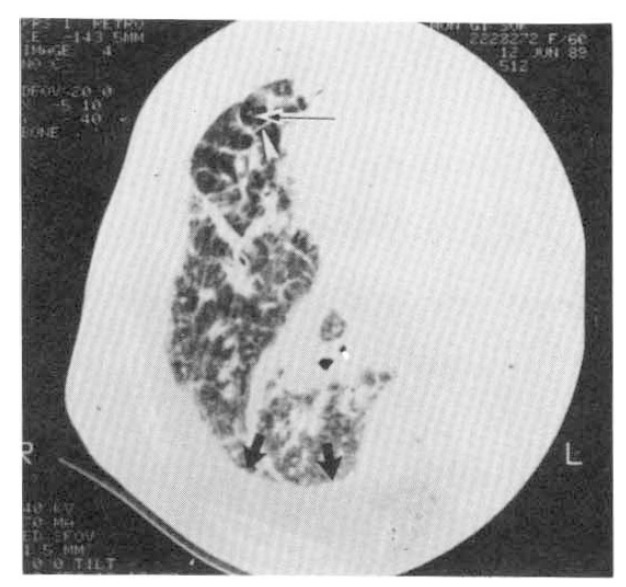
A high resolution CT obtained at right lower lung field (retrospective retargeting, FOV 20 cm, 140KVP, 510 mAs, 512×512 matrix, bone algorithm) shows prominent interlobular septa (white arrowhead) and a central core structure (thin arrow), as well as a diffuse, ill-defined, increased hazy density. Increased density between the rib and the lung (thick black arrows) is due to pleural effusion.
Because lymphangitic metastasis was suggested on chest CT and gastrointestinal tumor was also suspected, bronchoscopy with transbronchial lung biopsy was done. There was no endobronchial lesion. After the procedure, the patient complained of dyspnea, and inspiratory crackle was heard on the entire lung field. A chest radiograph revealed pulmonary edema and increased pleural effusion. The dyspnea and pulmonary edema was improved by aminophylline and diuretics. Three days later, the patient suddenly experienced dizziness after standing up and loss of consciousness, and cardiac standstill developed. Despite cardiopulmonary resuscitation, the patient expired on the 22nd hospital day.
A transbronchial lung biopsy specimen revealed diffuse hyaline thickening of the alveolar septa and blood vessel walls. Polarizing microscopy following Congo-red stain revealed apple-green birefringence at the vascular walls and along the alveolar septa (Fig. 5). Electron microscopic observation revealed a massive deposit of fibrillar material in the alveolar septum (Fig. 6).
Fig. 5.
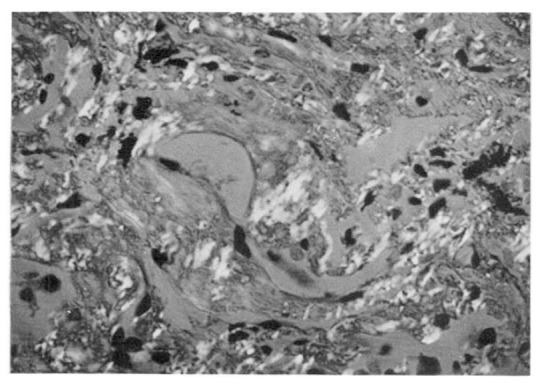
Polarizing microscopy following Congo-red stain reveals apple-green birefringence at the vascular walls and along the alveolar septa (×400).
Fig. 6.
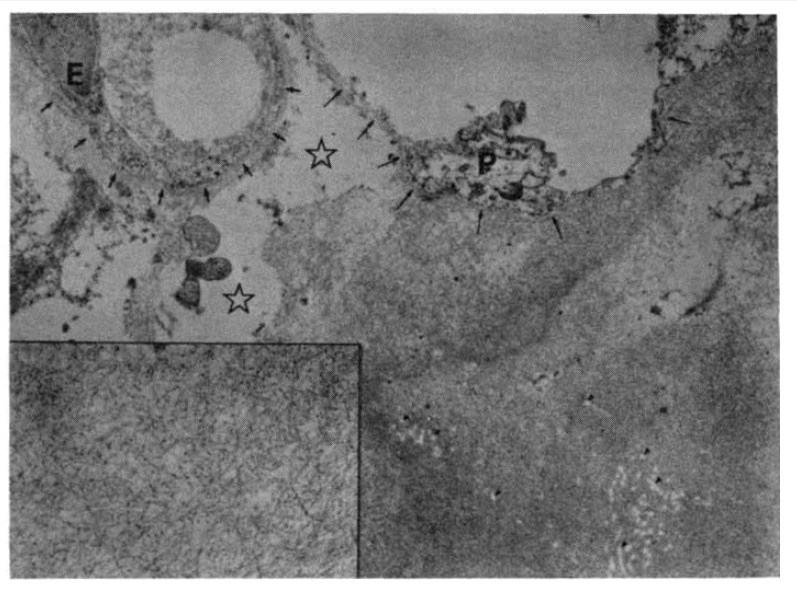
Electron microscopic observation of the lung reveals massive deposition of fibrillar material in the alveolar septum. The alveolar basement membrane (long arrows) is incorporated into the amyloid substance. Retractile white spots and fibers (tiny arrowheads) in the amyloid pool are preexisting collagen fibers. Free space (stars) in the interstitium is due to interstitial edema. Alveolar epithelial cells (P) are artifactually deformed. (E: endothelial cells; smaller arrows: capillary basement membrane). (Bar represents 1 um, ×14,000)
(Inset) Magnification of the amyloid substance reveals nonbranching fibrills of approximately 100 nm in length and 10 nm in diameter. Granular deposits, less than 10 nm in diameter, are scattered also. (Bar represents 0.1 um, ×56,000).
DISCUSSION
Pulmonary deposition of amyloid was first described by Virchow in 1857. Thereafter three major types of pulmonary amyloidosis have been described: the tracheobronchial, parenchymal nodular and diffuse alveolar septal types4). Minimal or moderate focal involvement of the lung by amyloid deposits have been reported in up to 88% of the patients with primary systemic amyloidosis6). However, diffuse alveolar septal amyloidosis is extremely rare5,7).
In our case, a chest radiograph revealed bilateral diffuse interstitial infiltration which was aggravated during a period of one year, and high resolution CT of the lung showed diffuse thickening of the septal lines, prominence of bronchovascular core structure and bilateral pleural thickening with pleural effusion. A pulmonary function test showed mainly a restrictive pattern and a mildly decreased diffusing capacity. Biopsy revealed diffuse hyaline thickening of the alveolar septum and blood vessel wall on light microscope, along with apple-green birefringence on polarizing microscope following Congo-red staining. Considering the clinical course and laboratory findings, it is certain that this case belongs to the category of diffuse alveolar septal amyloidosis.
The prognosis for diffuse alveolar septal amyloidosis is reported to be grave. The diagnosis of most patients is discovered at autopsy, and even if found while the patients is alive, most show an inexorable progression to respiratory failure within two years8).
The cause of death in pulmonary amyloidosis is also related to cardiac involvement9,10) and there have been reports that cardiac involvement tends to parallel pulmonary involvement6). Our case showed congestive heart failure, cardiac arrhythmia and a granular sparkling appearance of the myocardium on echocardiography.
Although there was no pathologic confirmation, it is evident that this case involved the heart, and sudden death was related to cardiac amyloidosis, especially with conduction system involvement that was evident by ECG and Holter monitoring.
Diagnosis of systemic amyloidosis could be done by simple and safe methods, such as rectal biopsy or subcutaneous fat aspiration, if the clinician suspects that entity of disease11). But pulmonary amyloidosis, especially the diffuse alveolar septal type, is extremely rare and very difficult to confirm in living patients12). In English literature there are six cases reported of diffuse alveolar septal amyloidosis for which ante-mortem diagnosis was made by transbronchial lung biopsy up until 198712–15).
In our case, a roentgenogram of the gastrointestinal tracts showed multiple nodules, and a high resolution CT of the lung suggested the possibility of lymphangitic metastasis. In cases of amyloidosis, transbronchial lung biopsies pose the risk of massive bleeding. But since we couldn’t completely exclude the possibility of malignancy with systemic manifestations, we decided to perform fiberoptic bronchoscopy and transbronchial lung biopsy. Fortunately, we could confirm the diagnosis of diffuse alveolar septal amyloidosis without serious complication. The dyspnea aggravated after bronchoscopy was probably due to congestive heart failure and soon improved.
Kline et al. described three cases of pulmonary parenchymal amyloidosis diagnosed by transbronchial lung biopsy via the flexible fiberoptic bronchoscope12). Although there was a report that serious complications appeared during bronchoscopy and transbronchial lung biopsy in one patient with amyloidosis15), on the whole, bronchoscopy and transbronchial lung biopsy are considered relatively safe methods if carried out with caution16). We recommend that bronchoscopy be carried out under the monitoring of ECG and oxygen saturation and that an emergency operation team for open thoracotomy stand by during the procedure to control any massive bleeding.
Amyloid infiltrates in the gastrointestinal tracts have been reported frequently. Some patients have masses that are interpreted roentgenographically as carcinoma or multiple polyps17,18). In our case, a multiple polypoid lesion shown on a gastrointestinal roentgenogram was possibly due to amyloid deposits even though pathologically not proved. Our case also showed nephrotic syndrome, a tingling sensation in both hands, gastrointestinal disturbance and orthostatic hypotension suggesting involvement of the kidneys and peripheral nerves.
In conclusion, this patient had pulmonary diffuse alveolar septal amyloidosis confirmed by transbronchial lung biopsy, which also involved the heart, kidneys, gastrointestinal tracts and peripheral nerves. We suggest that when diffuse interstitial lung disease is present with systemic signs, such as nephrotic syndrome or cardiac arrhythmia, amyloidosis should be considered as a possible diagnosis and transbronchial lung biopsy be implemented as a useful confirmative diagnostic tool.
REFERENCES
- 1.Glenner GG. Amyloid deposits and amyloidosis: The beta fibrillosis. N Engl J Med. 1980;302:1283–1292. 1333–1343. doi: 10.1056/NEJM198006053022305. [DOI] [PubMed] [Google Scholar]
- 2.Cohen AS, Shirahama T, Sipe JD, Skinner M. Amyloid proteins, precursors, mediator, and enhancer. Lab Invest. 1983;48:1–4. [PubMed] [Google Scholar]
- 3.Kyle RA, Greipp PR. Amyloidosis (AL): Clinical and laboratory features in 229 cases. Mayo Clin Proc. 1983;58:665–683. [PubMed] [Google Scholar]
- 4.Kanada DJ, Sharma OP. Long-term survival with diffuse interstitial pulmonary amyloidosis. Am J Med. 1979;67:879–882. doi: 10.1016/0002-9343(79)90748-4. [DOI] [PubMed] [Google Scholar]
- 5.Poh SC, Tjia TS, Seah HC. Primary diffuse alveolar septal amyloidosis. Thorax. 1975;30:186–191. doi: 10.1136/thx.30.2.186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Smith RS, Hutchins GM, Moore GW, Humphrey RL. Type and distribution of pulmonary parenchymal and vascular amyloid. Am J Med. 1979;66:86–104. doi: 10.1016/0002-9343(79)90488-1. [DOI] [PubMed] [Google Scholar]
- 7.Hardy TJ, Myerowitz RL, Bender BL. Diffuse parenchymal amyloidosis of lungs and breast. Arch Pathol Lab Med. 1979;103:583–585. [PubMed] [Google Scholar]
- 8.Hui AN, Koss MN, Hochholzer L, Wehunt WD. Amyloidosis presenting in the lower respiratory tract. Arch Pathol Lab Med. 1986;110:212–218. [PubMed] [Google Scholar]
- 9.Buja LM, Khoi NB, Roberts WC. Clinically significant cardiac amyloidosis. Am J Cardiol. 1970;26:394–405. doi: 10.1016/0002-9149(70)90736-8. [DOI] [PubMed] [Google Scholar]
- 10.Falk RH, Rubinow A, Cohen AS. Cardiac arrhythmias in systemic amyloidosis: Correlation with echocardiographic abnormalities. J Am Coll Cardiol. 1984;3:107–113. doi: 10.1016/s0735-1097(84)80436-2. [DOI] [PubMed] [Google Scholar]
- 11.Westermark P, Stenkvist B. A new method for the diagnosis of systemic amyloidosis. Arch Intern Med. 1983;132:522–523. [PubMed] [Google Scholar]
- 12.Kline LR, Dise CA, Ferro TJ, Hansen-Flaschen JH. Diagnosis of pulmonary amyloidosis by transbronchial biopsy. Am Rev Respir Dis. 1985;132:191–194. doi: 10.1164/arrd.1985.132.1.191. [DOI] [PubMed] [Google Scholar]
- 13.Eisenberg R, Sharma OP. Primary pulmonary amyloidosis: An unusual case with 14 years survival. Chest. 1986;89:889–891. doi: 10.1378/chest.89.6.889. [DOI] [PubMed] [Google Scholar]
- 14.Road JD, Jacques J, Sparling JR. Diffuse alveolar septal amyloidosis presenting with recurrent hemoptysis and medial dissection of pulmonary arteries. Am Rev Respir Dis. 1985;132:1368–1370. doi: 10.1164/arrd.1985.132.6.1368. [DOI] [PubMed] [Google Scholar]
- 15.Strange C, Heffner JE, Collins BS, Brown FM, Sahn SA. Pulmonary hemorrhage and air embolism complicating transbronchial biopsy in pulmonary amyloidosis. Chest. 1987;92:367–369. doi: 10.1378/chest.92.2.367. [DOI] [PubMed] [Google Scholar]
- 16.Pereira W, Kovnat DM, Snider GL. A prospective cooperative study of complications following flexible fiberoptic bronchoscopy. Chest. 1978;73:813–816. doi: 10.1378/chest.73.6.813. [DOI] [PubMed] [Google Scholar]
- 17.Gilat T, Spiro H. Amyloidosis and the gut. Am J Dig Dis. 1968;13:619–633. doi: 10.1007/BF02232969. [DOI] [PubMed] [Google Scholar]
- 18.Pandarinath GS, Levine SM, Sorokin JJ, Jacoby JH. Selective massive amyloidosis of the small intestine mimicking multiple tumors. Radiology. 1978;129:609–610. doi: 10.1148/129.3.609. [DOI] [PubMed] [Google Scholar]


