Figure 2.
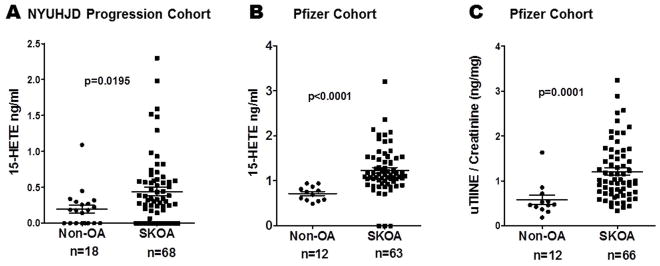
Baseline plasma 15-hydroxyeicosatetraenoic acid (15-HETE) is elevated in NYUHJD progression (A) and Pfizer validation cohorts (B). C) Baseline urinary type II collagen neoepitope (uTIINE) is elevated in Pfizer validation cohort [LC-MS/MS (Liquid Chromatography-Tandem Mass Spectrometry)]. The solid horizontal bar in each group represents the mean value. Non-parametric, Mann-Whitney test was performed to calculate statistical significance using GraphPad Prism 4.0. The p values for cases versus controls are shown for each biomarker.
Mean (±SD) 15-HETE (ng/ml) levels in NYUHJD progression SKOA cases were 0.44±0.48 (0.0–2.3) and in controls and 0.22±0.26 (0.0–1.1), respectively. Mean (±SD) 15-HETE (ng/ml) levels in Pfizer SKOA cases were 1.3±0.45 (0.71–3.2), and in controls 0.72±0.16 (0.49–0.94), respectively.
