Table S1.
Physical, chemical, and biologic characteristics of vitamin E and caffeine
| Active compounds | α-tocopherol or vitamin E | Caffeine |
|---|---|---|
| Chemical structure |
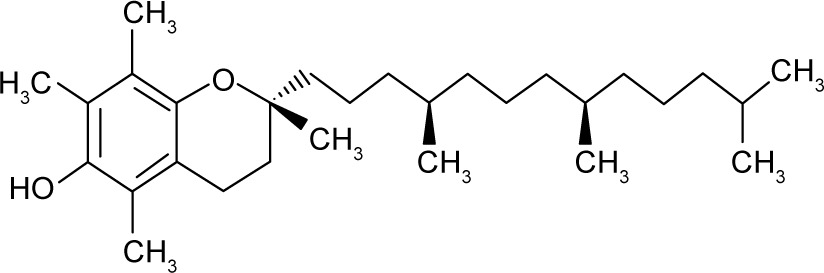
|
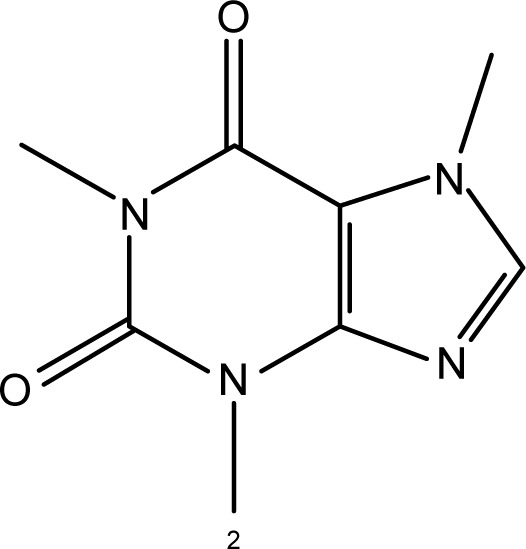
|
| Chemical group | Tocopherols3 | Xanthine4 |
| IUPAC name | (2R)-2,5,7,8-Tetramethyl-2-[(4R,8R)-(4,8,12-trimethyltridecyl)]-6-chromanol5 | 1,3,7-trimethyl-3,7-dihydro-1H--purine-2,6-dione4 |
| Molecular formula | C29H50O25 | C8H10N4O24 |
| Molar mass (g/mol) | 430.715 | 194.194 |
| Log P | ~8.8–10.55 | ~−0.07/−0.554 |
| Water solubility | Insoluble in water, but water-dispersible5 | ~2.16E+004 mg/L (at 25°C)4 |
| Skin distribution | The distribution rate of vitamin E along the skin layers is not similar and Thiele et al6 described an increased quantity of vitamin E in the lower SC. | Although caffeine has an efficient penetration in the skin, the distribution rate of caffeine along the skin layers is not similar. After application of caffeine emulsion, a tape stripping test was made and the SC showed an increased quantity of caffeine comparatively to epidermis and dermis.7 |
| Biological activity/mechanism of action | Antioxidants,8 being important radical scavengers and protecting the cells against lipid peroxidation.9 α-tocopherol is the isoform of vitamin E with the most significant antioxidant activity and with the highest bioavailability due to a strong affinity to α-tocopherol transfer protein and low rate of metabolism.10 | Inhibition of phosphodiesterase, antagonist of adenosine receptors or purinergic P1 receptors, and modulation of calcium handling.2,11,12 |
| Therapeutic action on skin disorders/diseases | Yellow nail syndrome, epidermolysis bullosa, cutaneous ulcers, wound healing,13 and photoprotection.14 Anticancer activity.15 | Psoriasis16 and stimulation of lipolysis in epidermal adipocytes for the treatment of cellulite.17 Caffeine has also been studied for the topical treatment of herpes simplex virus and malignant tumors in mice.12 |
Abbreviations: IUPAC, International Union of Pure and Applied Chemistry; SC, stratum corneum.
