Figure 3.
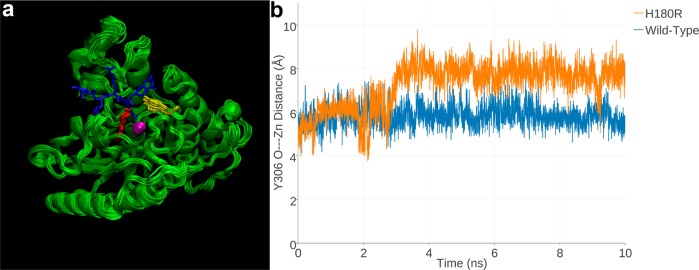
(a) Superimposed 1 ns snapshots from the 10 ns MD simulation of Zn2+-bound H180R HDAC8, with a tetrapeptide assay substrate (blue) superimposed for reference (from the structure of the H143A HDAC8–substrate complex, PDB accession code 3EWF). Zn2+ is a magenta sphere, R180 is red, and Y306 is yellow. (b) Y306 fluctuates ∼2 Å away from the “in” conformation required for catalysis in H180R HDAC8 relative to its fluctuations in the wild-type enzyme over the course of the 10 ns MD simulation. The fluctuations of Y306 in the MD simulation of Zn2+-free H180R HDAC8 are just slightly less (Figure S2, Supporting Information).
