Figure 2.
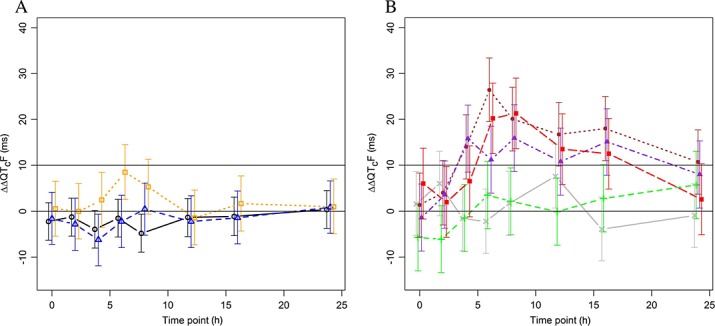
Placebo-adjusted, change from time matched baseline QTcF (ΔΔQTcF; mean ± 90%CI). Results are based on an analysis of variance model for repeated measurements with terms for dose level, time point and their interaction. A) Period 1 (OZ439 only). Number of subjects: 24, eight and seven subjects in the 100 mg, 300 mg and 800 mg groups, respectively.  800 mg OZ439,
800 mg OZ439,  300 mg OZ439,
300 mg OZ439,  100 mg OZ439. B) Period 2 (OZ439 + PQP). Number of subjects: eight subjects in 100 mg OZ439 + 160 mg PQP and 100 mg OZ439 + 1440 mg PQP, seven subjects in 100 mg OZ439 + 480 mg PQP and 300 mg OZ439 + 1440 mg PQP and six subjects in 800 mg OZ439 + 1440 mg PQP groups.
100 mg OZ439. B) Period 2 (OZ439 + PQP). Number of subjects: eight subjects in 100 mg OZ439 + 160 mg PQP and 100 mg OZ439 + 1440 mg PQP, seven subjects in 100 mg OZ439 + 480 mg PQP and 300 mg OZ439 + 1440 mg PQP and six subjects in 800 mg OZ439 + 1440 mg PQP groups.  800 mg OZ439 + 1440 mg PQP,
800 mg OZ439 + 1440 mg PQP,  300 mg OZ439 +1440 mg PQP,
300 mg OZ439 +1440 mg PQP,  100 mg OZ439 +1440 mg PQP,
100 mg OZ439 +1440 mg PQP,  100 mg OZ439 + 480 mg PQP,
100 mg OZ439 + 480 mg PQP,  100 mg OZ439 + 160 mg PQP
100 mg OZ439 + 160 mg PQP
