Fig. 10.
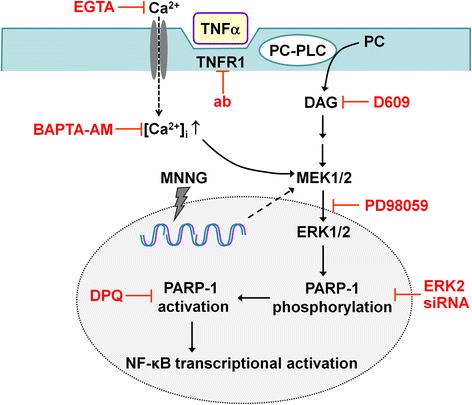
The schematic summary of TNFα-induced PARP-1 activation pathway in glial cells. PARP-1 activation induced by TNFα does not involve DNA damage, which is a classical inducer of PARP-1 activation. A MNNG-induced DNA damage activation pathway differs from a TNFα-induced one, but they both share the requirement of ERK2-mediated PARP-1 phosphorylation. TNFα binding to its receptor TNFR1 induces calcium influx, and activates phosphotidyl choline-specific phospholipase (PC-PLC) leading to phosphotidyl choline cleavage to diacyl glycerol (DAG). This process leads to activation of mitogen-activated protein kinase pathway (MEK1/2-ERK1/2) and ERK2-mediated phosphorylation of PARP-1, which allow PARP-1 activation. Enzymatically active PARP-1 co-activates NF-κB transcriptional activation promoting pro-inflammatory secretion profile. TNFα-induced PARP-1 activation was prevented by calcium chelators (EGTA, BAPTA-AM), TNFR1-neutralizing antibody (ab), inhibitors of PC-PLC-DAG (D609), MEK1/2-ERK1/2 (PD98059), PARP (DPQ) and ERK2 siRNA, while MNNG-induced PARP activation was affected only by inhibitors of ERK2 and PARP. TNFα-induced NF-κB transcription activation was prevented by inhibitors of PC-PLC-DAG, ERK1/2 and PARP
