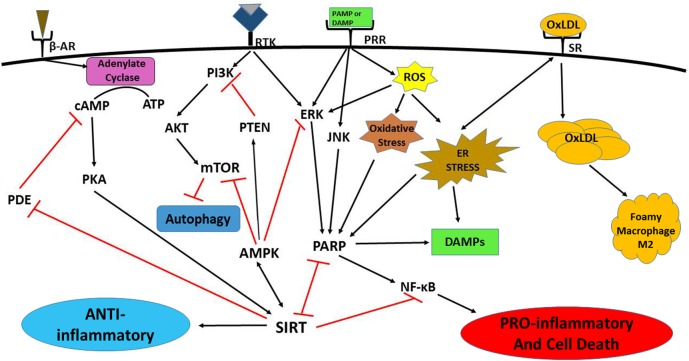
Figure 1.
Signaling pathways of immunometabolism and its dysregulation. Growth receptors (including receptor tyrosine kinases) and pattern recognition receptor (PRRs) induce mechanistic target of rapamycin (mTOR) and poly(ADP-ribose) polymerase (PARP) activation through phosphatidylinositol 3-kinase (PI3K)/serine-threonine protein kinase (AKT) and c-Jun N-terminal kinase (JNK)/extracellular signal-regulated kinase (ERK) signaling, respectively, to stimulate inflammation, largely by upregulation of nuclear factor kappa-light-chain-enhancer of activated B cells (NF-kB). Phosphatidylinositol-3, 4, 5,-trisphosphate 3-phosphatase (PTEN) directly inhibits PI3K activation. Sirtuin (SIRT) and AMP-activated protein kinase (AMPK) cross-activate each other. SIRT dampens inflammation by blocking PARP directly. SIRT signaling also downregulates NF-kB activation. AMPK inhibits PARP through suppression of ERK signaling. AMPK stimulates PTEN, blocks mTOR and induces autophagy. Increased reactive oxygen species (ROS)/oxidative cellular stress induces ERK signaling and PARP activation, and endoplasmic reticulum stress (ERS) increases PARP activation. Both types of stress lead to inflammation, cell damage, and death, and damage-associated molecular pattern (DAMP) molecule release. DAMPs further increase these signaling patterns, resulting in a vicious cycle of progressive inflammation and cell death. The stress responses also lead to increased uptake of oxidized low-density lipoprotein (ox-LDL) in macrophages via scavenger receptors. Increased ox-LDL causes lipid droplet formation that may lead to foam cell development. Foamy macrophages are most often M2 polarized, producing a hypoinflammatory response and increasing susceptibility to Mycobacterium tuberculosis infection. β-Adrenergic and some G-protein–coupled receptors can activate adenylate cyclase that in turn increases protein kinase A (PKA) and SIRT activities. SIRT activation can also inhibit cyclic adenosine monophosphate (cAMP) phosphodiesterase (PDE).