Abstract
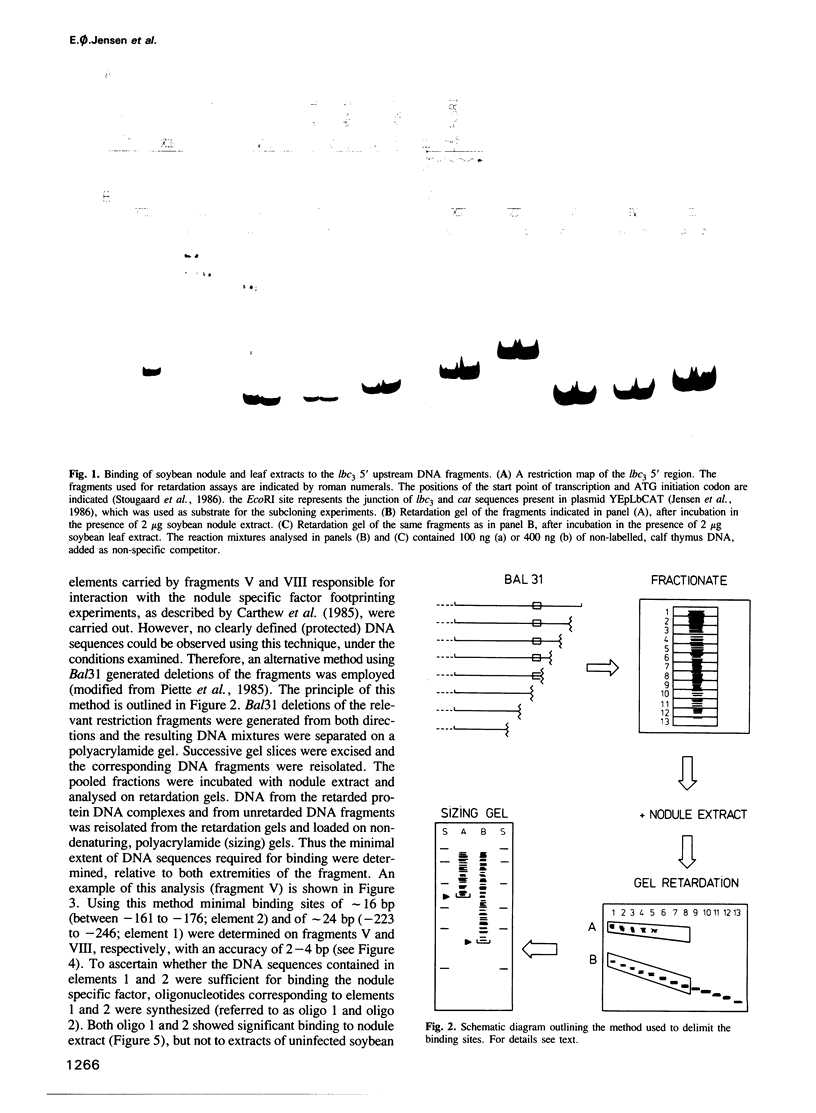
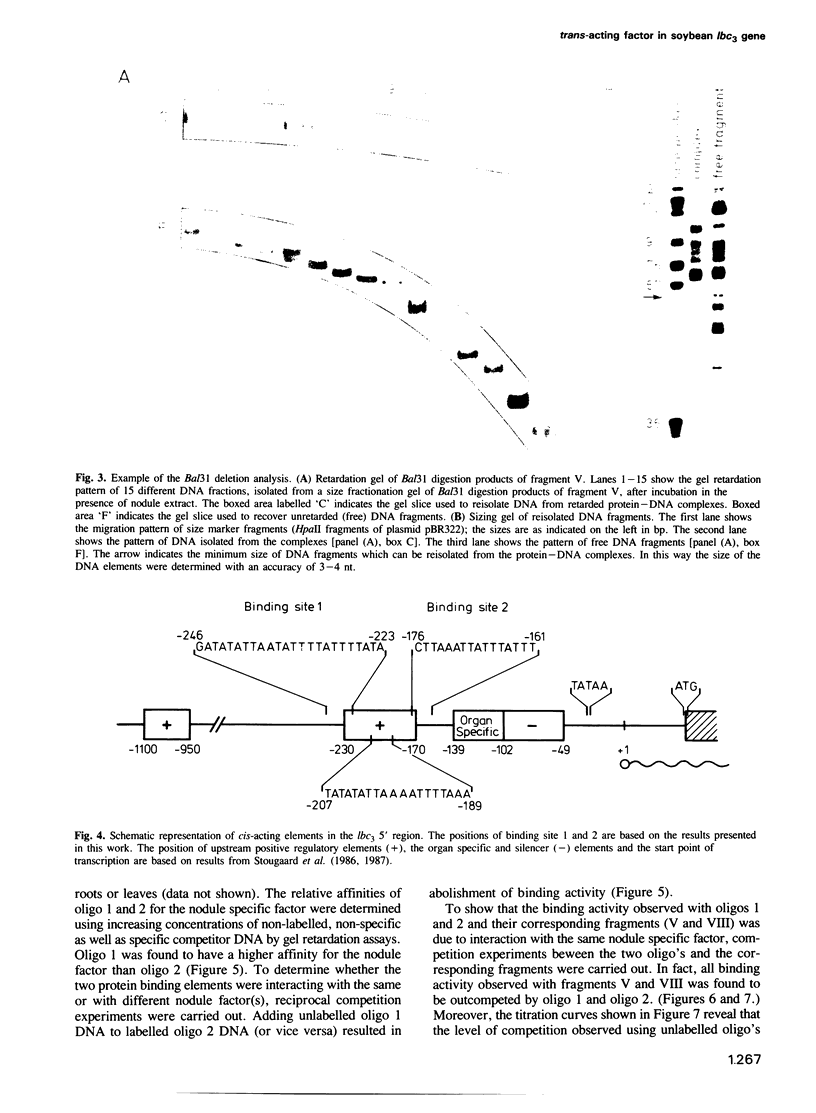
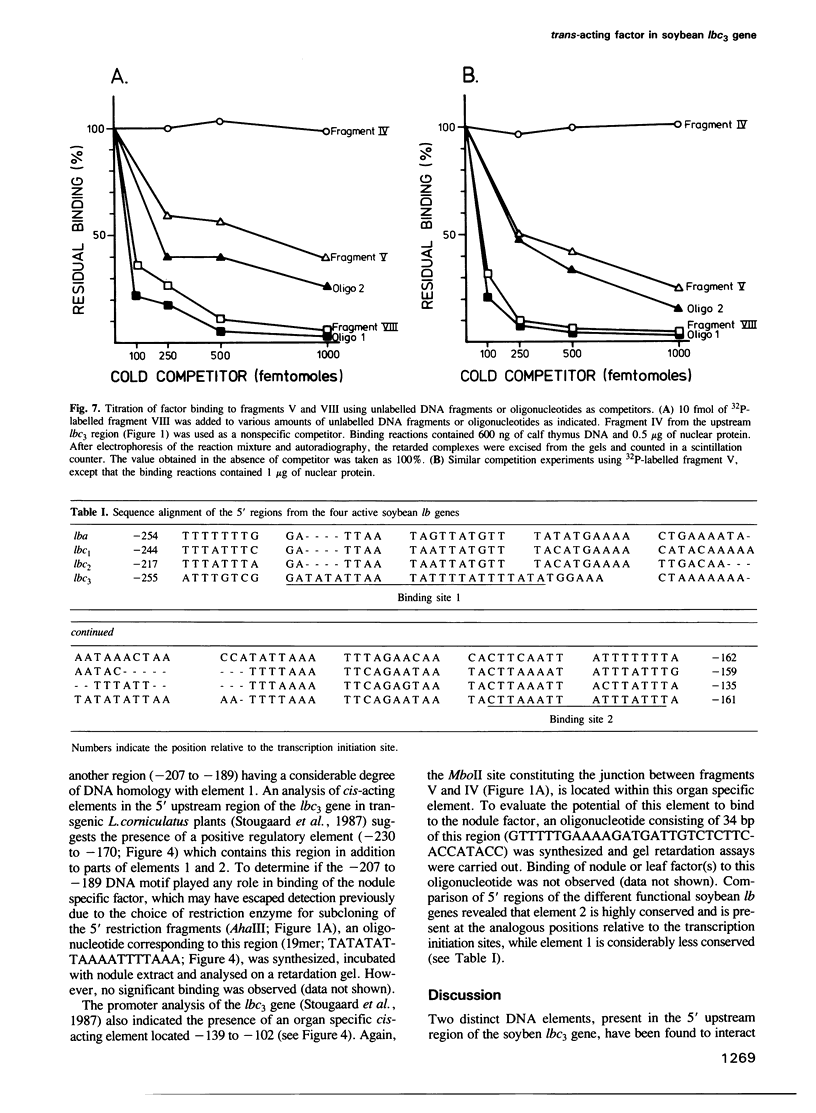
Nuclear extracts from soybean nodules, leaves and roots were used to investigate protein–DNA interactions in the 5' upstream (promoter) region of the soybean leghaemoglobin lbc3 gene. Two distinct regions were identified which strongly bind a nodule specific factor. A Bal31 deletion analysis delimited the DNA elements responsible for the binding of this factor, which map at nucleotides –223 to –246 (element 1) and –161 to –176 (element 2), relative to the start point of transcription. Competition experiments strongly suggest that both elements bind to the same nodule specific factor, but with different affinities. Elements 1 and 2 share a common motif, although their AT-rich DNA sequences differ. Element 2 is highly conserved at an analogous position in other soybean lb gene 5' upstream regions.
Keywords: leghaemoglobin, trans-acting factor, organ specificity, promoter elements, nodules
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Carthew R. W., Chodosh L. A., Sharp P. A. An RNA polymerase II transcription factor binds to an upstream element in the adenovirus major late promoter. Cell. 1985 Dec;43(2 Pt 1):439–448. doi: 10.1016/0092-8674(85)90174-6. [DOI] [PubMed] [Google Scholar]
- Dignam J. D., Lebovitz R. M., Roeder R. G. Accurate transcription initiation by RNA polymerase II in a soluble extract from isolated mammalian nuclei. Nucleic Acids Res. 1983 Mar 11;11(5):1475–1489. doi: 10.1093/nar/11.5.1475. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fried M., Crothers D. M. Equilibria and kinetics of lac repressor-operator interactions by polyacrylamide gel electrophoresis. Nucleic Acids Res. 1981 Dec 11;9(23):6505–6525. doi: 10.1093/nar/9.23.6505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garner M. M., Revzin A. A gel electrophoresis method for quantifying the binding of proteins to specific DNA regions: application to components of the Escherichia coli lactose operon regulatory system. Nucleic Acids Res. 1981 Jul 10;9(13):3047–3060. doi: 10.1093/nar/9.13.3047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hyldig-Nielsen J. J., Jensen E. O., Paludan K., Wiborg O., Garrett R., Jørgensen P., Marcker K. A. The primary structures of two leghemoglobin genes from soybean. Nucleic Acids Res. 1982 Jan 22;10(2):689–701. doi: 10.1093/nar/10.2.689. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jensen E. O., Marcker K. A., Villadsen I. S. Heme regulates the expression in Saccharomyces cerevisiae of chimaeric genes containing 5'-flanking soybean leghemoglobin sequences. EMBO J. 1986 May;5(5):843–847. doi: 10.1002/j.1460-2075.1986.tb04293.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jofuku K. D., Okamuro J. K., Goldberg R. B. Interaction of an embryo DNA binding protein with a soybean lectin gene upstream region. Nature. 1987 Aug 20;328(6132):734–737. doi: 10.1038/328734a0. [DOI] [PubMed] [Google Scholar]
- Legocki R. P., Verma D. P. Identification of "nodule-specific" host proteins (nodoulins) involved in the development of rhizobium-legume symbiosis. Cell. 1980 May;20(1):153–163. doi: 10.1016/0092-8674(80)90243-3. [DOI] [PubMed] [Google Scholar]
- Marcker A., Lund M., Jensen E. Ø, Marcker K. A. Transcription of the soybean leghemoglobin genes during nodule development. EMBO J. 1984 Aug;3(8):1691–1695. doi: 10.1002/j.1460-2075.1984.tb02033.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Piette J., Kryszke M. H., Yaniv M. Specific interaction of cellular factors with the B enhancer of polyoma virus. EMBO J. 1985 Oct;4(10):2675–2685. doi: 10.1002/j.1460-2075.1985.tb03987.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spector T. Refinement of the coomassie blue method of protein quantitation. A simple and linear spectrophotometric assay for less than or equal to 0.5 to 50 microgram of protein. Anal Biochem. 1978 May;86(1):142–146. doi: 10.1016/0003-2697(78)90327-5. [DOI] [PubMed] [Google Scholar]
- St Schell J. Transgenic plants as tools to study the molecular organization of plant genes. Science. 1987 Sep 4;237(4819):1176–1183. doi: 10.1126/science.237.4819.1176. [DOI] [PubMed] [Google Scholar]
- Stougaard J., Sandal N. N., Grøn A., Kühle A., Marcker K. A. 5' Analysis of the soybean leghaemoglobin lbc(3) gene: regulatory elements required for promoter activity and organ specificity. EMBO J. 1987 Dec 1;6(12):3565–3569. doi: 10.1002/j.1460-2075.1987.tb02686.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wiborg O., Hyldig-Nielsen J. J., Jensen E. O., Paludan K., Marcker K. A. The nucleotide sequences of two leghemoglobin genes from soybean. Nucleic Acids Res. 1982 Jun 11;10(11):3487–3494. doi: 10.1093/nar/10.11.3487. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yanisch-Perron C., Vieira J., Messing J. Improved M13 phage cloning vectors and host strains: nucleotide sequences of the M13mp18 and pUC19 vectors. Gene. 1985;33(1):103–119. doi: 10.1016/0378-1119(85)90120-9. [DOI] [PubMed] [Google Scholar]







