Abstract
Purpose
Mefenamic acid (MEF) as a non-steroidal anti-inflammatory drug is used as a medication for relieving of pain and inflammation. Radiation-induced inflammation process is involved in DNA damage and cell death. In this study, the radioprotective effect of MEF was investigated against genotoxicity induced by ionizing radiation in human blood lymphocytes.
Materials and Methods
Peripheral blood samples were collected from human volunteers and incubated with MEF at different concentrations (5, 10, 50, or 100 µM) for two hours. The whole blood was exposed to ionizing radiation at a dose 1.5 Gy. Lymphocytes were cultured with mitogenic stimulation to determine the micronuclei in cytokinesis blocked binucleated lymphocyte.
Results
A significant decreasing in the frequency of micronuclei was observed in human lymphocytes irradiated with MEF as compared to irradiated lymphocytes without MEF. The maximum decreasing in frequency of micronuclei was observed at 100 µM of MEF (38% decrease), providing maximal protection against ionizing radiation.
Conclusion
The radioprotective effect of MEF is probably related to anti-inflammatory property of MEF on human lymphocytes.
Keywords: Mefenamic acid, DNA damage, Radiation-protective agents, Micronucleus test
Introduction
Exposure to ionizing radiation (IR) is associated with increased DNA damage and leads to stress response and inflammation, even carcinogenesis and cell death. IR produces free radicals and other reactive substance and induces reactive oxygen species (ROS) which is main mechanism involved in DNA damage. ROS interacts with critical macromolecules such as DNA and oxidizes DNA [1,2]. There is a cross talk between ROS mediated inflammation and DNA damage. IR modulates several signaling pathways resulting in transcription factor activation. In response to DNA damage, IR activates inflammatory process through expression of proinflammatory cytokines such as interleukin-1 and 6 (IL-1 and IL-6) and mediators of cytokines such as nuclear factor-kappa B and tumor necrosis factor-alpha (TNF-α) [3,4]. Theses cytokines has an anti-apoptotic effect and promotes cell proliferation [5,6]. Several studies demonstrated that antioxidants scavenged free radicals in the irradiated cells and resulted in decreased inflammation process [7,8,9].
Mefenamic acid (MEF) is a non-steroidal anti-inflammatory drug (NSAID) relieving dental and menstrual pains. It is typically prescribed for oral administration. MEF provided significant protection against the elevation of lipid peroxidation, protein oxidation, levels of TNF-α and IL-1β and finally decreased inflammation in the brain of rat [10,11]. In vitro , MEF could also reduce ROS production, lipid peroxidation, and protein oxidation [12,13]. IR induced genotoxicity is wellknown to increase the risk of developing malignancy and to be linked to IR induced inflammatory processes suggesting that an anti-inflammatory drug may protect IR induced DNA damages. The effect of anti-inflammatory MEF has not been clearly revealed yet on irradiated normal human cells. Thus we investigated the IR induced genotoxicity on human normal lymphocytes priming with or without MEF.
Materials and Methods
1. Blood treatment
After obtaining permission from research and ethical committees of the Mazandaran University of Medical Sciences, this study was performed. Healthy, non-smoking three male volunteers, aged from 20 to 28 years were enrolled in this study. Twelve milliliters of whole blood were collected in heparinized tubes and divided in centrifuge tubes at 0.9 mL each. Blood samples were primed with 100 µL solution of MEF (Alhavi Pharmaceutical Company, Tehran, Iran) at the concentrations of 5, 10, 50, or 100 µM. These samples were incubated for two hours at 37℃. MEF was dissolved in dimethyl sulfoxide (DMSO; Merck Company, Darmstadt, Germany) and diluted in RPMI cultural medium (Gibco, Carlsbad, CA, USA). DMSO concentration was same in control and MEF solutions (0.2%).
2. Ionizing radiation and micronucleus test
Whole blood samples in tubes were irradiated with 6 MV X-ray beam produced by a linear accelerator (Primus; Siemens, Erlangen, Germany) at a dose of 1.5 Gy with a dose rate of 1.9 Gy/min. Samples from three volunteers were allocated to the control (non-irradiated samples). After irradiation, subsequently, 0.5 mL of each sample (control and irradiated samples in duplicate) was added to 4.4 mL of RPMI 1640 culture medium (Gibco), which contained a mixture of 10% fetal calf serum, 100 µL phytohemagglutinin (Gibco). All cultures were incubated at 37℃ in a humidified atmosphere of 5% CO2 and 95% air. Cytochalasin B (final concentration, 6 µL/mL; Sigma-Aldrich, St. Louis, MO, USA) was added after 44 hours of culture. Following 72 hours of incubation, the cells were collected by centrifugation, re-suspended in cold 0.75 M potassium chloride. Cells were immediately fixed in a fixative solution as made of methanol: acetic acid (6:1) two times. The fixed cells were dropped onto clean microscopic slides, air-dried and stained with 10% Giemsa (Merck) solution. All slides were evaluated at 100× magnification in order to determine the frequency of micronuclei in the cytokinesisblocked binucleated cells with a well-preserved cytoplasm [14]. For each treated group from each volunteer, a total of 1,000 binucleate cells (in the duplicate cultures) were examined to record the frequency of micronuclei-containing cells. A total of 3,000 binucleated lymphocytes were counted in each treated group from three volunteers, and totally 30,000 binucleated lymphocytes were counted for ten treated groups in this study.
3. Statistical analysis
For each volunteer, the frequency of IR-induced micronuclei was determined at each concentration of MEF. The data were analyzed with Student t-test. A probability value of 0.05 was accepted to denote significance.
Results
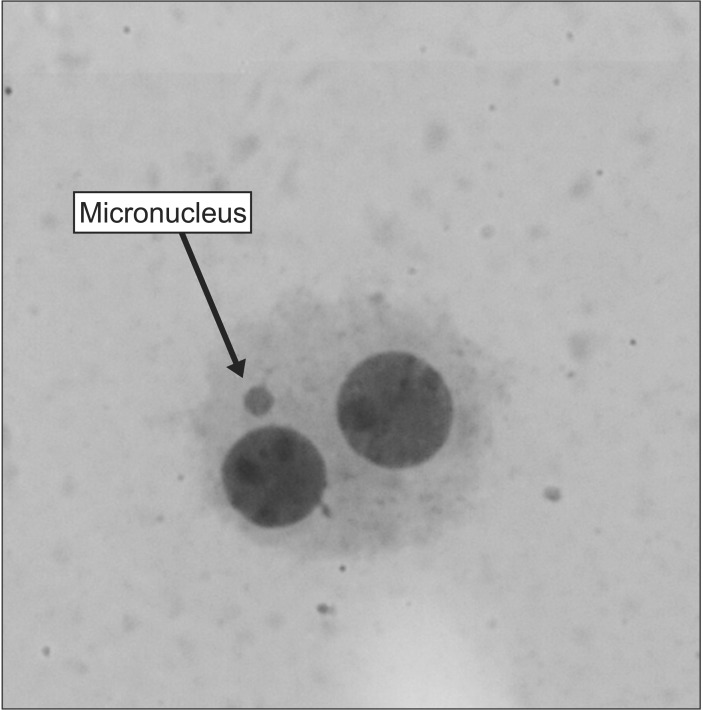
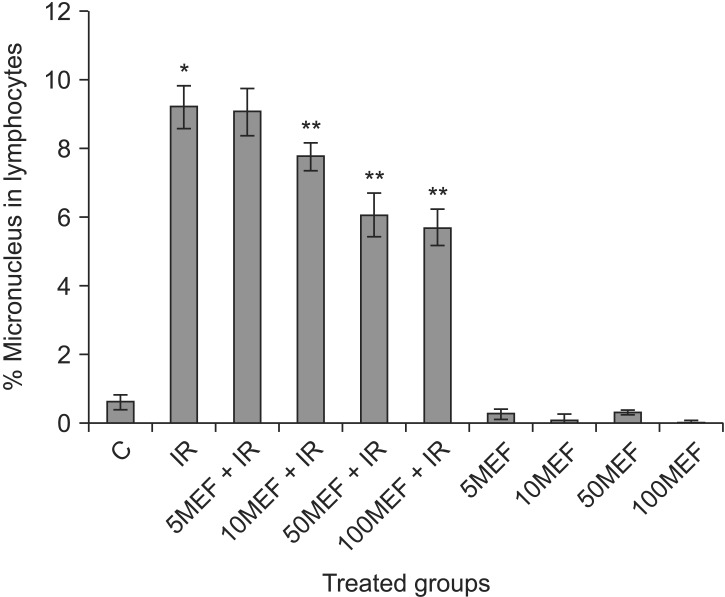
A typical binucleated lymphocyte with micronucleus is shown in Fig. 1. The mean frequency of micronuclei in irradiated three volunteers was 9.23 ± 0.63, while that in non-irradiated control samples was 0.63 ± 0.21. It showed a statistically significant (p < 0.001) increase (14-fold rise) in the frequency of micronuclei in irradiated samples at dose of 1.5 Gy (Table 1). In irradiated samples with MEF, the frequency of micronuclei at the concentrations of 5, 10, 50, or 100 µM were 9.10 ± 0.71, 7.80 ± 0.40, 6.10 ± 0.62, and 5.73 ± 0.51, respectively (Fig. 2 and Table 1). The data demonstrate that irradiated samples with MEF at concentrations of 10, 50, and 100 µM exhibited a significant decrease in the frequency of micronuclei as compared to irradiated samples without MEF. Total micronuclei frequencies were reduced by 1.4%, 15%, 34%, and 38% in irradiated samples with MEF at concentrations of 5, 10, 50, or 100 µM, respectively, compared to irradiated samples without MEF (Table 1). Among irradiated samples with MEF, a statistically significant difference in micronuclei was observed between doses 10 and 50 µM of MEF. It is interesting, the frequency of micronuclei in non-irradiated sample with MEF at concentration of 10 and 100 µM was significantly lower than control group (p < 0.05).
Fig. 1. A typical binucleated lymphocyte with micronucleus in our experiments.
Table 1. The frequency of micronuclei induced in vitro by 1.5 Gy X-ray radiation (IR) in cultured blood lymphocytes at different doses of mefenamic acida).
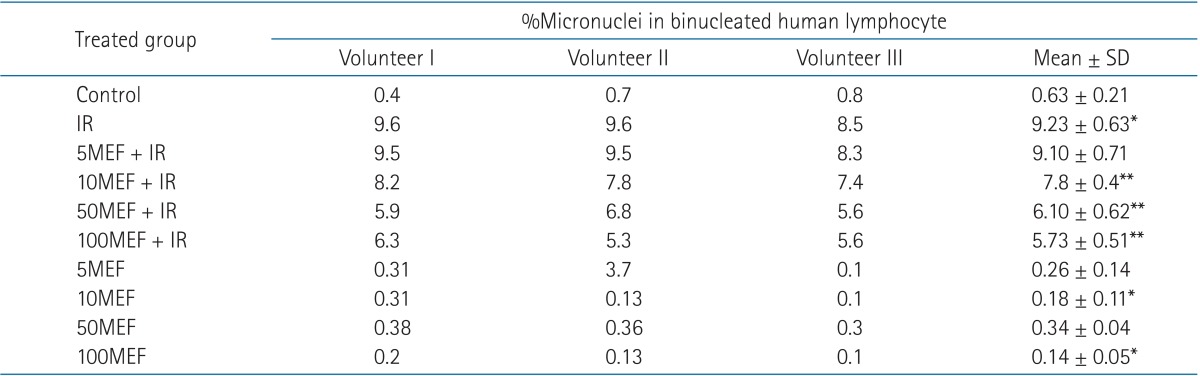
SD, standard deviation; IR, ionizing radiation; 5MEF, 5 µM mefenamic acid; 10MEF, 10 µM mefenamic acid; 50MEF, 50 µM mefenamic acid; 100MEF, 100 µM mefenamic acid.
a)1,000 binucleated lymphocyte were examined in each sample, and 3,000 binucleated lymphocyte from three volunteers in each group.
*p < 0.05 compared to control, **p < 0.05 compared to IR.
Fig. 2. In vitro protection by mefenamic acid (MEF) at different concentrations (5, 10, 50, and 100 µM) against ionizing radiation (IR) induced genetic damage in cultured whole blood lymphocytes. The data represent average ± standard deviation from three volunteers. C, control; 5MEF, 5 µM mefenamic acid; 10MEF, 10 µM mefenamic acid; 50MEF, 50 µM mefenamic acid; 100MEF, 100 µM mefenamic acid. *p < 0.05 compared to control, **p < 0.05 compared to IR.
Discussion and Conclusion
In this study, we showed priming of human lymphocytes with MEF markedly reduced genotoxicity induced by IR. The frequency of micronuclei was reduced with pretreatment of MEF. MEF is widely used as an anti-inflammatory drug for relieving of pain and inflammation [15]. MEF belongs to NSAID, and the mechanism of action is partially attributed to capability of inhibiting the cyclooxygenase-2 (COX-2) enzyme, an essential component of the inflammatory process [16,17]. MEF has an inhibitory effect on the proliferation of cancer cells such as liver and colon cancer cells. Different mechanisms are proposed for anti-cancer effect of MEF to be caspase-3 pathway and inhibiting of the synthesis of hyaluronic acid and cyclooxygenase [18,19,20,21]. COX-2 is frequently over expressed in various cancer tissues, thus inhibition of COX-2 by NSAID is suggested as a strategy for cancer treatment and prevention [20,21,22,23,24]. While inflammatory environment is linked to DNA damage and death in normal cells, proinflammatory cytokines are involved in cancer cell growth [25,26]. Inflammatory cytokines secreted by tumor activate the necessary pathways to facilitate cancer cells proliferation [27], It is recommended that blockade of inflammation process by NSAIDs inhibit cancer progression. In the normal cells, exposure to IR initiates secretion of inflammatory cytokines, which is involved in DNA damage [28,29]. Mukherjee et al. [30] showed that NSAIDs could reduce radiation-induced chromosomal instability in vivo. In this study we showed that MEF mitigated IR induced DNA damage in human lymphocytes. It is well documented that free radicals and inflammation are main triggers for IRinduced DNA damage. Normal cell is going to dysfunction in inflammatory process. It is cleared that anti-inflammatory effect of MEF by inhibiting of COX-2 and decreasing secretion of cytokines is main proposed mechanisms for radioprotective effect of MEF. Whereas MEF has anti-cancer effect, it likely protects, in vitro , normal cell against IR induced DNA damage. This result provides a new indication of MEF for protection of normal cells during radiation therapy in treatment of cancer patients. Future studies are needed to find the exact mechanism such as detailed pathways for the radioprotective effect of MEF.
Acknowledgments
This study was supported by a grant from Mazandaran University of Medical Sciences, Sari, Iran. This research was the subject of a PharmD thesis of Reyhaneh Nobakht as a PharmD student of Mazandaran University of Medical Sciences.
Footnotes
Conflict of Interest: No potential conflict of interest relevant to this article was reported.
References
- 1.Cadet J, Wagner JR. Oxidatively generated base damage to cellular DNA by hydroxyl radical and one-electron oxidants: similarities and differences. Arch Biochem Biophys. 2014;557:47–54. doi: 10.1016/j.abb.2014.05.001. [DOI] [PubMed] [Google Scholar]
- 2.Hosseinimehr SJ. Flavonoids and genomic instability induced by ionizing radiation. Drug Discov Today. 2010;15:907–918. doi: 10.1016/j.drudis.2010.09.005. [DOI] [PubMed] [Google Scholar]
- 3.Ozyurt H, Cevik O, Ozgen Z, et al. Quercetin protects radiationinduced DNA damage and apoptosis in kidney and bladder tissues of rats. Free Radic Res. 2014;48:1247–1255. doi: 10.3109/10715762.2014.945925. [DOI] [PubMed] [Google Scholar]
- 4.Yang HJ, Youn H, Seong KM, et al. Psoralidin, a dual inhibitor of COX-2 and 5-LOX, regulates ionizing radiation (IR)-induced pulmonary inflammation. Biochem Pharmacol. 2011;82:524–534. doi: 10.1016/j.bcp.2011.05.027. [DOI] [PubMed] [Google Scholar]
- 5.Chishti AA, Baumstark-Khan C, Hellweg CE, Reitz G. Imaging of nuclear factor kappaB activation induced by ionizing radiation in human embryonic kidney (HEK) cells. Radiat Environ Biophys. 2014;53:599–610. doi: 10.1007/s00411-014-0541-x. [DOI] [PubMed] [Google Scholar]
- 6.Saltman B, Kraus DH, Szeto H, et al. In vivo and in vitro models of ionizing radiation to the vocal folds. Head Neck. 2010;32:572–577. doi: 10.1002/hed.21212. [DOI] [PubMed] [Google Scholar]
- 7.Das U, Manna K, Sinha M, et al. Role of ferulic acid in the amelioration of ionizing radiation induced inflammation: a murine model. PLoS One. 2014;9:e97599. doi: 10.1371/journal.pone.0097599. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Manna K, Das U, Das D, et al. Naringin inhibits gamma radiation-induced oxidative DNA damage and inflammation, by modulating p53 and NF-kappaB signaling pathways in murine splenocytes. Free Radic Res. 2015;49:422–439. doi: 10.3109/10715762.2015.1016018. [DOI] [PubMed] [Google Scholar]
- 9.Rithidech KN, Tungjai M, Reungpatthanaphong P, Honikel L, Simon SR. Attenuation of oxidative damage and inflammatory responses by apigenin given to mice after irradiation. Mutat Res. 2012;749:29–38. doi: 10.1016/j.mrgentox.2012.08.001. [DOI] [PubMed] [Google Scholar]
- 10.Armagan G, Turunc E, Kanit L, Yalcin A. Neuroprotection by mefenamic acid against D-serine: involvement of oxidative stress, inflammation and apoptosis. Free Radic Res. 2012;46:726–739. doi: 10.3109/10715762.2012.669836. [DOI] [PubMed] [Google Scholar]
- 11.Asanuma M, Nishibayashi-Asanuma S, Miyazaki I, Kohno M, Ogawa N. Neuroprotective effects of non-steroidal antiinflammatory drugs by direct scavenging of nitric oxide radicals. J Neurochem. 2001;76:1895–1904. doi: 10.1046/j.1471-4159.2001.00205.x. [DOI] [PubMed] [Google Scholar]
- 12.Armagan G, Kanit L, Yalcin A. Effects of non-steroidal antiinflammatory drugs on D-serine-induced oxidative stress in vitro. Drug Chem Toxicol. 2012;35:393–398. doi: 10.3109/01480545.2011.633086. [DOI] [PubMed] [Google Scholar]
- 13.Corda S, Laplace C, Vicaut E, Duranteau J. Rapid reactive oxygen species production by mitochondria in endothelial cells exposed to tumor necrosis factor-alpha is mediated by ceramide. Am J Respir Cell Mol Biol. 2001;24:762–768. doi: 10.1165/ajrcmb.24.6.4228. [DOI] [PubMed] [Google Scholar]
- 14.Fenech M. The in vitro micronucleus technique. Mutat Res. 2000;455:81–95. doi: 10.1016/s0027-5107(00)00065-8. [DOI] [PubMed] [Google Scholar]
- 15.Moll R, Derry S, Moore RA, McQuay HJ. Single dose oral mefenamic acid for acute postoperative pain in adults. Cochrane Database Syst Rev. 2011;(3):CD007553. doi: 10.1002/14651858.CD007553.pub2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Xu XC. COX-2 inhibitors in cancer treatment and prevention, a recent development. Anticancer Drugs. 2002;13:127–137. doi: 10.1097/00001813-200202000-00003. [DOI] [PubMed] [Google Scholar]
- 17.Prusakiewicz JJ, Duggan KC, Rouzer CA, Marnett LJ. Differential sensitivity and mechanism of inhibition of COX-2 oxygenation of arachidonic acid and 2-arachidonoylglycerol by ibuprofen and mefenamic acid. Biochemistry. 2009;48:7353–7355. doi: 10.1021/bi900999z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.August EM, Nguyen T, Malinowski NM, Cysyk RL. Non-steroidal anti-inflammatory drugs and tumor progression: inhibition of fibroblast hyaluronic acid production by indomethacin and mefenamic acid. Cancer Lett. 1994;82:49–54. doi: 10.1016/0304-3835(94)90145-7. [DOI] [PubMed] [Google Scholar]
- 19.Teicher BA, Korbut TT, Menon K, Holden SA, Ara G. Cyclooxy-genase and lipoxygenase inhibitors as modulators of cancer therapies. Cancer Chemother Pharmacol. 1994;33:515–522. doi: 10.1007/BF00686511. [DOI] [PubMed] [Google Scholar]
- 20.Woo DH, Han IS, Jung G. Mefenamic acid-induced apoptosis in human liver cancer cell-lines through caspase-3 pathway. Life Sci. 2004;75:2439–2449. doi: 10.1016/j.lfs.2004.04.042. [DOI] [PubMed] [Google Scholar]
- 21.Hixson LJ, Alberts DS, Krutzsch M, et al. Antiproliferative effect of nonsteroidal antiinflammatory drugs against human colon cancer cells. Cancer Epidemiol Biomarkers Prev. 1994;3:433–438. [PubMed] [Google Scholar]
- 22.Gurpinar E, Grizzle WE, Piazza GA. COX-Independent Mechanisms of Cancer Chemoprevention by Anti-Inflammatory Drugs. Front Oncol. 2013;3:181. doi: 10.3389/fonc.2013.00181. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Ganesh R, Marks DJ, Sales K, Winslet MC, Seifalian AM. Cyclooxygenase/lipoxygenase shunting lowers the anti-cancer effect of cyclooxygenase-2 inhibition in colorectal cancer cells. World J Surg Oncol. 2012;10:200. doi: 10.1186/1477-7819-10-200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.de Groot DJ, de Vries EG, Groen HJ, de Jong S. Non-steroidal anti-inflammatory drugs to potentiate chemotherapy effects: from lab to clinic. Crit Rev Oncol Hematol. 2007;61:52–69. doi: 10.1016/j.critrevonc.2006.07.001. [DOI] [PubMed] [Google Scholar]
- 25.Licandro G, Ling Khor H, Beretta O, et al. The NLRP3 inflammasome affects DNA damage responses after oxidative and genotoxic stress in dendritic cells. Eur J Immunol. 2013;43:2126–2137. doi: 10.1002/eji.201242918. [DOI] [PubMed] [Google Scholar]
- 26.Ruan D, So SP. Prostaglandin E2 produced by inducible COX-2 and mPGES-1 promoting cancer cell proliferation in vitro and in vivo. Life Sci. 2014;116:43–50. doi: 10.1016/j.lfs.2014.07.042. [DOI] [PubMed] [Google Scholar]
- 27.Shigdar S, Li Y, Bhattacharya S, et al. Inflammation and cancer stem cells. Cancer Lett. 2014;345:271–278. doi: 10.1016/j.canlet.2013.07.031. [DOI] [PubMed] [Google Scholar]
- 28.Mukherjee D, Coates PJ, Lorimore SA, Wright EG. Responses to ionizing radiation mediated by inflammatory mechanisms. J Pathol. 2014;232:289–299. doi: 10.1002/path.4299. [DOI] [PubMed] [Google Scholar]
- 29.Guerra L, Guidi R, Frisan T. Do bacterial genotoxins contribute to chronic inflammation, genomic instability and tumor progression. FEBS J. 2011;278:4577–4588. doi: 10.1111/j.1742-4658.2011.08125.x. [DOI] [PubMed] [Google Scholar]
- 30.Mukherjee D, Coates PJ, Lorimore SA, Wright EG. The in vivo expression of radiation-induced chromosomal instability has an inflammatory mechanism. Radiat Res. 2012;177:18–24. doi: 10.1667/rr2793.1. [DOI] [PubMed] [Google Scholar]