Abstract
Following a period of quiescence at the end of last century, the study of microsaccades has now regained strong impetus and broad attention within the vision research community. This new wave of interest, partly fueled by the advent of a new generation of high-resolution eyetrackers, has attracted new researchers and led to new ideas. Old hypothesis have been revisited and new ones formulated. This article is designed to serve as a practical guide for researchers in the field. Because of the history of the field and the difficulty of measuring very small eye movements, the study of microsaccades presents peculiar challenges. Here, we summarize some of the main challenges and describe methods for assessing and improving the quality of the recordings. Furthermore, we examine how these experimental challenges have influenced analysis of the visual functions of microsaccades and critically review current evidence on three long-debated proposals: the maintenance of fixation, the prevention of visual fading, and the exploration of fine spatial detail.
Keywords: Eye movements, saccade, ocular drift, fovea, visual acuity
1 Introduction
Vision is an active process. In humans, like in many other species, photoreceptors are not uniformly distributed across the retina, but their density progressively declines starting from a small region approximately at the center of the fovea. As a consequence, humans acquire visual information during brief periods of fixation separated by saccades, the rapid eye movements that enable inspection of the objects of interest with this high-acuity region.
The term “fixation”, however, is only partially accurate. Close examination of motor activity in the intervals in between saccades reveals that the eye is never stationary. During these periods, small eye movements, collectively known as fixational eye movements, incessantly perturb the gaze position. These movements occur even when attempting to maintain steady gaze on a single point. They consist of the continual alternation between miniature saccades (microsaccades or fixational saccades) and periods of erratic and relatively slow eye drifts.
Studies on fixational eye movements have primarily focused on microsaccades, which are easier to detect and measure (but see Kuang et al., 2012 for an example of recent study on drift). Microsaccades have drawn considerable interest both because they seem to occur without the observer's awareness and because of their small amplitude. Given that the primary function of eye movements is to inspect the target with the high-acuity region of the retina, what could be the function of saccades so small that maintain this region on the same object?
After being the subject of continuous investigation for large part of last century, interest in microsaccades waned in the 1990s, to then surge back into a full wave of active research this century, when a new generation of non-invasive and easy-to-operate eyetrackers with resolution sufficient to examine small saccades became available. As part of this new wave of interest, old hypothesis have been revisited and new ones proposed. This broader access to the study of small eye movements has, however, not always being accompanied by the necessary appreciation of the facts already established by the previous literature and/or full consideration of the challenges inherent in the study of very small eye movements (see Collewijn and Kowler, 2008 for an excellent discussion of these issues). These are serious concerns, as failure to pay attention to present challenges and previous accomplishments comes at the risk of adding confusion, together with new knowledge, to the field.
The purpose of this article is not to provide yet another review on microsaccades, but to serve as a practical guide to the study of small eye movements and as an introductory source for new researchers in the field. In the following pages, we analyze critical challenges presented by microsaccades and describe some possible solutions. We also examine how experimental challenges have influenced the quest for the visual functions of eye movements and summarize our viewpoint on three long-debated hypotheses. Our conclusions are simple: (a) we agree with previous observations that great caution is necessary in extrapolating laboratory results to more natural conditions; and (b) argue that when the artificial condition of sustained fixation is not enforced and observers can move their eyes normally, most microsaccades serve an exploratory function similar to that of larger saccades.
2 Challenges associated with the study of microsaccades
Studying microsaccades requires more than a high-resolution eyetracker and a pool of committed subjects. Because of the long and complex history of the field, special attention needs to be dedicated to the design of the experimental conditions, the choice of controls, and the events that are selected as microsaccades. Furthermore, because of the extremely small size of the considered movements, technical knowledge of the recording methodology is required to a level that goes beyond the standard information provided by most eyetracker manufacturers. In this section, we discuss practical challenges associated with the study of microsaccades which may not be immediately obvious to new researchers in the field.
2.1 Defining microsaccades
What is a microsaccade? Difficulties in studying these small movements emerge even from their very definition, as little agreement exists on how they should be identified. Different studies have used the term in considerably different ways, and common definitions unfortunately depend on the specific task and experimental conditions under investigation, making it difficult to generalize scientific conclusions.
Microsaccades are often introduced as the “involuntary saccades that occur spontaneously during intended fixation” (e.g., Rolfs, 2009 excellent review of the topic). This statement is typically accompanied by a subordinate clause specifying an amplitude threshold, which distinguishes microsaccades (saccades with amplitudes below threshold) from regular saccades (those with amplitudes above threshold). Although frequently used in the field, this definition implicitly comes with drawbacks: its dependence on the subject's intention (note the terms: “involuntary”, “spontaneously”, “intended”) makes it little objective and prone to different interpretations. More importantly, this definition leads to the use of unnatural viewing conditions, which, as explained below, have contributed to ongoing confusion on the visual functions of microsaccades.
Consequences of a definition that relies on volition
A first difficulty in applying the microsaccade definition above originates from what is really meant by the term “involuntary”. A common interpretation is that microsaccades are reflexive movements, and lack of awareness is often reported as evidence for such nature. Another interpretation is that microsaccades are unnecessary movements, because the projection of the stimulus is already on the preferred retinal locus of fixation. But both considerations are doubtful. Are microsaccades really “involuntary”?
As nicely pointed out by Collewijn and Kowler (2008), lack of awareness does not imply lack of voluntary control. Many movements which one would not categorize as involuntary are routinely made without explicit awareness, including saccades. Furthermore, microsaccades show clear signs of voluntary control. For example, they can be made in response to small displacements of the fixated target (Timberlake et al., 1972; Wyman and Steinman, 1973; Havermann et al., 2014) and to look in specified directions (Haddad and Steinman, 1973; Ko et al., 2010). As we will discuss later in this article, microsaccades precisely relocate gaze toward nearby regions of interest in high acuity tasks (Section 3.3) and may be voluntarily executed also during sustained fixation, perhaps to compensate for fixation errors (Cornsweet, 1956). Indeed, in agreement with this hypothesis, it has long been known that microsaccades become less frequent by simply changing the instruction from “fixate” to “hold the eyes still” (Fiorentini and Ercoles, 1966; Steinman et al., 1967), and a similar rate reduction also occurs by removing the fixation marker (Cherici et al., 2012). Thus, microsaccades may not be as involuntary as one would intuitively assume. We will focus on this point in Section 3.1.
There is another, more fundamental problem which follows from a microsaccade definition that relies on volition. Since the experimenter has normally no way to determine whether the subject intends to maintain fixation, the definition given above can in practice only be used in experiments in which fixation is explicitly enforced. This requirement is typically achieved by asking subjects to maintain fixed gaze on a small cue (commonly a single point) for prolonged periods of time, a condition we will refer to as sustained fixation. This condition is a frequent requirement in vision research experiments, but rarely, if ever, happens in natural tasks, when the periods of intersaccadic fixation only last a few hundreds of milliseconds. Sustained fixation has long been known to affect the pattern of fixational eye movements (Steinman et al., 1973) including microsaccades (Kowler and Steinman, 1980). Thus, the standard definition of microsaccades implicitly comes with specific and unnatural experimental requirements that alter oculomotor activity, the very focus of the investigation. In the following of this paper, we will often use the term “fixational saccades” to refer to the microsaccades performed under conditions in which observers are explicitly asked to keep their gaze at a fixed location.
Dealing with sustained fixation
Sustained fixation may also complicate the interpretation of experimental results in other ways. Sometimes this condition serves as a reference relative to which results obtained under other, perhaps more natural, conditions are evaluated. This use of an unnatural baseline may influence scientific conclusions. For example, a lower microsaccade rate observed in other tasks may not necessarily imply a suppression of microsaccades. Sustained fixation tends to yield frequent microsaccades in most observers (Cherici et al., 2012; Thaler et al., 2013), even if no stimulus other than the fixation marker is displayed (Fig. 4A).
Figure 4. Characteristics of fixational saccades.
(A) Fixational saccades (saccades measured during sustained fixation with amplitudes in the range 3′-30′) tend to be more frequent and smaller during strict fixation on 4′ dot than during relaxed fixation on a uniform field (N= 14; *p < 0.05, paired two-tailed t-test. Data from Cherici et al, 2012). (B) Fixational saccades and drift move gaze in opposite directions. Distribution of the fixational saccade displacements relative to the directions of the preceding drift segments. All drifts were aligned at 0° angle. (C) Effect of stabilizing the fixation cue on the retina. Mean saccade rates during fixation on 5′ dot which either moved normally (unstabilized) of remained immobiled (stabilized) on the retina. The background (a natural image) was always stabilized. Errorbars represent 95% confidence intervals (*p < 0.05, paired z-test). Modified from Poletti and Rucci (2010).
In other cases, sustained fixation only serves as an auxiliary condition combined with another perceptual task, which is the primary focus of the experiment and the task to which investigators presumably dedicate their attention. Under these circumstances, the visual and oculomotor systems are simultaneously engaged in two activities: executing the task of interest and maintaining fixation. Both activities may trigger microsaccades, yet their occurrence may be erroneously attributed to the task of interest without consideration of the underlying requirement for maintaining fixation. This type of confusion has likely contributed to the long-standing controversy over the visual functions of microsaccades.
The possible influence of the explicit requirement of maintaining accurate fixation constitutes a general problem in vision research, which goes beyond the specific area of microsaccades. Various studies have now pointed out that fixational saccades may act as confounding factors in a number of situations. For example, Yuval-Greenberg et al. (2008) showed that a certain class of gammaband EEG responses, traditionally associated with higher cognitive functions, is time-locked with the onset of microsaccades, possibly reflecting a saccadic spike potential rather than higher-order processes. Indeed, several characteristics of this EEG signal mimic the dynamics of microsaccades. Herrington et al. (2009) reported that the occurrence of microsaccades can explain part of the correlation measured between neural activity and behavior. Therefore, microsaccades should be taken into account when examining the activity of populations of neurons underlying perceptual decisions. Recently, Hafed (2013) argued that some perceptual effects normally considered the result of attentional shifts might instead be partly driven by microsaccades. These studies emphasize that the ongoing influence of oculomotor activity in perceptual tasks should never be disregarded, especially when requesting strict fixation, a condition that elicits a particularly high number of small saccades.
The importance of studying microsaccades under more natural conditions has been strongly advocated by Steinman and colleagues in a series of papers (Skavenski et al., 1979; Malinov et al., 2000; Steinman, 2003). These authors have long pointed out that the requirement for sustained fixation is not the only factor contributing to unnatural requirements. The operation of head immobilization, often necessary to enable precise measurement of very small eye movements, also affects oculomotor activity (Skavenski et al., 1979; Aytekin et al., 2014; see Section 3.2). Our point here is not that one should avoid studying sustained fixation at all costs. Much has been learned about the visual and motor systems by studying eye movements in this condition. But (a) explicit consideration should be given to its action as a confounding factor, and (b) caution is necessary in extrapolating findings obtained under sustained fixation to more natural settings.
Challenges in Defining microsaccades under more natural conditions
In more natural tasks in which fixation is not explicitly required, the distinction between voluntary and involuntary movements becomes entirely speculative: how can the experimenter determine whether fixation was intended? The previous definition can no longer be adopted. For this reason, studies on free-viewing tasks commonly define microsaccades purely based on an amplitude criterion: microsaccades are all the saccades with amplitude smaller than a pre-selected threshold. This is a more neutral approach, one that does not rely on assumptions about the subject's intentions.
A new challenge, however, emerges, since no consensus exists on how the amplitude threshold should be selected. While the specific characteristics of saccades obviously change with the stimulus and task (von Wartburg et al., 2007; Ko et al., 2010), distributions of saccadic amplitudes have generally been found to be unimodal. An example is given in Fig. 1A, which shows the average distribution of saccade amplitudes during free examination of natural scenes. No clear-cut separation exists between saccades and microsaccades in these data, raising (a) the practical question of how one can objectively select an amplitude threshold; and (b) the more fundamental question of whether any subdivision is at all warranted.
Figure 1. Saccade amplitudes.
Average probability distributions of saccadic amplitudes during free viewing of natural images (A) and during sustained fixation (B). The inset in A zooms in on the distribution of saccades smaller than 30′. In B subjects maintained fixation on a 4′ dot. Data represent averages ± SEM of the amplitude distributions of individual subjects (N = 14). Triangles indicate mean values. Eye movements were recorded by means of a Dual-Purkinje Image (DPI) eyetracker. Adapted from Cherici et al, (2012) and Kuang et al, (2012).
An empirical—but questionable—approach to this problem has traditionally been to set a threshold based on the amplitude distribution of the saccades observed during sustained fixation. The largest acceptable microsaccade is then taken to correspond to a given percentile (e.g., the 90-th percentile) of this distribution.1 But, this approach comes with its own problems, as lack of agreement seems to occur even on the range of saccades humans make under sustained fixation.
Early studies uniformly reported narrow amplitude distributions for fixational saccades, and correspondingly used the term microsaccades to indicate saccades smaller than a few minutes of arc, typically 12′ (Ditchburn and Ginsborg, 1953; Nachmias, 1959; Boyce, 1967; Ditchburn and Foley-Fisher, 1967; St.Cyr and Fender, 1969). In contrast, several recent studies have reported substantially broader distributions of fixational saccades, which has led to a considerable increment in the microsaccade amplitude threshold. It is not uncommon to see in the recent literature an extension of the range of saccades labeled as microsaccades up to 1-1.5° (e.g., Valsecchi et al., 2007; Kliegl et al., 2009; Privitera et al., 2014) and even 2° (Martinez-Conde et al., 2006), an increment of one order of magnitude with respect to the past. Obviously, this drastic change in definition has to be kept clear in mind when comparing results across studies and/or attempting to answer unsolved questions from the classical literature, as older studies were much more restrictive than the more recent ones in their definition of microsaccades (see Collewijn and Kowler, 2008 on this issue). A large amplitude threshold is particularly problematic when investigating microsaccades under natural tasks. Small saccades with amplitudes above 1° completely change the portion of the visual field covered by the foveola, the high-acuity rod-free region of the fovea. Therefore, there seems to be no reason to separate these movements from larger saccades, nor to believe that they may serve different functions.
It is unclear why some recent studies have extended the range of “microsaccades” to such large amplitudes, but several possibilities come to mind. One obvious difference is the experimental equipment. Classical studies traditionally relied on accurate but invasive methods for recording eye movements, such as the scleral coil technique (Collewijn et al., 1975; Malinov et al., 2000), whereas more recent ones have used less invasive, but also less well characterized, video-based eyetrackers (see Section 2.2). Differences in the way these tools handle post-saccadic overshoots and saccade/drift transitions may affect estimation of saccade amplitudes. It has also been speculated that a possible source for this deviation is the use of naïve and inexperienced subjects in the later studies, which contrasts with the tendency of earlier studies to rely on highly experienced observers, often the experimenters themselves (Rolfs, 2009). Less accurate fixation has indeed been measured in inexperienced observers, but the difference does not seem sufficient to explain the large change in threshold (Cherici et al., 2012). Fig. 1B, shows the average amplitude distribution of fixational saccades measured by means of a high-resolution eyetracker (a Dual Purkinje Image (DPI) tracker, Fourward Technology) over a relatively large pool of observers, almost all of them inexperienced. As shown by these data, 63% of saccades are below 20′ and 90% below 31′. Based on these data, a 30′ threshold appears to be a reasonable choice.
A definition based on anatomy and resolution
It would be important for the field to agree on a common definition of microsaccades in natural tasks. To this end, we argue that a meaningful criterion can be based on anatomy, rather than on saccade amplitude distributions, and propose a definition that consists of two, rather than one, thresholds: an upper amplitude threshold based on the size of the foveola; and a lower threshold based on the characteristics of the eyetracker used in the study. Microsaccades are then defined as all saccades with amplitudes within these two boundaries.
Because there is a clear distinction at the functional and anatomical level between the foveola and the rest of the retina (Curcio et al., 1990), a reasonable approach is to separate the small saccades that maintain the object of interest within this region—i.e., those that cause a large overlap in the stimulus present within the foveola before and after the movement—from the larger ones which do not. According to Curcio et al. (1990), the average horizontal diameter of the rod-free region is approximately 1.25°. Thus saccades up to 30′ in amplitude give more than a 50% overlap between pre- and post-saccadic images. Based on this criterion, we have routinely defined microsaccades as saccades with amplitudes up to 0.5° in our studies. Because these movements yield little change in the retinal stimulus, their functions are unclear. In Section 3.3, we argue that these small movements, even movements smaller than 20′ which yield a 66% overlap in the pre- and post-saccadic images, serve the same exploratory function as larger saccades.
Whereas studies in the field always specify an upper boundary on the amplitude that can be accepted as microsaccade, they rarely set a lower boundary. It is not rare to find studies reporting 1′ microsaccades measured with devices that clearly do not possess this level of resolution. However, providing a minimum amplitude threshold is as important as setting an upper boundary. This is because the eyetracker limits the smallest movement that can be reliably detected, causing misclassifications of the events close to this limit. These unreliable events should obviously be discarded from data analysis. In our studies, we typically use a lower threshold of 3′. Although the resolution of DPI eyetracker, as measured with an artificial eye, is approximately 1′ (see section 2.2 and Fig. 2), we conservatively work with a threshold three times larger than the measured limit to minimize the influence of other sources of errors that might be present when recording from a real eye (e.g., small head movements).
Figure 2. Evaluating the quality of recordings.
(A) An example of raw eye movement data recorded with a DPI eyetracker (black trace). Eye movements are compared to the signal recorded while tracking a stationary artificial eye (red trace). Shaded regions mark microsaccades. (B) The eyetracker's output obtained while moving the artificial eye by a square wave signal of 2′ amplitude and 1 Hz frequency.
2.2 Resolving small eye movements
Another important challenge in studying microsaccades is the determination of the degree by which experimental measurements of oculomotor activity can be trusted. Researchers have gone to great lengths for improving algorithms for detecting microsaccades. But these algorithms are little effective if the quality of the original signals is poor. For example, checks of binocularity of microsaccades—a test sometimes used to ensure that the selected events are genuine eye movements—can be easily fooled by head movement artifacts.
In general, given the small size of fixational eye movements, recordings of high quality are necessary to measure microsaccades. This implies the use of very sensitive eyetrackers capable of achieving fine spatial and temporal resolutions, requirements that are usually limited to the most sophisticated and expensive devices. Furthermore, several steps are required to ensure that the events labeled as microsaccades correspond to real eye movements and are not artifacts in the recordings. Here we describe some of these steps and review the parameters and methods commonly used to measure the quality of an eyetracker.
Sensitivity, precision and resolution
Like for any measurement instrument, multiple parameters describe the quality of an eyetracker. Terms like sensitivity, resolution, and precision are sometimes used as synonyms in the literature, but they are actually meant to indicate different characteristics. Here, as it is common in the engineering literature, we use sensitivity to indicate the ratio between changes in the input and the output signals. For example, the sensitivity of an analog eyetracker is given by the ratio between the change in the output voltage and the eye rotation that produced this change. Sensitivity is often measured to ensure linearity, a critical feature which guarantees that the system maintains similar functional characteristics throughout its operating range.
In general, the higher the sensitivity the more likely it is that a small eye movement can be detected, but this is not always the case. The actual detection limit of the system—the smallest change in input that yields a detectable change in output—goes under the name of resolution. Sensitivity and resolution are related: a system with high resolution also has high sensitivity, but the opposite is not necessarily true. For example, a system with very high gain, but some hysteresis, would possess high sensitivity and low resolution. These quantities should not be confused with the precision of a measurement, which, following the definition of the International Organization for Standardization, represents the degree to which repeated measurements yield the same results. Note that even a high-resolution system will possess low precision, if its output has high variability.
Analog and digital eyetrackers
Understandably, the quality of an eyetracker depends both on its technology and on the specific method that it implements. Like in most measurement fields, a fundamental change in eye-tracking technology came with the transition from analog to digital electronics, which enabled the development of a new generation of systems based on digital image processing algorithms. This change in technology marks the boundary between two different families of eyetrackers.
Traditional analog systems were generally developed by scientists, and their characteristics extensively examined. Many experimenters possessed deep knowledge of their apparatus and regularly checked its performance. Systems like the search coil and the DPI possess high resolution, precision, and speed, as well as excellent linearity. However, they are cumbersome, fragile, and difficult to use (some would say temperamental), requiring frequent maintenance and constant tuning of their components.
Video-based eyetrackers are primarily off-the-shelf products. They tend to be more robust and user-friendly than analog systems, but they also come with more severe limitations regarding speed and resolution and with less well characterized performances. The quality of these commercial systems critically depends on (a) their digital cameras, which intrinsically pose a trade-off between resolution and sampling frequency; and (b) the specific tracking algorithm and the way it is implemented. Unfortunately, only limited information is normally provided by the manufacturer on this latter component, making the eyetracker a sort of black box. As a consequence, the experimenter typically has little knowledge about how eye movement data are processed internally by the device and may even have no access to the raw data. He/she needs to rely on the system specifications for the quality of the recordings, but even fundamental parameters such as sensitivity/linearity and resolution may not be available or reported by the manufacturer in a non-rigorous manner.
A critical issue is the type of filtering performed internally by the eyetracker to improve the signal- to-noise ratio, which is only rarely disclosed. Filtering introduces temporal correlations and delays in the output, which the experimenter—without clear specifications from the manufacturer—may erroneously attribute to eye movements. Internal filtering also critically affects post-saccadic overshoots, possibly introducing errors in estimation of saccadic amplitudes. Given the many positive features of video eyetrackers, these considerations emphasize the need for the scientific community to invest more effort into the rigorous characterization of their performance as well as to develop open-source systems with clear specifications.
Resolution vs. internal noise
A successful marketing strategy for manufacturers has been to quantify the resolution of the eyetracker by means of its internal noise, which some report to be as low as 0.01°. The root mean square of the ouput signal measured in the presence of a static artificial eye is ∼0.005° in the DPI eyetracker (Crane and Steele, 1985) and between ∼0.001-1° in different types of video eyetrackers (Holmqvist et al., 2011). This quantity, however, is only marginally informative. It does not represent the system's resolution, but the ideal limit in resolution which could be achieved in theory. In practice, many factors contribute to worsen the resolution of the eyetracker way beyond this limit. In digital systems, which by their very nature tend to possess little noise, use of the internal noise can actually be misleading for measuring performance. For example, a system with a huge quantization step would give very little noise, but would obviously also be poorly sensitive.
Measuring resolution with an articial eye
A better approach, one that gets closer to a real estimate of resolution, consists in controlling the motion of an artificial eye by means of a high-precision manipulator in order to measure the minimum rotation that can be reliably detected (see example in Fig. 2B). It is by means of this method that Crane and Steele (1985) estimated that the DPI eyetracker can resolve changes in the signal as small as 1′. This procedure was routinely executed in the past for analog eyetrackers, but similar methods for estimating resolution are rarely applied to commercial video eyetrackers.
Measurements with artificial eyes are also important for fine-tuning data processing during analysis. For example, spectral estimation of the signal-to-noise ratio distribution has enabled us to tailor the degree of filtering necessary to separate eye movements and noise in our recordings. This has led to us to confidently conclude that ocular drift moves the image on the retina at faster speed than commonly assumed (Cherici et al., 2012). Similar types of analysis are difficult with video eyetrackers, where the internal filtering performed by the system is rarely disclosed.
While the use of a precisely controlled artificial eye is essential for a more realistic assessment of the eyetracker's performance, it should be clear that also these measurements overestimate the system's capabilities. The human eye differs from an artificial eye in many ways, and various factors contribute to lower the quality of eye-tracking in real experiments. In video systems, changes in pupil size (Wyatt, 2010; Drewes et al., 2012; Wildenmann and Schaeffel, 2013; Choe et al., 2014), the color of the iris (Nyström et al., 2013), lack of proper head immobilization and/or slippage of head-mounted camera (Kolakowski and Pelz, 2006; Houben et al., 2006) have already been shown to have a negative influence on the precision of measurements. Yet experiments not always control for these factors. For example, often the head of the observer is free or loosely restrained, a condition which should be expected to drastically lower resolution and precision. Head movements are problematic also for the DPI and coil eyetrackers (Crane and Steele, 1985; Robinson, 1963)—but not for the Revolving Field Monitor (Epelboim et al., 1995)—even though the DPI eyetracker is by design less sensitive to head movements than current video systems. For this reason, in our experiments, we routinely immobilize the head of the observers by means of a head-rest and a custom dental-imprint bite-bar. This approach is necessary to ensure accurate detection of very small saccades.
Measuring microsaccades
The specific algorithm one needs to use for detecting and measuring microsaccades unavoidably depends on the eyetracker and the quality of the data. With high-quality recordings, such as those provided by the DPI eyetracker, a simple criterion based on ocular speed and displacement amplitude is very effective. But attention needs to be paid to the method followed for estimating velocity, as the operation of differentiation enhances the influence of noise (prior low-pass filtering is required). Various speed-estimation algorithms exist in the literature, including some suited for real-time operation (Janabi-Sharifi et al., 2000).
In our studies, we use the eye speed to segment oculomotor traces into separate periods of ocular drifts and saccades. Periods with peak velocity higher than 3°/s are labeled as possible saccadic events, and their amplitude defined as the modulus of the vector connecting the two locations at which eye speed becomes greater (saccade onset) and lower (saccade offset) than 2°/s. Consecutive events closer than 15 ms are then merged together into a single saccade, a method which automatically excludes post-saccadic overshoots resulting from the damping of the eyetracker and possible movement of the lens (Deubel and Bridgeman, 1995; Stevenson and Roorda, 2005). Saccades with amplitude in the range 3′-30′ are then classified as microsaccades.
Traces recorded with more noisy eyetrackers may require more sophisticated algorithms for reliably extracting microsaccades. For example, blink-related artifacts in the recorded traces may easily be confused for eye movements if not handled correctly. Users of binocular video-based systems may also want to impose a check for binocularity. Although the presence of disconjugate microsaccades has been documented in monkeys when stimuli are viewed in depth (Horn and Cullen, 2012), monocular events measured when subjects view stimuli that lay on the same depth plane, as common in psychophysics experiments, are likely to be recording artifacts (Collewijn and Kowler, 2008).
2.3 Localizing gaze
Determination of where exactly the observer looks is another important element in the study of microsaccades. For example, much discussion over the functions of these movements has focused on whether they compensate for, or contribute to, fixation errors. Answer to this question clearly requires accurate localization of the line of sight—i.e., determination of which point in the scene projects to the center of the preferred retinal locus of fixation. Accurate gaze localization is also critical in many experiments involving gaze-contingent manipulations of the input to the retina.
As explained in this section, localization of the line of sight represents an insidious challenge. Its error does not just depend on the eyetracker, but on both the instrument and the transformation that maps its native output into degrees of visual angle. This transformation is typically obtained with a calibration procedure, which by itself may yield a region of uncertainty as large as the foveola. We describe below a gaze-contingent calibration procedure which helps narrowing this region of uncertainty.
Trueness
Following the terminology suggested by the International Organization for Standardization, the quality of gaze localization can be quantified by means of the trueness of the output angular signal, defined as the inverse of the deviation between the real orientation of the line of sight and the estimated one. This quantity depends on several characteristics of the eyetracker, and is influenced by a variety of factors, such as long-term drifts in the device, the presence of head movements, and, in video eyetrackers, changes in pupil size (Wyatt, 2010; Drewes et al., 2012; Wildenmann and Schaeffel, 2013).
Trueness should not be confused with the resolution of the machine: gaze localization is often only approximate even in systems capable of resolving very small eye movements. In the literature, gaze localization errors have been reported to be less than 0.1° for the DPI eyetracker (Deubel and Schneider, 1996) and in the range 0.3-1° for the most common video eyetrackers (Holmqvist et al., 2011; Blignaut and Wium, 2014). But again, these numbers are likely to seriously underestimate the real errors: trueness is particularly difficult to measure, because it requires the independent determination of a reference value for gaze position. Thus, great caution is necessary in making claims about where exactly the observer looks at the scale relevant for the study of microsaccades.
Errors resulting from calibration
Most eyetrackers do not directly measure gaze position in space. Rather, their raw signals need to be converted into angular coordinates in visual space by means of transformation determined during a preliminary calibration procedure. There are two types of errors that result from this transformation. First, the quality of the conversion obviously depends on how the mapping function relates to the (possibly nonlinear) eyetracker's behavior across its working area. Second, errors result from fixational eye movements during the procedure, when observers are typically required to fixate on various points at known positions. In inexperienced observers the physiological instability of fixation can be considerable, covering an area as large as 0.5 deg2 (Cherici et al., 2012).
An example of the uncertainty caused by eye movements is given in Fig. 3. A standard calibration was repeated multiple times. At every repetition, the observer pressed a button when accurately fixating on each of the dots of a 3×3 grid. However, because of fixational eye movements, at any given point of the calibration grid, a different value was recorded for every repetition of the procedure. In the example in Fig. 3B, even though the subject was highly experienced, the resulting variability covered an area of 451 arcmin2. Much larger areas can be expected with inexperienced or less careful observers. Thus, even with a highly linear and high-resolution eyetracker as the DPI, significant errors in gaze localization may occur. To further complicate things, these errors are not systematic, but vary across experimental sessions every time that the calibration procedure is repeated.
Figure 3. Uncertainty in gaze localization.
The line of sight is usually determined by means of a preliminary calibration routine. (A) A standard calibration. The subject sequentially looks at a series of points at known positions and presses a button when accurately fixating on each point. Note that eye movements are always present during each fixation, preventing exact localization of the line of sight. (B) An example of the calibration errors resulting from eye movements. Each dot represents the offset between the gaze position at the time of button press and the fixated marker in an individual repetition of the calibration procedure. The 95% confidence ellipse with its area (arcmin2) and Gaussian fits of the marginal distributions with their standard deviations are also shown. (C) A gaze-contingent refinement of the calibration. The subject fixates again on each point of the grid and corrects for possible offsets (blue arrow), while the estimated center of gaze is displayed in real time on the monitor (red cross). Once the error has been corrected, the subject presses a button, and the offset is recorded. (D) These corrections are incorporated into the calibration function, significantly improving gaze localization. Modified from Poletti et al, (2013).
A gaze-contingent calibration procedure
To circumvent this problem, we have recently developed a calibration procedure that effectively improves gaze localization by almost one order of magnitude compared to standard methods (Ko et al., 2010; Poletti et al., 2013). This procedure relies on a technique of gaze-contingent display, in which the stimulus on the display is updated according to the observer's eye movements. To this end, we use EyeRIS, a custom-developed hardware/software system which acquires eye movement signals from the eyetracker, processes them in real time, and updates the display according to the desired combination of estimated oculomotor variables (Santini et al., 2007).
Our calibration procedure consists of two steps. In the first phase, subjects sequentially fixate on a grid of nine points, equispaced within the working area of the display, as it is customary in eye-tracking experiments (Fig. 3A). This gives us a first approximation of the gaze-to-angle mapping, which is then refined in a gaze-contingent manner. In the second phase of the calibration procedure, subjects fixate again on each of the nine points of the grid, but this time they also see our current estimate of the line of sight based on the previous phase of the calibration. This estimated location of the center of gaze is displayed in real time on the monitor as a retinally-stabilized red cross (Fig. 3C). While maintaining fixation, subjects use dedicated controls on the EyeRIS joypad to adjust the position of this red cross until it matches their perceived center of gaze. These adjustments are then incorporated into the offsets and gains of a bilinear interpolation.
As shown in Fig. 3D, this procedure considerably reduces the dispersion of gaze localization compared to the standard procedure. In our experiments, we routinely repeat this procedure several times during each session to correct for possible drifts resulting from the apparatus and/or microscopic head adjustments which may occur even with a bite-bar. This approach has enabled us to examine in greater detail how microsaccades move the line of sight (Ko et al., 2010) and how they affect foveal vision (Poletti et al., 2013).
3 Visual functions of microsaccades
Over the course of more than half a century of research, many ideas have been put forward to explain the existence of microsaccades. In the second part of this article, we build upon the technical observation of the previous section to critically analyze the status of three well-known hypotheses. These proposals have argued for critical roles of microsaccades in: (1) the maintenance of accurate fixation; (2) the prevention of image fading—the gradual disappearance of the visual percept experienced in the absence of retinal image motion; and (3) vision of fine spatial detail. To provide tangible demonstrations of concepts and ideas, we will draw examples from data collected in our own experiments whenever possible.
3.1 Fixation maintenance
One of the oldest hypotheses on microsaccades is that these movements play a role in the maintenance of accurate fixation. This proposal has received considerable experimental support, some of which is reviewed here.
Historically, it was first proposed by Cornsweet (1956) that fixational saccades may correct for displacements caused by ocular drifts which move the point of fixation away from the preferred locus on the retina. Subsequent findings, however, argued against this proposal, suggesting that both drift and microsaccades create, as well as correct for, fixation errors (Nachmias, 1959; St.Cyr and Fender, 1969). It was also shown that microsaccades are not really necessary for maintaining fixation: one can fixate accurately even in the absence of saccades by means of ocular drift alone— thus a form of slow control (Steinman et al., 1967, 1973). Many recent observations support the notion that ocular drift is under some degree of motor control (e.g., Møller et al., 2006; Cherici et al., 2012). Thus, if saccades are indeed part of the strategy by which humans maintain fixation, they are probably not the only component of this process, but act together with ocular drift.
Determination of whether microsaccades correct for retinal errors requires accurate localization of the line of sight. As pointed out in Section 2.3, this is a tricky operation, which may yield a region of uncertainty as large as the foveola. This uncertainty should be kept in mind when analyzing reports of absolute displacement of the stimulus on the retina (e.g., Beeler, 1967; St.Cyr and Fender, 1969). Because of this difficulty, many studies have investigated the function of fixational saccades using relative, rather than absolute, coordinates. Examples include analysis of: (a) the start and landing positions of microsaccades relative to the mean of the spatial distribution of gaze (Cornsweet, 1956); (b) the direction of each microsaccade relative to the direction of the preceding drift (Cherici et al., 2012); and (c) the statistics of relative displacements (Engbert and Kliegl, 2004). These approaches provide very useful indications, but do not directly estimate fixation errors. To reach a more conclusive answer, accurate knowledge of how stimuli move on the retina with respect to the preferred locus of fixation will be necessary (Putnam et al., 2005).
Despite the difficulty in localizing the line of sight, a considerable body of evidence supports the idea that microsaccades are generally used to maintain accurate fixation on a marker when observers are instructed to do so. Analyses of the time series of displacements indicate that fixational saccades tend to bring the line of sight away from its current position in a way that is faster than Brownian motion at short time-scales—as one may expect, given the characteristics of saccades—but slower than Brownian motion at longer time delays (Engbert and Kliegl, 2004). Thus, microsaccades appear to help constraining fixation in the long term. Furthermore, microsaccades are highly influenced by the presence/absence of a clear fixation target. The data in Fig. 4A show that microsaccades are significantly more frequent and smaller when subjects attempt to maintain accurate fixation on a fixation marker (a small dot; strict fixation) rather than at the center of a uniform field without the marker (relaxed fixation). The directions of drift and subsequent microsaccades are also anti-correlated during strict fixation, but not during relaxed fixation (see Fig. 4B; Poletti and Rucci, 2010; Cherici et al., 2012). In addition, fixational saccade rate tends to be positively correlated with ocular drift speed, and their amplitudes negatively correlated with drift curvature (Cherici et al., 2012). All these characteristics are exactly those that one would expect if fixational saccades are part of the oculomotor fixation strategy.
The idea that microsaccades and drift work together to maintain fixation also explains results obtained in retinal stabilization experiments. Microsaccades are less frequent and ocular drifts larger during sustained fixation on an unstabilized dot when the background is stabilized (i.e., maintained immobile on the retina; Poletti and Rucci, 2010). This result makes sense if microsaccades compensate for retinal displacements, because estimation of fixation errors is more difficult in this condition. The visual system can no longer rely on the normal motion of the entire image on the retina, but only on the displacement of the fixation marker from the preferred retinal locus. Furthermore, the microsaccade rate drops even further if the fixation marker is also stabilized on the retina together with the background (Fig. 4C). Again, this decrement is to be expected under the hypothesis that fixational saccades tend to be corrective, since the projection of the target always remains close to the preferred retinal locus in this condition, and saccades are no longer needed to correct for retinal displacements.
Support for the idea that microsaccades contribute to maintaining fixation also comes from neurophysiology. Until recently, it was commonly assumed that fixation was an inhibitory process preventing the occurrence of saccades toward possible peripheral targets (Dorris and Munoz, 1995). In this view, the rostral deep layers of the superior colliculus maintain sustained activity during fixation, which then declines after the offset of the fixation cue, disinhibiting saccades toward other locations in the visual field. An alternative model has been recently proposed, according to which the rostral superior colliculus controls the maintenance of fixation using a population code similar to that employed for the generation of saccades (Hafed et al., 2009; Hafed and Krauzlis, 2012; Goffart et al., 2012). According to this proposal, fixational saccades are the results of transient imbalances of activity in the colliculi. A fixation error would lead to such imbalance and to the generation of a microsaccade to restore equilibrium.
All these results support the notion that—although not strictly necessary (Steinman et al., 1967, 1973),—fixational saccades normally help maintaining accurate fixation on point. While these studies reveal fundamental principles on the mechanisms underlying the production of microsaccades, we remind the reader that strict, prolonged fixation on a single point does not happen during normal viewing. Therefore, it is critical to interpret these findings in terms of their implications for natural viewing conditions. A possible implication of these results is the existence of a very narrow preferred retinal locus of fixation that the visual system normally attempts to relocate on the object of interest. We will discuss this hypothesis in detail in Section 3.3.
3.2 Prevention of image fading
In the middle of last century, it became possible for the first time to immobilize stimuli on the retina, a laboratory condition known as retinal stabilization (Ditchburn and Ginsborg, 1952; Riggs and Ratliff, 1952; Yarbus, 1967). These experiments revealed that visual percepts tend to progressively lose contrast and may eventually disappear completely in the absence of retinal image motion (Koenderink, 1972; Kelly, 1979; Tulunay-Keesey, 1982). Fading of dim peripheral stimuli can also be experienced during steady and prolonged fixation without retinal stabilization, an effect first described by Troxler in 1804.
Following these findings, it was suggested by Ditchburn et al. (1959) that microsaccades serve the function of preventing visual fading during fixation. These researchers conducted a clever experiment: they (a) used retinal stabilization to immobilize an image on the retina so that it would fade, and (b) moved the stabilized image by means of different trajectories designed to replicate the characteristics of microsaccades and ocular drift. Their goal was to determine which of these two types of motion would be the most efficient in preserving visibility of the stimulus. As shown in Fig. 5A, Ditchburn et al. (1959) noticed that fading was more frequent when the stimulus moved sinusoidally at very low speeds and that relatively fast displacements made the stimulus visible for a considerable fraction of the time. Based on these results, they concluded that only microsaccades were effective in counteracting visual fading.
Figure 5. Microsaccades and image fading.
(A) Ditchburn et al., (1959) visibility data replotted as a function of the speed of retinal image motion rather than displacement amplitude. In this experiment, an otherwise stabilized stimulus was moved to yield on the retina a sinusoidal trajectory with fixed frequency (0.55 Hz) and variable amplitude. The red line represents the level of visibility, an index of the fraction of time that the stimulus remained visible, as a function of the mean (top) and peak speed (bottom) of the imposed motion. The visibility levels measured by Ditchburn under complete retinal stabilization and when the stimulus was moved to replicate a microsaccade are also shown. Recent analyses (Cherici et al, 2010) have shown that the eye drifts faster than assumed by Ditchburn, yielding velocities which account for better visibility than microsaccades. (B) Microsaccades rates under three different fading conditions. Fading was simulated by progressively manipulating the image in one of two ways: by lowering its contrast (Contrast) or low-pass filtering its content (Frequency). In both conditions, microsaccades rates are compared to those measured when movies were played backwards, so that the contrast or the frequency band of the image progressively increased (No Fading conditions). Mean microsaccade rates measured under retinal stabilization (Stabilized) and during normal unstabilized presentations of the same images (Normal) are also shown. Rates are not affected by a fading stimulus and are lower under retinal stabilization (⋆ p < 0.05 paired t-test; adapted from Poletti and Rucci, 2010).
The idea that microsaccades prevent the fading experienced under retinal stabilization quickly gained widespread notoriety. It continues to remain popular nowadays, and has received new interest with the recent claims that microsaccades are critical for maintaining visibility both in the periphery and at center of the visual field (Martinez-Conde et al., 2006; McCamy et al., 2012). However, the plausibility of this theory has been debated among oculomotor researchers for a long time (Kowler and Steinman, 1980; Steinman, 2003; Collewijn and Kowler, 2008; Poletti and Rucci, 2010; Kagan, 2012). At the heart of the debate is the question of how to extrapolate results obtained under artificial laboratory settings to natural viewing conditions, when fixations are brief, stimuli normally possess higher contrasts, and many other motor activities besides microsaccades contribute to retinal image motion.
Multiple considerations seem to speak against this hypothesis. We summarize ten of them below:
Careful examination of the data reported by Ditchburn et al. (1959)—the original study that led to this hypothesis—reveals that even these data do not argue for a privileged function of microsaccades in preventing image fading. Ditchburn and colleagues modeled drift as a low-frequency oscillation and measured the percentage of time that a stimulus was visible as a function of the amplitude of displacement. Fig. 5 replots Ditchburn's original data as a function of speed rather than amplitude. This graph clarifies that full visibility was already achieved with a speed of ∼40′/s. At the time of Ditchburn's study it was commonly assumed that ocular drift would provide retinal speeds substantially lower than this value. This assumption was based on manual measurements of displacements over relatively long intervals, an operation which, because of the curvature of ocular drift, significantly underestimates instantaneous speed. It is now known that 40′/s is well within the normal range of mean drift speed (Cherici et al., 2012; Kuang et al., 2012). Therefore, when re-examined with current knowledge, these classical data do not support a special role for microsaccades. They suggest, in fact, that ocular drift is very effective in maintaining visibility.
In line with the previous observation, it should be pointed out that other studies with experiments similar to those of Ditchburn et al. (1959) did not reach the same conclusions. For example Gerrits and Vendrik (1970) precisely controlled the retinal motion of an otherwise stabilized stimulus by using a small electric motor mounted on a contact lens. In contrast with the previous report, these authors found that continuous slow motion was more effective than abrupt shifts in preserving visual perception. They concluded that drifts are more effective than microsaccades in preventing visual fading.
It has long been observed that outside of the laboratory, when the head is not restrained as it is often necessary in studies of small eye movements, uncompensated or partially-compensated head and body movements also contribute to the motion of the retinal image providing a range of retinal speeds far larger than those necessary for preserving visibility (Steinman, 2003). We recently reconstructed the motion of the retinal image during natural head-free fixation by using data collected by Steinman and colleagues with the revolving field monitor (Epelboim et al., 1995), to our knowledge the only eyetracker with demonstrated capability of resolving 1′ movements without requiring immobilization of the head. The results of this analysis confirm that the image on the retina normally moves much faster than Ditchburn and colleagues assumed, reaching speeds of ∼2°/s for a fixated nearby object (Aytekin et al., 2014). These results further suggest that perceptual fading is not a concern under natural viewing conditions.
Qualitative reports during different types of paralysis are in agreement with the observation at point 3. Fading is not experienced when eye movements are suppressed by means of retro-bulbar block, a procedure that leaves head movements unaltered (Stevens et al., 1976). In contrast, fading does occur under complete body paralysis (Stevens et al., 1976) (but see Whitham et al. (2011)).
Unlike entoptic images (Coppola and Purves, 1996), images of external stimuli seem to fade slowly over a period at least one order of magnitude longer than the duration of natural fixation. Indeed, controversies even exist on whether or not perfectly stabilized images disappear completely in the long term (Arend and Timberlake, 1986; Ditchburn, 1987). The attenuation in contrast sensitivity measured during prolonged viewing of stabilized stimuli (many seconds or even minutes; Koenderink, 1972; Kelly, 1979; Tulunay-Keesey, 1982) is not observed with brief stimulus exposures comparable to the duration of natural fixation (Tulunay-Keesey, 1982; Rucci et al., 2007). These findings suggest that image fading is not a concern during the brief periods of natural fixation.
In a seminal study, Cornsweet (1956) found no correlation between the frequency of microsaccades and the disappearance of a retinally-stabilized stimulus. This classical finding may appear to conflict with those of more a recent report with sustained fixation of unstabilized stimuli (McCamy et al., 2012). But also this later study actually found little effect on visibility for saccades smaller than 30′, a result compatible with Cornsweet's classical data.
Fading of the stimulus does not lead to an increment in the number of microsaccades. Fig. 5B shows the mean rates of microsaccades collected in three separate experiments which examined the effects of: (1) changing the contrast of the stimulus, (2) varying its frequency content, and (3) eliminating retinal image motion. All three conditions resulted in fading stimuli: the first two simulated perceptual fading by manipulating the image; the third condition focused on the endogenous fading resulting from retinal stabilization. Microsaccade rates did not increase in any of these conditions, not even when close attention to the vanishing stimulus was explicitly enforced (Poletti and Rucci, 2010).
The data in Fig. 5B indicate that microsaccades are not elicited by decrements in visibility. Perhaps, a closed feedback loop is unnecessary, and the baseline microsaccade rate is already set to a level sufficient to prevent fading. But this seems unlikely. The speed of fading depends on stimulus contrast, so that no single optimal baseline rate would exist. Furthermore, the data in Fig. 5B show that elimination of retinal image motion does affect microsaccade rates: microsaccades are significantly less (rather than more) frequent under retinal stabilization, a condition which impairs visibility. Similar results were previously reported by Cornsweet (1956). While these findings are difficult to justify in terms of the fading hypothesis, they find an immediate explanation with the proposal that microsaccades normally contribute to the maintenance of fixation, as discussed in Section 3.1.
As summarized later in Section 3.3, microsaccades move the line of sight toward nearby regions of interest during execution of high-acuity tasks. In these circumstances, the precise landing location of a microsaccade matters: discrimination of fine spatial patterns is equally impaired in the absence of microsaccades and when microsaccades occur but are not effective in shifting the preferred retinal locus of fixation on the stimulus (see Fig. 7D). Thus, in these tasks, microsaccades help by positioning the projection of the object of interest on the retina, not by enhancing visibility with their transients.
In agreement with the previous points, saccades smaller than 30′ appear to have no effect on contrast sensitivity. A recent study that specifically measured contrast thresholds in the presence and absence of microsaccades found no differences between these two conditions neither at low nor high spatial frequencies (Mostofi et al., 2014).
Figure 7. Microsaccades and foveal vision.
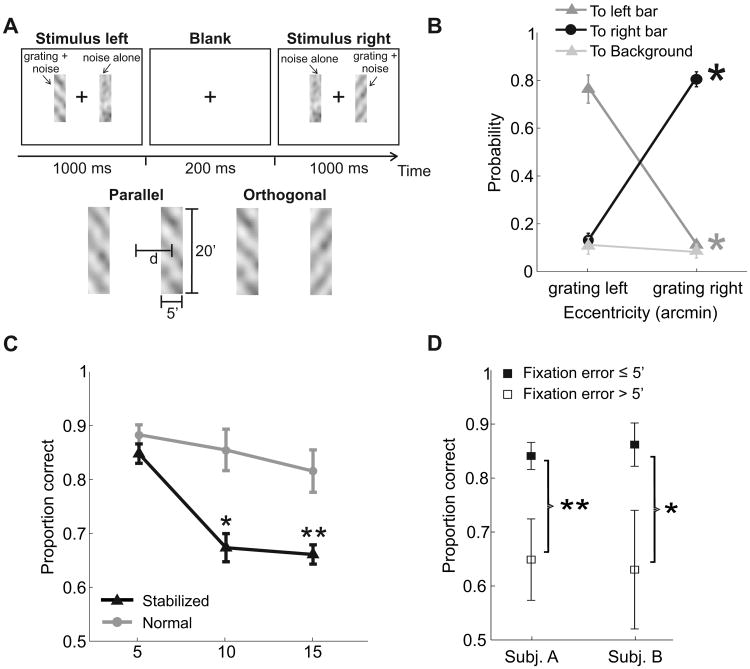
(A) In a forced-choice task, subjects reported whether two gratings (11 cycles/degree tilted by ±45°) were parallel or orthogonal. Gratings appeared within two rectangular noise bars centered at the desired eccentricity d. They were displayed sequentially first in the left and then in the right bar while subjects maintained fixation at the center of the display (cross). (B) Proportions of microsaccades landing on one of the two bars at 15′ eccentricity and on the background region during the two periods of grating presentation. Error bars represent SEM. Asterisks mark significant differences between the probabilities of landing in a given region of the image in the two temporal intervals (p < 0.01; two-tailed paired t test). (C) Average subject performance (N=4) as a function of the stimulus eccentricity in the two conditions. In each condition, asterisks mark statistically significant differences with respect to the proportions of correct responses at 5′ (*p < 0.05; **p < 0.005; two-tailed paired t test). (D) Comparison of discrimination performances in the trials in which microsaccades brought the center of gaze within 5′ from both bars (Fixation error ≤5′) relative to the trials in which microsaccades did occur but were not as effective in bringing the center of gaze on the stimulus (Fixation error > 5′). Data refer to normal (unstabilized) viewing of stimuli at 15′ eccentricity. Results for two subjects are shown. Adapted from Poletti et al, (2013)
Many other observations, not reviewed here, also speak against the idea that image fading represents a concern for the visual system and/or an important function of microsaccades during natural viewing. In our view, while much has certainly happened, little seems to have actually changed since Nachmias's conclusion many years ago that: “fading prevention might just be an incidental, if not accidental role of microsaccades” (Nachmias, 1961).
3.3 Microsaccades and ne spatial vision
It was first proposed by Cunitz and Steinman (1969) that microsaccades might serve to precisely redirect a preferred small locus on the retina. These authors wrote: “We would like to propose that many “fixation” microsaccades serve the same purpose of large “voluntary” saccades, viz they are used to make very small shifts in the line of regard so that an attended feature in the visual array falls at a preferred locus within the center of best vision” (from Cunitz and Steinman, 1969).
This idea was later left aside following reports that microsaccades tend to be suppressed during the execution of finely guided visuomotor tasks. Winterson and Collewijn (1976) noticed that, when observers threaded a sewing needle or aimed a rifle, microsaccade rates decreased just before the end of a trial, even when the task was performed successfully. Microsaccades were less frequent in these tasks than during sustained fixation. Similar results were also reported by Bridgeman and Palca (1980) in a comparable, but purely visual, task. These findings led to the notion that microsaccades are detrimental for high-acuity vision.
Several observations, however, caution that this conclusion might have been premature. First, as observed before, the artificial condition of sustained fixation elicits frequent microsaccades in many observers and may not serve as the optimal reference baseline. Second, a reduction in the rate of microsaccades when the thread enters the needle could be caused by the need to avoid peri-saccadic perceptual impairments (such as saccadic suppression and mislocalization) at a critical time in the task, or by other unrelated factors, such as a change in attention at the completion of the task. It does not imply that the microsaccades occurring at earlier times did not provide useful information. Was Cunitz and Steinman's proposal discarded too quickly?
3.3.1 Threading a needle
To reexamine the role of microsaccades in fine spatial vision, we conducted an experiment similar to those of the previous studies, while using modern technology to better control experimental conditions and more closely examine the impact of microsaccades. In this experiment, observers threaded a needle in a virtual environment. As illustrated in Fig. 6A, they used a joystick to insert a thin horizontal bar (the thread) into a small aperture at the center of a stationary vertical bar (the needle). Like the real threading of a needle, the stimulus was very small, covering an area smaller than 1 deg2.
Figure 6. Microsaccades precisely relocate gaze.
(A) Subjects moved a horizontal bar (the thread) toward the small gap in a vertical bar (the needle). The panel on the right and its enlargement show an example of the spatial distribution of fixations. Fixations were primarily allocated to the thread (blue circles) and the eye of the needle (green circles). The red crosses mark the thread trajectory. (B) Mean instantaneous frequency and amplitude of microsaccades during the course of the trial. The trial starts at t = 0 and lasts 17.5 s. The two horizontal lines represent mean microsaccade rates during sustained fixation (dashed line) and free viewing of natural scenes (dotted line). (C) Starting and landing points of microsaccades. (D) conditional probabilities of adjustments following different types of microsaccades. All data refer to saccades smaller than 20′. Error bars represent SEM. Adapted from Ko et al. (2010).
This experimental setup enabled replication of the previous results in the literature when their specific conditions were implemented. In agreement with previous reports, the frequency of microsaccades decreased toward the end of the trial when the thread reached the needle as in Winterson and Collewijn (1976), or when the subject maintained fixation on the eye of the needle for the entire duration of the trial as in Bridgeman and Palca (1980). The mean rate of microsaccades was also lower than that measured during sustained fixation, as previously reported. However, microsaccades were frequent at earlier times in a trial, when the thread had not reached the needle and subjects were still involved in adjusting its alignment. Furthermore, the microsaccade rate was five times higher than the rate exhibited by the same observers while free-viewing natural images. Thus, the condition chosen as reference is critical for evaluating microsaccade frequency.
To better examine the effects of microsaccades, we took several precautions. First, we restricted motion control of the thread to the vertical axis to ensure that changes in the thread position reflected a perceived misalignment. On the horizontal axis, the thread approached the needle at slow constant speed. Second, to avoid possible changes in attention at the completion of the task, we terminated each trial when the thread was close to, but still had not reached, the needle. Third, we used EyeRIS, our system for gaze-contingent control (Santini et al., 2007), together with the gaze-contingent calibration developed in our laboratory (Fig. 3) to precisely localize microsaccades both in time and space. This enabled us to determine how microsaccades moved the line of sight and their timing of occurrence relative to the adjustments in the thread-needle alignment.
Our results are summarized in Fig. 6. Microsaccades became progressively smaller during the course of a trial, a change that reflected the progressive closing of the distance between the thread and the needle. They also became increasingly more frequent as the thread approached the needle (Fig. 6B). Thus, the characteristics of microsaccades followed ongoing changes in the stimulus. Careful localization of the line of sight revealed that microsaccades shifted gaze in a very precise manner. We classified each intersaccadic interval as a fixation on a different part of the image according to the mean eye position. Fixations were clustered in specific and salient locations of the image: most fixations were either on the thread or around the eye of the needle. Very few fixations fell far from these two regions. Microsaccades were often used to relocate the line of sight between the thread and the needle. They very rarely landed on other regions of the scene (Fig. 6C).
These results suggest that observers used microsaccades to acquire useful information from the scene. To confirm this hypothesis, we examined whether a link existed between microsaccades and adjustments in the thread's position. We estimated the probabilities of correcting the thread/needle alignment immediately after a microsaccade and the probabilities of performing microsaccades immediately after changing the thread's position. We found that subjects were more likely to realign the thread after executing a microsaccade that shifted the preferred retinal locus from one object (the thread or the needle) to the other (see Fig. 6D). In contrast, microsaccades were likely to move the preferred retinal locus toward the thread after executing an adjustment. These data corroborate the idea that subjects performed microsaccades to judge the alignment between the thread and the needle. They provide strong support to Cunitz and Steinman's proposal that microsaccades are exploratory movements like larger saccades.
3.3.2 Why microsaccades?
The findings described in the previous section raise an important question. As well expressed by Kowler and Collewijn in a commentary article: “A judiciously chosen central fixation location should have provided the vantage point needed to evaluate the relative position of the critical details (needle and thread) and permit the appropriate adjustments. […] Subjects preferred to make saccades. Why? What was the benefit of this strategy?” (Kowler and Collewijn, 2010).
A possible answer to this question is that the preferred retinal locus centered by microsaccades facilitates fine spatial judgments. This proposal would provide an explanation not only for the results of Fig. 6, but also for previously reported improvements in fine spatial discrimination measured in the presence of microsaccades (Rucci and Desbordes, 2003). Support for this proposal comes from anatomical examinations of the human retina. While individual variability exists, the region of maximum cone concentration is on average restricted to an area with a radius of approximately 6′. Outside this region, cone density falls steeply with increasing eccentricity even within the foveola (Curcio et al., 1990). Thus, fine spatial vision may benefit by properly centering this region.
Yet, this idea conflicts with the common assumption that, unlike in the periphery, visual functions within the central fovea are approximately uniform. In fact, direct psychophysical evaluation with stimuli at very small eccentricity angles has given mixed results. Some studies have found a decline in performance with eccentricity, whereas others have reported minimal changes (Weymouth et al., 1928; Millodot, 1972). Mapping foveal vision is, however, a very challenging task. Fixational eye movements yield—as described in Fig. 3—a region of uncertainty in gaze localization and also move the stimulus over many photoreceptors, if brief flashes are not used. Both effects effectively prevent isolation of closely spaced regions on the retina and are likely to homogenize measurements at adjacent locations. These factors may have contributed to the notion of approximately uniform foveal vision.
To determine whether fine spatial vision is optimal at the preferred retinal locus, we recently developed a forced-choice task designed to confine stimulation at fixed eccentricities, and relied on the higher precision given by methods for gaze-contingent display to map performance across the foveola. In this experiment, subjects reported whether two sequentially presented bars (two narrow rectangular noise fields) contained gratings that were parallel or orthogonal (Fig. 7A)
Fig. 7C shows performance when subjects were simply asked to maintain fixation at the center of the display and stimuli were presented at three different, but very small distances from this point. Results varied little with eccentricity. Percentages of correct discrimination with stimuli at 10′ and 15′ from the point of fixation were very similar to those measured with bars at only 5′. Without recording eye movements, these data would appear to give support to the idea that vision is approximately uniform within the foveola. However, close examination of fixational eye movements revealed that, as in the experiment of Fig. 6, microsaccades were not random, but were driven by the stimulus and the ongoing demands of the task. Subjects used microsaccades to shift the line of sight toward the stimulus (Fig. 7B). They looked to the left side of the monitor when the grating appeared in the left bar and to the right side of the monitor when the grating was displayed in the right bar. This difference in gaze position was visible even with stimuli at 5′ eccentricity. Thus, stimuli did not remain at a fixed eccentricity on the retina, but moved, because of microsaccades, toward the preferred retinal locus.
Fig. 7C also shows performance in the same task when stimuli were forced to remain at their fixed retinal eccentricity by means of retinal stabilization. In this condition, the display was updated in real time according to the subject's eye movements to maintain the stimulus immobile on the retina, thus avoiding contamination from multiple retinal regions. Percentages of correct judgments now dropped rapidly with eccentricity, and reduction in performance was already present at just 10′ eccentricity. This effect was caused by the absence of precise microsaccades: it was also visible in the normal (unstabilized) condition both in the rare trials in which microsaccades did not occur and also when microsaccades did occur but failed to bring the projection of the stimulus onto the preferred retinal locus of fixation. Performance was significantly lower in the trials in which microsaccades did not shift the center of gaze within 5′ from the stimulus (Fig. 7D). Thus, the perceptual benefit of microsaccades was not given by their temporal transients or a general “refresh” of the image associated with microsaccades. It was the consequence of precisely positioning the stimulus of interest on the retina.
These results show that (a) fine spatial vision deteriorates very rapidly with eccentricity outside of a preferred retinal locus; (b) observers precisely relocate this locus by means of microsaccades in high-acuity tasks. These small gaze shifts appear useful in the examination of fine spatial detail.
4 Some conclusions and some open questions
Following a period of incubation, the study of fixational eye movements has reemerged to a prominent place within the vision research community. This new wave of studies has generated a wealth of information. The neural mechanisms responsible for the generation of microsaccades, their impact on the responses of neurons in the visual system, and their consequences for vision are becoming progressively more clear.
Much of this work has emphasized the similarity between microsaccades and larger saccades, raising the question of whether it continues to make sense to preserve the traditional distinction between these two categories of movements. In addition to the functional similarity summarized in this article, it is now known that microsaccades and saccades are mediated by the same neural structures and motor mechanisms (Hafed et al., 2009). Furthermore, similar visually-driven and extraretinal modulations have been observed in neurons in the primary visual cortex for both fixational saccades and small voluntary saccades (Kagan et al., 2008). Even phenomena that were once considered to be exclusive domain of larger saccades, such as saccadic adaptation or peri-saccadic perceptual alterations (Hafed, 2013; Havermann et al., 2014), are now known to extend to the range of microsaccades.
In this article we have examined some of the challenges intrinsic to the study of microsaccades and expressed our viewpoint on some of the proposed functions. The difficulties in (1) recording small eye movements, (2) extrapolating results obtained under artificial laboratory conditions, and (3) determining how exactly microsaccades move the stimulus on the retina are some of the obstacles faced by researchers in the field. A real need exists for developing better and more standardized procedures to ensure recordings of high quality. We have argued that this process implies going beyond the eyetracker specifications, both because the manufacturer's data may not be accurate and because they may refer to ideal conditions that are very different from those of a real experiment. We remind the reader that the internal noise of an eyetracker is only one among the many elements contributing to the system's precision and advocate for the use of an artificial eye controlled by a high-precision manipulator to get more realistic estimation of resolution. While no ideal method exists, and recordings of an artificial and a real eye differ in many ways, a precisely controlled artificial eye provides a first important step toward a better characterization of the ideal limits of the eyetracker.
Another concern that we have addressed in this article is dealing with a definition of microsaccade that relies on volition. While the emerging body of literature indicates that a distinction between saccades and microsaccades is contrived under natural viewing conditions, it is in practice useful to have a dedicated term for labeling the eye movements that keep the stimulus within the central high-acuity region of the visual field. Rather than using amplitude thresholds based on debated distributions of “involuntary” saccades, we have suggested a more rigorous definition that assigns the term microsaccade to the saccades with amplitudes within two boundaries: an upper amplitude thresholds of 30′, which ensures that the overlap in the images contained within the foveola is above 50%; and a study-specific lower amplitude threshold, which excludes unreliable oculomotor events. This lower threshold depends on the characteristics of the eyetracker used in the study and should be set above the minimum rotation that can be reliably detected with an artificial eye controlled by a galvanometer.
In the previous pages, we have also summarized our view that, under natural viewing conditions, no functional distinction exists between most microsaccades and larger saccades. Microsaccades are an important element in the toolbox of oculomotor behaviors, as no other body movement, including movements of the head and eye, can shift retinal projections of objects within the fovea as rapidly and as accurately as a microsaccade. Perhaps, an obstacle to this realization has been an intuitive difficulty in visualizing angular eye rotations in terms of spatial shifts of the gaze location in the scene. To help forming an intuition, Fig. 8 provides various examples in which we routinely perform microsaccades of various sizes to shift the gaze across salient objects or features. According to the results of our recent experiments (Ko et al., 2010; Poletti et al., 2013), microsaccades are beneficial in many tasks in which stimuli are viewed at a distance. Practical examples include deciphering the facial expressions of people a few meters away from us, reading traffic signs, following the action at a sport event, and following a theater performance with the tickets that many researchers can afford nowadays.
Figure 8. Spatial consequences of microsaccades.
Displacements caused by microsaccades of various amplitudes when looking at objects at different distances. The circles refer to the three real-life examples illustrated in the figures on the margins.
While much progress has been made in understanding the functions of small eye movements, fundamental aspects remain unclear. We like to conclude this article with three open questions:
Is a poor control of microsaccades responsible for impairment in fine spatial vision? The finding that microsaccades precisely move a preferred retinal locus suggests that fine spatial vision may be compromised when microsaccades are not well executed. This raises the possibility for a causal relationship between the abnormal fixational eye movements and the reduced visual acuity that coexist in various disorders (Ciuffreda et al., 1979; Wark et al., 2008; Parkinson and Maxner, 2005; Abadi and Pascal, 1991; Simmers et al., 1999; Tilikete et al., 2011). Further research is necessary to investigate this emerging link between perception and action.
What is special about the preferred retinal locus? For simplicitly, in this article we have let the reader assume that the preferred retinal locus of fixation coincide with the region of highest cone density. But it is known that the center of the preferred retinal locus of fixation can be as far as 10′ from the point of highest cone density (Putnam et al., 2005). It is not clear what determines a preferred retinal locus, nor whether a link exists between individual variability in retinal anatomy (Curcio et al., 1990) and idiosyncratic patterns of microsaccades (Cherici et al., 2012).
How do head movements influence microsaccades? With possibly one exception (Epelboim et al., 1995), current eyetrackers require head immobilization to give reliable measurements of very small eye movements. It has long been argued that head immobilization may affect oculomotor activity (Skavenski et al., 1979; Epelboim et al., 1995) and even suggested that very small saccades may be laboratory artifacts (Malinov et al., 2000; Steinman et al., 2003). However, these pioneering head-free studies did not focus on the high-acuity tasks in which microsaccades are expected to be helpful. New experiments are necessary to investigate this question. More in general, it is time for the community to start investigating microsaccades under more natural settings without having to sacrifice the quality of oculomotor recordings.
Highlights.
Progress has been hindered by measurement artifacts and unnatural viewing conditions
The characteristics of commercial eye trackers should be evaluated more rigorously
During natural viewing, microsaccades precisely move a high-acuity retinal locus
Microsaccades and saccades serve similar visual functions
Acknowledgments
This work was supported by National Institutes of Health grant EY18363 and National Science Foundation grants 1127216 and 1420212.
Footnotes
Simply taking the amplitude of the largest saccade is clearly not a good strategy, as errors will always be present. For example, blinks may have been erroneously classified as saccades, or the subject may have been momentarily distracted and looked away from the fixation cue. These events will appear in the saccade amplitude distribution, but one does not want to count them in the definition of microsaccades. The use of a sufficiently high percentile solves this problem.
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- Abadi RV, Pascal E. Visual resolution limits in human albinism. Vision Research. 1991;21:1445–1447. doi: 10.1016/0042-6989(91)90063-b. [DOI] [PubMed] [Google Scholar]
- Arend LE, Timberlake GT. What is psychophysically perfect image stabilization? Do perfectly stabilized images always disappear? Journal of Optical Society of America, Series A. 1986;3:235–241. doi: 10.1364/josaa.3.000235. [DOI] [PubMed] [Google Scholar]
- Aytekin M, Victor JD, Rucci M. The visual input to the retina during natural head-free fixation. Journal of Neuroscience. 2014;34:12701–12715. doi: 10.1523/JNEUROSCI.0229-14.2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Beeler GWJ. Visual threshold changes resulting from spontaneous saccadic eye movements. Vision Research. 1967;7:769–775. doi: 10.1016/0042-6989(67)90039-9. [DOI] [PubMed] [Google Scholar]
- Blignaut P, Wium D. Eye-tracking data quality as affected by ethnicity and experimental design. Behavior Research Methods. 2014;46:67–80. doi: 10.3758/s13428-013-0343-0. [DOI] [PubMed] [Google Scholar]
- Boyce PR. Monocular fixation in human eye movement. Proceedings of the Royal Society of London Series B, Biological Sciences. 1967;167:293–315. doi: 10.1098/rspb.1967.0028. [DOI] [PubMed] [Google Scholar]
- Bridgeman B, Palca J. The role of microsaccades in high acuity observational tasks. Vision Research. 1980;20:813–817. doi: 10.1016/0042-6989(80)90013-9. [DOI] [PubMed] [Google Scholar]
- Cherici C, Kuang X, Poletti M, Rucci M. Precision of sustained fixation in trained and untrained observers. Journal of Vision. 2012;12:1–16. doi: 10.1167/12.6.31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Choe KW, Blake R, Lee SH. Correcting video-based eye tracking signals for pupil size artifacts. Journal of Vision. 2014;14:754. [Google Scholar]
- Ciuffreda KJ, Kenyon RV, Stark L. Fixational eye movements in amblyopia and strabismus. Journal of the American Optometric Association. 1979;50:1251–1258. [PubMed] [Google Scholar]
- Collewijn H, Kowler E. The significance of microsaccades for vision and oculomotor control. Journal of Vision. 2008;8:1–21. doi: 10.1167/8.14.20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Collewijn H, van der Mark F, Jansen TC. Precise recording of human eye movements. Vision Research. 1975;15:447–450. doi: 10.1016/0042-6989(75)90098-x. [DOI] [PubMed] [Google Scholar]
- Coppola D, Purves D. The extraordinarily rapid disappearance of entopic images. Proceedings of the National Academy of Sciences, USA. 1996;93:8001–8004. doi: 10.1073/pnas.93.15.8001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cornsweet TN. Determination of the stimuli for involuntary drifts and saccadic eye movements. Journal of the Optical Society of America. 1956;46:987–988. doi: 10.1364/josa.46.000987. [DOI] [PubMed] [Google Scholar]
- Crane HD, Steele C. Generation V Dual Purkinje-Image eyetracker. Applied Optics. 1985;24:527–537. doi: 10.1364/ao.24.000527. [DOI] [PubMed] [Google Scholar]
- Cunitz RJ, Steinman RM. Comparison of saccadic eye movements during fixation and reading. Vision Research. 1969;9:683–693. doi: 10.1016/0042-6989(69)90125-4. [DOI] [PubMed] [Google Scholar]
- Curcio CA, Sloan KR, Kalina RE, Hendrickson AE. Human photoreceptor topography. Journal of Comparative Neurology. 1990;292:497–523. doi: 10.1002/cne.902920402. [DOI] [PubMed] [Google Scholar]
- Deubel H, Bridgeman B. Perceptual consequences of ocular lens overshoot during saccadic eye movements. Vision Research. 1995;35:2897–2902. doi: 10.1016/0042-6989(95)00042-x. [DOI] [PubMed] [Google Scholar]
- Deubel H, Schneider WX. Saccade target selection and object recognition: Evidence for a common attentional mechanism. Vision Research. 1996;36:1827–1837. doi: 10.1016/0042-6989(95)00294-4. [DOI] [PubMed] [Google Scholar]
- Ditchburn RW. What is psychophysically perfect image stabilization? Do perfectly stabilized images always disappear?: Comment. Journal of Optical Society of America, Series A. 1987;4:405–406. doi: 10.1364/josaa.4.000405. [DOI] [PubMed] [Google Scholar]
- Ditchburn RW, Fender DH, Mayne S. Vision with controlled movements of the retinal image. Journal of Physiology. 1959;145:98–107. doi: 10.1113/jphysiol.1959.sp006130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ditchburn RW, Foley-Fisher JA. Assembled data in eye movements. Optica Acta. 1967;14:113–118. doi: 10.1080/713818024. [DOI] [PubMed] [Google Scholar]
- Ditchburn RW, Ginsborg BL. Vision with a stabilized retinal image. Nature. 1952;170:36–37. doi: 10.1038/170036a0. [DOI] [PubMed] [Google Scholar]
- Ditchburn RW, Ginsborg BL. Involuntary eye movements during fixation. The Journal of Physiology. 1953;119:1–17. doi: 10.1113/jphysiol.1953.sp004824. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dorris MC, Munoz DP. A neural correlate for the gap effect on saccadic reaction times in monkey. Journal of Neurophysiology. 1995;73:2558–2562. doi: 10.1152/jn.1995.73.6.2558. [DOI] [PubMed] [Google Scholar]
- Drewes J, Masson GS, Montagnini A. Shifts in reported gaze position due to changes in pupil size: Ground truth and compensation. Proceedings of the Symposium on Eye Tracking Research and Applications, ETRA; New York, NY, USA. 2012. p. 209212. [Google Scholar]
- Engbert R, Kliegl R. Microsaccades keep the eyes' balance during fixation. Psychological Science. 2004;15:431–436. doi: 10.1111/j.0956-7976.2004.00697.x. [DOI] [PubMed] [Google Scholar]
- Epelboim J, Kowler E, Steinman RM, Collewijn H, Erkelens CJ, Pizlo Z. When push comes to shove: Compensation for passive perturbation of the head during natural gaze shifts. Journal of Vestibular Research. 1995;5:421–442. [PubMed] [Google Scholar]
- Fiorentini A, Ercoles AM. Involuntary eye movements during attempted monocular fixation. Atti Fondazione Giorgio Ronchi. 1966:199–217. [Google Scholar]
- Gerrits HJ, Vendrik AJ. Artificial movements of a stabilized image. Vision Research. 1970;10:1443–1456. doi: 10.1016/0042-6989(70)90094-5. [DOI] [PubMed] [Google Scholar]
- Goffart L, Hafed ZM, Krauzlis RJ. Visual fixation as equilibrium: Evidence from superior colliculus inactivation. Journal of Neuroscience. 2012;32:10627–10636. doi: 10.1523/JNEUROSCI.0696-12.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haddad GM, Steinman RM. The smallest voluntary saccade: Implications for fixation. Vision Research. 1973;13:1075–1086. doi: 10.1016/0042-6989(73)90145-4. [DOI] [PubMed] [Google Scholar]
- Hafed ZM. Alteration of visual perception prior to microsaccades. Neuron. 2013;77:775–786. doi: 10.1016/j.neuron.2012.12.014. [DOI] [PubMed] [Google Scholar]
- Hafed ZM, Goffart L, Krauzlis RJ. A neural mechanism for microsaccade generation in the primate superior colliculus. Science. 2009;323:940–943. doi: 10.1126/science.1166112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hafed ZM, Krauzlis RJ. Similarity of superior colliculus involvement in microsaccade and saccade generation. Journal of Neurophysiology. 2012;107:1904–1916. doi: 10.1152/jn.01125.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Havermann K, Cherici C, Rucci M, Lappe M. Fine-scale plasticity of microscopic saccades. Journal of Neuroscience. 2014;34:11665–11672. doi: 10.1523/JNEUROSCI.5277-13.2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Herrington TM, Masse NY, Hachmeh KJ, Smith JE, Assad JA, Cook EP. The effect of microsaccades on the correlation between neural activity and behavior in middle temporal, ventral intraparietal, and lateral intraparietal areas. Journal of Neuroscience. 2009;29:5793–57805. doi: 10.1523/JNEUROSCI.4412-08.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Holmqvist K, Nyström M, Andersson R, Dewhurst R, Jarodzka H, de Weijer JV. Eye tracking: A comprehensive guide to methods and measures. Authors Oxford University Press; 2011. [Google Scholar]
- Horn MRV, Cullen KE. Coding of microsaccades in three-dimensional space by premotor saccadic neurons. Journal of Neuroscience. 2012;32:1974–1980. doi: 10.1523/JNEUROSCI.5054-11.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Houben MM, Goumans J, van der Steen J. Recording three-dimensional eye movements: scleral search coils versus video oculography. Investigative Ophthalmology & Visual Science. 2006;47:179–187. doi: 10.1167/iovs.05-0234. [DOI] [PubMed] [Google Scholar]
- Janabi-Sharifi F, Hayward V, Chen CJ. Discrete-time adaptive windowing for velocity estimation. IEEE Transactions on Control Systems Technology. 2000;8:1003–1009. [Google Scholar]
- Kagan I. Microsaccades and image fading during natural vision. Electronic response to McCamy et al. microsaccadic efficacy and contribution to foveal and peripheral vision. Journal of Neuroscience. 2012;32:9194–9204. doi: 10.1523/JNEUROSCI.0515-12.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kagan I, Gur M, Snodderly DM. Saccades and drifts differentially modulate neuronal activity in V1: Effects of retinal image motion, position, and extraretinal influences. Journal of Vision. 2008;8:1–25. doi: 10.1167/8.14.19. [DOI] [PubMed] [Google Scholar]
- Kelly DH. Motion and vision. I. Stabilized images of stationary gratings. Journal of Optical Society of America. 1979;69:1266–1274. doi: 10.1364/josa.69.001266. [DOI] [PubMed] [Google Scholar]
- Kliegl R, Rolfs M, Laubrock J, Engbert R. Microsaccadic modulation of response times in spatial attention tasks. Psychological Research. 2009;73:136–146. doi: 10.1007/s00426-008-0202-2. [DOI] [PubMed] [Google Scholar]
- Ko HK, Poletti M, Rucci M. Microsaccades precisely relocate gaze in a high visual acuity task. Nature Neuroscience. 2010;13:1549–1553. doi: 10.1038/nn.2663. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koenderink JJ. Contrast enhancement and the negative afterimage. Journal of the Optical Society of America. 1972;62:685–689. doi: 10.1364/josa.62.000685. [DOI] [PubMed] [Google Scholar]
- Kolakowski S, Pelz JB. Compensating for eye tracker camera movement. Proceedings of the ACM SIGCHI Eye Tracking Research & Applications Symposium; San Diego, California, United States. 2006. [Google Scholar]
- Kowler E, Collewijn H. The eye on the needle. Nature Neuroscience. 2010;13:1443–1444. doi: 10.1038/nn1210-1443. [DOI] [PubMed] [Google Scholar]
- Kowler E, Steinman RM. Small saccades serve no useful purpose: Reply to a letter by R. W. ditchburn. Vision Research. 1980;20:273–276. doi: 10.1016/0042-6989(80)90113-3. [DOI] [PubMed] [Google Scholar]
- Kuang X, Poletti M, Victor JD, Rucci M. Temporal encoding of spatial information during active visual fixation. Current Biology. 2012;20:510–514. doi: 10.1016/j.cub.2012.01.050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Malinov IV, Epelboim J, Herst AN, Steinman RM. Characteristics of saccades and vergence in two kinds of sequential looking tasks. Vision Research. 2000;40:2083–2090. doi: 10.1016/s0042-6989(00)00063-8. [DOI] [PubMed] [Google Scholar]
- Martinez-Conde S, Macknik SL, Troncoso XG, Dyar TA. Microsaccades counteract fading during fixation. Neuron. 2006;49:297–305. doi: 10.1016/j.neuron.2005.11.033. [DOI] [PubMed] [Google Scholar]
- McCamy MB, Otero-Millan J, Macknik SL, Yang Y, Troncoso XG, Baer SM, Crook SM, Martinez-Conde S. Microsaccadic efficacy and contribution to foveal and peripheral vision. Journal of Neuroscience. 2012;32:9194–9204. doi: 10.1523/JNEUROSCI.0515-12.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Millodot M. Variation of visual acuity in the central region of the retina. The British Journal of Physiological Optics. 1972;27:24–28. [PubMed] [Google Scholar]
- Møller F, Laursen ML, Sjølie AK. The contribution of microsaccades and drifts in the maintenance of binocular steady fixation. Graefe's Archive for Clinical and Experimental Ophthalmology. 2006;244:465–471. doi: 10.1007/s00417-004-0989-5. [DOI] [PubMed] [Google Scholar]
- Mostofi N, Boi M, Rucci M. Influence of microsaccades on contrast sensitivity: Theoretical analysis and experimental results. Journal of Vision. 2014;14:109. [Google Scholar]
- Nachmias J. Two-dimensional motion of the retinal image during monocular fixation. Journal of the Optical Society of America. 1959;49:901–908. doi: 10.1364/josa.49.000901. [DOI] [PubMed] [Google Scholar]
- Nachmias J. Determiners of the drift of the eye during monocular fixation. Journal of the Optical Society of America. 1961;51:761–766. doi: 10.1364/josa.51.000761. [DOI] [PubMed] [Google Scholar]
- Nyström M, Andersson R, Holmqvist K, van de Weijer J. The influence of calibration method and eye physiology on eyetracking data quality. Behavior Research Methods. 2013;45:272–288. doi: 10.3758/s13428-012-0247-4. [DOI] [PubMed] [Google Scholar]
- Parkinson J, Maxner C. Eye movement abnormalities in alzheimer disease: Case presentation and literature review. The American Orthopic Journal. 2005;55:90–96. doi: 10.3368/aoj.55.1.90. [DOI] [PubMed] [Google Scholar]
- Poletti M, Listorti C, Rucci M. Microscopic eye movements compensate for nonhomogeneous vision within the fovea. Current Biology. 2013;23:1691–1695. doi: 10.1016/j.cub.2013.07.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Poletti M, Rucci M. Fixational eye movements under various conditions of image fading. Journal of Vision. 2010;10(3):1–18. doi: 10.1167/10.3.6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Privitera CM, Carney T, Klein S, Aguilar M. Analysis of microsaccades and pupil dilation reveals a common decisional origin during visual search. Vision Research. 2014;95:43–50. doi: 10.1016/j.visres.2013.12.001. [DOI] [PubMed] [Google Scholar]
- Putnam NM, Hofer HJ, Doble N, Chen L, Carroll J, Williams DR. The locus of fixation and the foveal cone mosaic. Journal of Vision. 2005;7:632–639. doi: 10.1167/5.7.3. [DOI] [PubMed] [Google Scholar]
- Riggs LA, Ratliff F. The effects of counteracting the normal movements of the eye. Journal of the Optical Society of America. 1952;42:872–873. [Google Scholar]
- Robinson DA. A method of measuring eye movement using scleral seach coil in a megnetic field. IEEE Transactions on Biomedical Engineering. 1963;10:137–145. doi: 10.1109/tbmel.1963.4322822. [DOI] [PubMed] [Google Scholar]
- Rolfs M. Microsaccades: Small steps on a long way. Vision Research. 2009;49:2415–2441. doi: 10.1016/j.visres.2009.08.010. [DOI] [PubMed] [Google Scholar]
- Rucci M, Desbordes G. Contributions of fixational eye movements to the discrimination of briefly presented stimuli. Journal of Vision. 2003;3:852–864. doi: 10.1167/3.11.18. [DOI] [PubMed] [Google Scholar]
- Rucci M, Iovin R, Poletti M, Santini F. Miniature eye movements enhance fine spatial detail. Nature. 2007;447:852–855. doi: 10.1038/nature05866. [DOI] [PubMed] [Google Scholar]
- Santini F, Redner G, Iovin R, Rucci M. EyeRIS: A general-purpose system for eye movement contingent display control. Behavioral Research Methods. 2007;39:350–364. doi: 10.3758/bf03193003. [DOI] [PubMed] [Google Scholar]
- Simmers AJ, Gray LS, Winn B. The effect of abnormal fixational eye movements upon visual acuity in congenital nystagmus. Current Eye Research. 1999;18:194–202. doi: 10.1076/ceyr.18.3.194.5374. [DOI] [PubMed] [Google Scholar]
- Skavenski AA, Hansen RM, Steinman RM, Winterson BJ. Quality of retinal image stabilization during small natural and artificial body rotations in man. Vision Research. 1979;19:675–683. doi: 10.1016/0042-6989(79)90243-8. [DOI] [PubMed] [Google Scholar]
- St Cyr GJ, Fender DH. The interplay of drifts and flicks in binocular fixation. Vision Research. 1969;9:245–265. doi: 10.1016/0042-6989(69)90004-2. [DOI] [PubMed] [Google Scholar]
- Steinman RM. Gaze control under natural conditions. In: Chalupa LM, Werner JS, editors. The Visual Neurosciences. MIT Press; Cambridge: 2003. [Google Scholar]
- Steinman RM, Haddad GM, Skavenski AA, Wyman D. Miniature eye movement. Science. 1973;181:810–819. doi: 10.1126/science.181.4102.810. [DOI] [PubMed] [Google Scholar]
- Steinman RM, Pizlo Z, Forofonova TI, Epelboim J. One fixates accurately in order to see clearly not because one sees clearly. Spatial Vision. 2003;16:225–241. doi: 10.1163/156856803322467509. [DOI] [PubMed] [Google Scholar]
- Steinman R, Cunitz R, Timberlake G, Herman M. Voluntary control of microsaccades during maintained monocular fixation. Science. 1967;155:1577–1579. doi: 10.1126/science.155.3769.1577. [DOI] [PubMed] [Google Scholar]
- Stevens JK, Emerson RC, Gerstein GL, Kallos T, Neufeld GR, Nichols CW, Rosenquist AC. Paralysis of the awake human: Visual perceptions. Vision Research. 1976;16:93–98. doi: 10.1016/0042-6989(76)90082-1. [DOI] [PubMed] [Google Scholar]
- Stevenson SB, Roorda A. Biomedical Optics 2005. International Society for Optics and Photonics; 2005. Correcting for miniature eye movements in high resolution scanning laser ophthalmoscopy; pp. 145–151. [Google Scholar]
- Thaler L, Schtz AC, Goodale MA, Gegenfurtner KR. What is the best fixation target? The effect of target shape on stability of fixational eye movements. Vision Research. 2013;76:31–42. doi: 10.1016/j.visres.2012.10.012. [DOI] [PubMed] [Google Scholar]
- Tilikete C, Jasse L, Vukusic S, Durand-Dubief F, Vardanian C, Plisson D, Vighetto A. Persistent ocular motor manifestations and related visual consequences in multiple sclerosis. Annals of the New York Academy of Sciences. 2011;1233:327–334. doi: 10.1111/j.1749-6632.2011.06116.x. [DOI] [PubMed] [Google Scholar]
- Timberlake GT, Wyman D, Skavenski AA, Steinman RM. The oculomotor error signal in the fovea. Vision Research. 1972;12:1059–1064. doi: 10.1016/0042-6989(72)90027-2. [DOI] [PubMed] [Google Scholar]
- Tulunay-Keesey U. Fading of stabilized retinal images. Journal of the Optical Society of America. 1982;72:440–447. doi: 10.1364/josa.72.000440. [DOI] [PubMed] [Google Scholar]
- Valsecchi M, Betta E, Turatto M. Visual oddballs induce prolonged microsaccadic inhibition. Experimental Brain Research. 2007;177:196–208. doi: 10.1007/s00221-006-0665-6. [DOI] [PubMed] [Google Scholar]
- von Wartburg R, Wurtz P, Pflugshaupt T, Nyffeler T, Lthi M, Mri RM. Size matters: Saccades during scene perception. Perception. 2007;36:355–365. doi: 10.1068/p5552. [DOI] [PubMed] [Google Scholar]
- Wark HA, Garell PC, Walker AL, Basso MA. A case report on fixation instability in parkinson's disease with bilateral deep brain stimulation implants. Journal of Neurology, Neurosurgery & Psychiatry. 2008;79:443–447. doi: 10.1136/jnnp.2007.117192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weymouth FW, Hines DC, Acres LH, Raaf JE, Wheeler MC. Visual acuity within the area centralis and its relation to eye movements and fixation. American Journal of Ophthalmology. 1928;11:947–960. [Google Scholar]
- Whitham EM, Fitzgibbon SP, Lewis TW, Pope KJ, Delosangeles D, Clark CR, Lillie P, Hardy A, Gandevia SC, Willoughby JO. Visual experiences during paralysis. Frontiers in Human Neuroscience. 2011;160 doi: 10.3389/fnhum.2011.00160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wildenmann U, Schaeffel F. Variations of pupil centration and their effects on video eye tracking. Ophthalmic and Physiological Optics. 2013;33:634–641. doi: 10.1111/opo.12086. [DOI] [PubMed] [Google Scholar]
- Winterson B, Collewijn H. Microsaccades during finely guided visuomotor tasks. Vision Research. 1976;16:1387–1390. doi: 10.1016/0042-6989(76)90156-5. [DOI] [PubMed] [Google Scholar]
- Wyatt HJ. The human pupil and the use of video-based eyetrackers. Vision Research. 2010;50:1982–1988. doi: 10.1016/j.visres.2010.07.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wyman D, Steinman RM. Latency characteristics of small saccades. Vision Research. 1973;13:2173–2175. doi: 10.1016/0042-6989(73)90195-8. [DOI] [PubMed] [Google Scholar]
- Yarbus AL. Eye Movements and Vision. Plenum Press; New York: 1967. [Google Scholar]
- Yuval-Greenberg S, Tomer O, Nelken I, Deouell LY. Transient induced gamma-band response in EEG as a manifestation of miniature saccades. Neuron. 2008;58:429–441. doi: 10.1016/j.neuron.2008.03.027. [DOI] [PubMed] [Google Scholar]