Highlights
-
•
Novel organic selenium compounds presented low toxicity over C. elegans survival.
-
•
Defecation cycles were decreased by Novel organic selenium compounds, except C1.
-
•
Novel organic selenium compounds, except C1 decreased AChE enzyme activity.
-
•
All compounds tested prevented from juglone stress.
-
•
C2 selenium compound decreased GST-4::GFP fluorescence under juglone stress.
Keywords: Caenorhabditis elegans, Diphenyl diselenide, Novel organic selenium compounds, GST-4::GFP, AChE, Juglone
Abstract
Organic selenium compounds possess numerous biological properties, including antioxidant activity. Yet, the high toxicity of some of them, such as diphenyl diselenide (DPDS), is a limiting factor in their current usage. Accordingly, we tested four novel organic selenium compounds in the non-parasite nematode Caenorhabditis elegans and compared their efficacy to DPDS. The novel organic selenium compounds are β-selenoamines 1-phenyl-3-(p-tolylselanyl)propan-2-amine (C1) and 1-(2-methoxyphenylselanyl)-3-phenylpropan-2-amine (C2) and analogs of DPDS 1,2-bis(2-methoxyphenyl)diselenide (C3) and 1,2-bisp-tolyldiselenide (C4). Synchronized worms at the L4 larval stage were exposed for one hour in M9 buffer to these compounds. Oxidative stress conditions were induced by juglone (200 μM) and heat shock (35 °C). Moreover, we evaluated C. elegans behavior, GST-4::GFP (glutathione S-transferase) expression and the activity of acetylcholinesterase (AChE). All tested compounds efficiently restored viability in juglone stressed worms. However, DPDS, C2, C3 and C4 significantly decreased the defecation cycle time. Juglone-induced GST-4::GFP expression was not attenuated in worms pretreated with the novel compounds, except with C2. Finally, AChE activity was reduced by DPDS, C2, C3 and C4. To our knowledge, this is study firstly showed the effects of C1, C2, C3 and C4 selenium-derived compounds in C. elegans. Low toxic effects were noted, except for reduction in the defecation cycle, which is likely associated with AChE inhibition. The juglone-induced stress (reduced viability) was fully reversed by compounds to control animal levels. C2 was also efficient in reducing the juglone-induced GST-4::GFP expression, suggesting the latter may mediate the stress induced by this compound. Future studies could be profitably directed at addressing additional molecular mechanisms that mediate the protective effects of these novel organic selenium compounds.
1. Introduction
Organic selenium compounds have been recognized for their immunosuppressive, anti-inflammatory, anti-cancer, anti-depressive, as well as neuro, cardio and hepatoprotective activities in a myriad of diseases associated with increased oxidative stress conditions [1]. Two of the main studied selenium-containing compounds, ebselen and diphenyl diselenide (DPDS), possess strong antioxidant activity, which has been associated with their ability to mimic glutathione peroxidase-like activity (GPx), as well as also by acting as substrates of thioredoxin reductase (TrxR) [1], [2]. Nevertheless, the toxicity of these molecules has limited their usage for pharmacological purposes in vertebrate animals [3]. Their toxicity is primarily characterized by enzymatic inhibition of acetylcholinesterase (AChE) and δ-aminolevulinic acid dehydratase (δ-ALA-D) [3], [4]. Accordingly, the need exists for the development of novel organic selenium compounds that provide pharmacological efficacy absent toxicity.
The present study focused on two different classes of organic selenium compounds, namely, monoselenides or β-selenoamines 1-phenyl-3-(p-tolylselanyl)propan-2-amine (C1) and 1-(2-methoxyphenylselanyl)-3-phenylpropan-2-amine (C2) and analogs of DPDS 1,2-bis(2-methoxyphenyl) diselenide (C3) and 1,2-bisp-tolyldiselenide (C4). These novel mono and diselenides have already been shown to possess antioxidant properties in vitro, associated with the the selenol formation [5].
The non-parasite nematode Caenorhabditis elegans (C. elegans) has been extensively used in toxicology, pharmacology and molecular biology studies, predominantely given its genetic power [6]. Amongst its many advantages are short life cycle, transparency, stereotypical development, compact genome, ease of breeding and small size [7]. Selenium compounds have been studied in this model, addressing their efficacy against oxidative stress-induced toxicity and attenuation of Aβ1-42 (amyloid-β) expression [8]. Seleno-xylofuranosides have also been shown to attenuate Mn-induced toxicity, by regulating the DAF-16/FOXO signaling pathway, and ebselen was shown to be effective in reversing Mn-induced reduction in survival and lifespan in C. elegans [9], [10].
Here, we assessed the toxicity of novel organic selenium compounds and DPDS in C. elegans. We evaluated survival, behavior, reproduction and AChE activity in response to oxidative stress and heat shock, addressing the efficacy of these novel compounds in attenuating toxicity.
2. Materials and methods
2.1. Organic selenium compounds
The organic selenium compounds 1-phenyl-3-(p-tolylselanyl) propan-2-amine (C1), 1-(2-methoxyphenylselanyl)-3-phenylpropan-2-amine (C2), 1,2-bis (2-methoxyphenyl) diselenide (C3), and 1,2-bisp-tolyldiselenide (C4) (Fig. 1) were synthesized according to Salman et al. and the chemical purity (99.9%) was assessed by hydrogen and carbon nuclear magnetic resonance and gas chromatography [11]. We compared the toxicity and efficacy of these novel compounds with a classical organic selenium compound, DPDS. All compounds were dissolved in dimethyl sulfoxide (DMSO).
Fig. 1.
Chemical structure and nomenclature of the β-selenoamines (C1 and C2), diphenyl diselenide (DPDS) and analogs (C3 and C4).
2.2. Worm maintenance
C. elegans Bristol N2 (wild-type) and CL2166 (N2; dvIs19 (Pgst-4::gfp; rol-6), glutathione S-transferase), were provided by the Caenorhabditis Genetic Center (CGC, University of Minnesota). All strains were grown at 20 °C on NGM plates (1.7% agar, 2.5 mg mL-1peptone, 25 mM NaCl, 50 mM KH2PO4 pH 6.0, 5 μg mL-1 cholesterol, 1 mM CaCl2, 1 mM MgSO4) with fresh Escherichia coli OP50 as food source [12]. For each experiment, synchronized populations were obtained by disruption of gravid adults. Worms were grown to the L4 larval stage on NGM/OP50-seeded plates.
2.3. C. elegans experimental treatments
L4 larval stage worms were exposed to the organic selenium compounds for one hour at different concentrations. Worms were transferred to a 1.5 mL conical tubes containing M9 buffer along with each of the compounds or DMSO (vehicle, at 1% maximum). Worms were kept under constant shaking for oxygenation during all phases of the liquid treatments. After one hour, animals were washed three times with M9 and transferred to NGM plates seeded with OP50. Worms were left for 30 min to acclimate on the plates prior to the behavioral assays, and for twenty-four hours for the survival assay and reproductive profile assessments.
2.4. Concentration range
Survival curves were generated for treatment (twenty-four hours) at 5, 25, 50, 100, 200 and 400 μM. Worms were considered dead when no movement response was obtained upon gentle touch and/or pharyngeal pumping was unnoted. Four experiments were individually performed in duplicates with approximately one hundred worms per treatment. After performing the dose-response curve for each of the organic selenium compounds, we chose the non-toxic dose of 200 μM for further assays (mentioned below).
2.5. Pharyngeal pumping
Pharyngeal pumping was individually verified for ten seconds at three time points in animals seeded over bacteria at 22 ± 2 °C [13]. The results were expressed as pharyngeal pumping/min. Ten worms were used in each experiment, with three independent replicates.
2.6. Defecation cycle
After treatment with organic selenium compounds, animals were washed with M9 and transferred to a fresh NGM plate seeded with E. coli OP50 [13]. The mean of three defecation cycles from each animal was used as indirect measurement of intestinal traffic. This experimental procedure was performed at 22 ± 2 °C with ten worms, and independently replicated in triplicate worm preparations.
2.7. Reproductive profile
C. elegans reproductive profile was analyzed twenty four hours after treatment. Egg-production represents the amount of eggs inside the uterus in each animal and egg-laying is represents the average of eggs laid over two hours. The results were expressed as the number of eggs/worm and eggs/worm/2 h respectively. Ten worms were used in each experiment, and independently replicated in triplicate worm preparations.
2.8. Body bends
L4 treated worms were individually transferred to E.coli-free NGM plates and allowed to move freely. After one min of adaptation, the number of body bends was scored in a 60-s time interval. A body bend was defined as a direction change in propagation of the posterior pharynx bulb along the y-axis, assuming the worm was traveling along the x axis [14]. Ten worms were observed at 22 ± 2 °C, and each experiment was independently replicated in triplicate worm preparations.
2.9. AChE activity
AChE activity in N2 L4 larval stage worms was analyzed with a colorimetric assay [15] with some modifications [16]. Following exposure to the organic selenium compounds, animals were washed three times in M9 buffer and transferred to microcentrifuge tubes. The samples were frozen three times in liquid nitrogen prior to sonication 9 × 10-s with 10-s breaks on ice at 30% amplitude, spun for 30 min at 15,000 rpm, and the supernatants (lysates) collected. Protein content was determined as previously described [17] with bovine serum albumin (BSA) as standard. A 160-μL portion of the sample was mixed with 1200 μL of 0.25 mM 5,5-dithiobis (2-nitrobenzoic acid) (DTNB), and 40 μL of 156 mM acetylthiocholine iodide (ASChI) and incubated at 30 °C for 5 min. The rate of change in absorbance was measured at 405 nm at 30-s intervals for one min by spectrophotometry. Kinetic measurements were recorded and converted to total cholinesterase activity using the extinction coefficient for the colored product, 5-thio-2-nitro-benzoic acid (II) [15]. Experiments were performed in duplicates on three different days with ∼500 worms per group.
2.10. Survival assay over juglone-induced oxidative stress
Worms at the L4 larval stage were treated with 200 μM of each compound as described above (∼500 worms per group). After three washes in M9, the worms were picked and transferred to tubes containing 100 μM juglone (5-hydroxy-1,4-naphthoquinone) or ethanol (EtOH, 1% maximum) [8]. After one hour of exposure under constant shaking, the worms were washed again with M9 and transferred to NGM plates where survival was examined twenty four hours later following the same criteria described above.
2.11. Stress induction of the GST-4 promoter
Worms at L4 larval stage were treated with each of the compounds (200 μM) for one hour and gently washed in M9 buffer. Next, animals were treated for one hour with juglone (50 μM) followed by three washes with M9, and transferred to a black microplate. For each measurement, twenty adult hermaphrodites were assessed for GFP fluorescence with a SpectraMax® i3 Multi-Mode Microplate Reader. Prior to the analysis, worms were concentrated in the middle of the wells by gently shaking the plate. GFP excitation was performed at 488 nm and emission was measured at 525 nm. In addition, a sample of treated worms was microscopically examined with an Olympus IX81 Inverted Microscope at 20× magnification using the green filter for fluorescence. The results were expressed as Arbitrary Fluorescence Units (AFU).
2.12. Heat shock stress
After pre-treatment with organic selenium compounds, worms were kept for four hours on food at 35 °C as a stress condition. Survival was assessed consecutively each hour during the heat shock, and results were expressed as% of survival (of a total of 100 worms per treatment). Approximately one hundred worms were counted per group in each independent experiment. Each experiment was independently replicated in triplicate worm preparations.
2.13. Minimum inhibitory concentration (MIC) in E. coli OP50
To verify whether the organic selenium compounds has influence on E.coli OP50 growth, we assessed the MIC. Bacteria was seeded on plates with Mueller Hinton agar and allowed to grow for twenty four hours at 37 °C. Next, we prepared suspensions of microorganisms in Mueller Hinton broth. The MIC was performed according to Clinical and Laboratory Standards Institute [18]. A total of 50 μL of the standardized microorganism suspension was placed in each test well of a 96-well microtiter plate, along with an equal volume of compound to be tested at different concentrations. We performed a broth control, a growth control, and a compound vehicle control to which the results were compared. The plates were incubated for twenty four hours at 37 °C. The MIC was considered as the lowest concentration of the test product able to inhibit the growth of microorganisms evidenced by the use of 2,3,5 triphenyltetrazolium chloride 1%.
2.14. Protein quantification
Protein concentration was estimated by the Bradford method with bovine serum albumin as the standard [17].
2.15. Statistical analysis
All data are expressed as means ± S.E.M for each experimental group. Determination of statistical significance was performed by one-way analysis of variance (ANOVA) followed by Newman–Keuls Multiple Comparison Test where appropriate. Differences between groups were considered to be significant at p < 0.05.
3. Results
3.1. C. elegans behavior under organic selenium compounds exposure
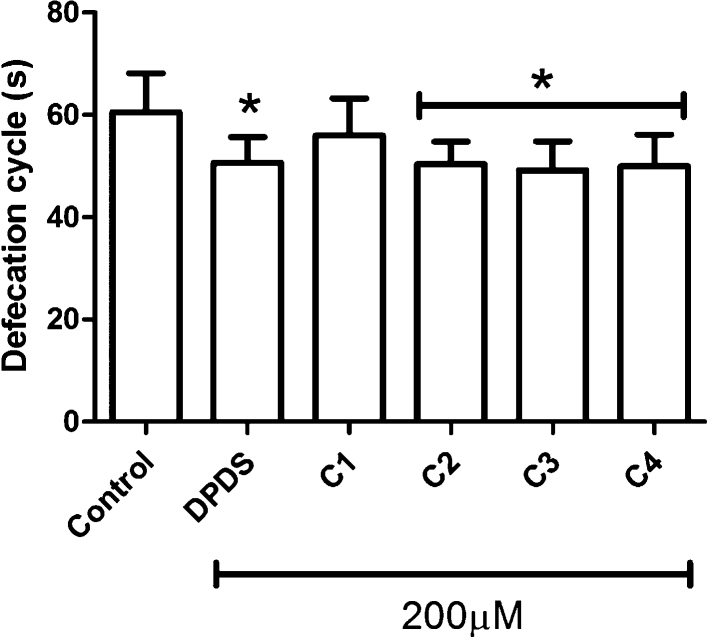
Pre-treatment with DPDS and the organic selenium compounds did not interfere with C. elegans egg-laying, egg-production, pharyngeal pumping and body bends (data not shown). However, treatment with the organic selenium compounds, excepted C1 produce a significantly decreased the defecation cycle when compared to non-treated worms (Fig. 2).
Fig. 2.
C. elegans behavior upon treatment with novel organic selenium compound.
Intervals of defecation cycles (s) were assessed at the L4 larval stage 30 min after the compoundś exposure on NGM plates. * indicates statistical significance (p < 0.05) compared to the control group by means of one-way ANOVA followed by Newman–Keuls Multiple Comparison Test (mean ± SEM). Ten animals were observed in three independent experiments.
3.2. C. elegans AChE activity following exposure to organic selenium compounds
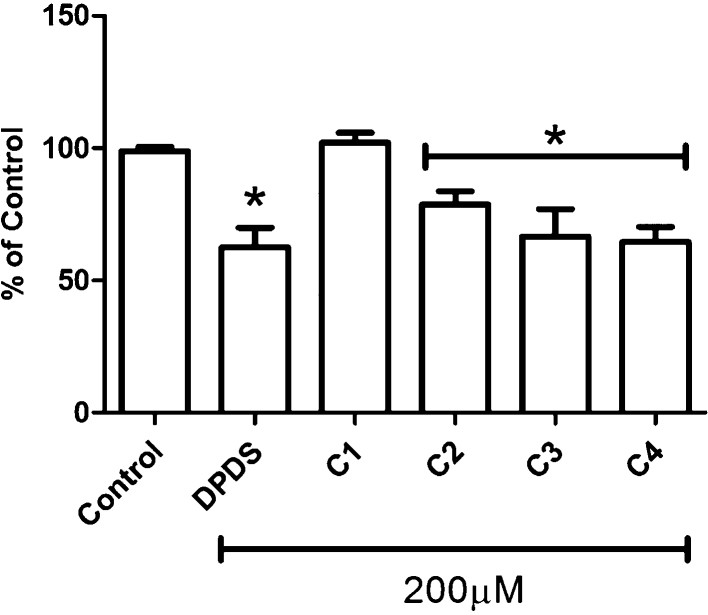
C2, DPDS, C3 and C4 significantly diminished AChE activity (p < 0.05), compared to the control group, (Fig. 3). C1, which contains a p-methyl inclusion, failed to alter AChE activity. The absolute control value was 41.34 ± 1.67 μmol/min/mg protein.
Fig. 3.
AChE enzyme activity was significantly reduced by treatment with novel organic selenium compounds, except C1, in C. elegans.
The activity of AChE was studied in L4 larval stage worms following novel organic selenium compound treatment. * indicates statistical significance (p < 0.05) compared to the control group with one-way ANOVA followed by Newman–Keuls Multiple Comparison Test (mean ± SEM). Five hundred animals were used in three independent experiments.
3.3. Organic selenium compounds protect against juglone-induced oxidative stress
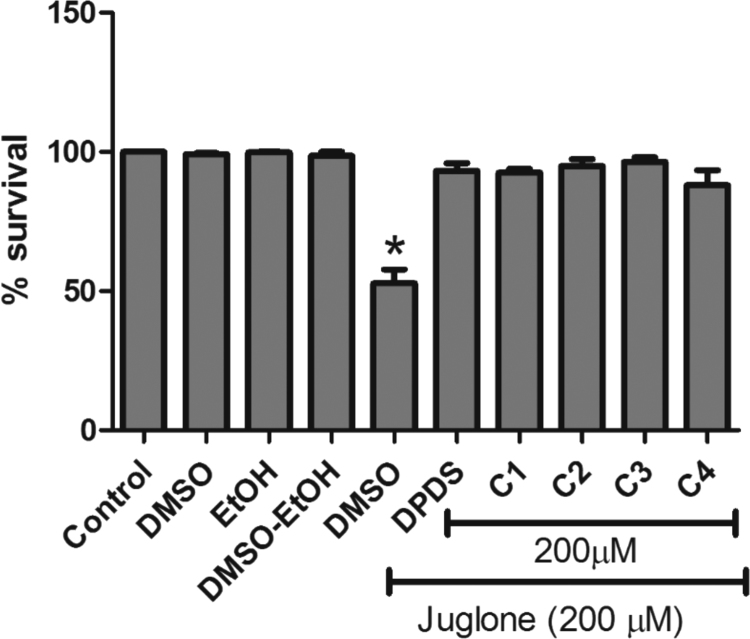
Juglone treatment significantly decreased the survival rate in C. elegans (Fig. 4, p < 0.05 compared to the control worms). Pre-treatment with DPDS or the organic selenium compounds protected the worms against juglone-induced toxicity, restoring viability to levels indistinguishable from controls.
Fig. 4.
C. elegans survival was not affected by 200 μM treatment with novel organic selenium compounds.
Worms were treated with each of the compounds for 1 h or vehicle (DMSO for organic selenium compound, EtOH for juglone and M9 buffer for control). After three washes in M9, the worms were transferred to NGM plates and the viability assessed 24 h later. Results are shown as% of survival of total worms (mean ± SEM) of three independent experiments. * indicates statistical significance (p < 0.05) compared to the control group with one-way ANOVA followed by Newman-Keuls Multiple Comparison Test.
3.4. Effect of organic selenium compounds on the GST-4 promoter fluorescence
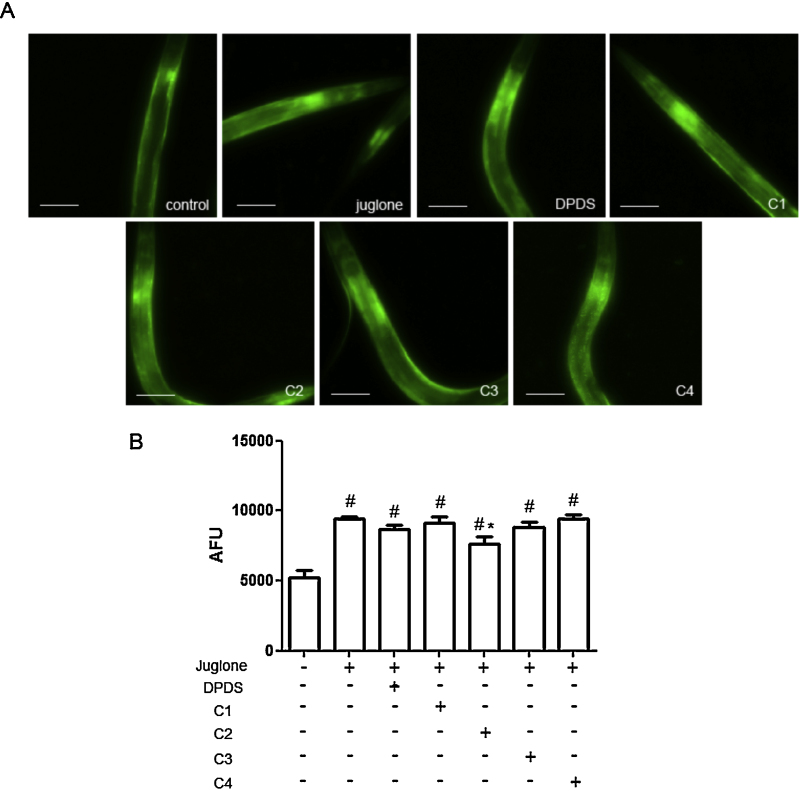
The fluorescence of GST-4::GFP transgenic worms was quantified after pre-treatment with organic selenium compounds followed by juglone stress induction (Fig. 5A and B). In addition, only C2 was able to prevent the juglone-induced GST-4 increase in fluorescence (p < 0.05, C2 in comparison to juglone treated group).
Fig. 5.
GST-4::GFP expression is induced by juglone and suppressed by the C2 organic selenium compound.
Worms were pre-treated with each of the compounds for 1 h (200 μM), washed three times in M9 and subjected to juglone treatment (200 μM) for 1 h. GST-4::GFP fluorescence was microscopically evaluated with an Olympus IX81 Inverted Microscope at 20× magnification, using the green filter for fluorescence (A) and with the SpectraMax® i3 Multi-Mode Microplate Reader (B) # represents statistically significant (p < 0.001) difference compared to control and * indicates statistically significant (p < 0.05) difference compared to the juglone treated group. Analysis was carried out by means of one-way ANOVA followed by Newman–Keuls Multiple Comparison Test (mean ± SEM). Twenty animals were observed in four independent experiments.
3.5. Organic selenium compounds protect against heat shock stress
The heat shock stress produced a time-dependent decrease in worm survival. In addition, treatment with mono- or diselenides failed to increase C. elegans viability over the heat shock induced stress (data not shown).
3.6. Effects of organic selenium compounds on E. coli OP50
The organic selenium compounds did not cause a significant antimicrobial effect on E. coli OP50 growth. In addition, all compounds tested at a concentration range between 5 and 400 μM showed analogous levels to the positive bacteria control group (data not shown).
4. Discussion
A number of organic selenium compounds have been studied, assessing their efficacy in attenuating oxidative stress generation [3], [19]. Their antioxidant capacity is predominantly ascribed to their mimetic activity of endogenous antioxidant enzymes, as well as the production of selenol group [20], [21]. Nevertheless, several studies have also reported enhanced toxicity inherent to these organic selenium compounds, likely related to the catalytic oxidation of thiol groups from glutathione (GSH) or different proteins and enzymes [1], [3]. Accordingly, the search for efficacious organic compounds has yet to materialized.
Herein, structural modifications to classical selenium compounds, i.e., ebselen and DPDS, were made to synthesize novel molecules with inherent capacity to produce selenol groups, higher stability and low toxicity [21], [22]. Our group has shown that β-selenoamines and anologs of DPDS possess enhanced in vitro antioxidant properties, such as a significant elevation in both GPx and TRxR activity [5], [23]. In this regard, we assessed biological and toxicological activities of these novel organic selenium compounds (in comparison with DPDS) in the C. elegans model.
To the best of our knowledge, this is the first trial study which addressed the effects of these novel organic selenium compounds C1, C2, C3 and C4 on a whole organism. Our studies suggest lack of effects of these compounds on C. elegans survival at doses as high as 200 μM (data not shown). Furthermore, the novel organic selenium compounds did not affect reproduction, feeding and movement behaviors (data not shown). Interestingly, we observed DPDS-, C2-, C3- and C4-induced decrease on the frequency of defecation cycles (Fig. 2). This behavior is a stereotypic motor program that in the nematode occurs approximately every 50 s [24]. Each cycle begins with a posterior body contraction that drives gut contents to the anterior part of the worm and is followed by an anterior body muscle contraction that pushes expels the gut contents toward the posterior of the animal, followed by an enteric muscle contraction that expels gut contents [7]. Among many pathways which orchestrate the defecation motor program in C. elegans [7], we affirmed herein that novel selenium compounds interfere with cholinergic system (Fig. 3). The neurotransmitter ACh has been found at low ranges in cha-1 mutants (cha-1 choline acetyltransferase) [25], which exhibit an increase in the interval between defecation cycles. The tested compounds, except C1, caused a significant reduction in the AChE activity (Fig. 3), suggests that greater availability of synaptic ACh is responsible for the noted decrease the defecation cycle time. Furthermore, C1 could not diminish the AChE activity (Fig. 3), which corroborates with the absence of effects over defecation cycle interval (Fig. 2). The AChE reduction caused by selenium compounds, such as ebselen and DPDS, have been also been reported in previous studies using rat brains in vitro and mouse ex vivo, respectively [26], [27].
We further characterized whether the novel organic selenium compounds attenuated juglone-induced stress. Our findings demonstrate that juglone reduced the worms' viability by ∼50%, and that pre-treatment with organic selenium compounds fully abolished this effect to levels indistinguishable from controls. This effect was inherent to all the tested novel organic selenium compounds, showing full protection against juglone-induced toxicity (Fig. 4).
Juglone is a reactive oxygen-generating naphthoquinone and it has been previously studied in C. elegans to define genes that promote oxidative stress resistance [28], [29]. Among many stress-related genes, gst-4 is upregulated upon juglone exposure [30]. Our data corroborate the ability of juglone to potently induce GST-4::GFP expression, an effect which was significantly reduced by C2 (Fig. 5). C2's efficacy in attenuating juglone-induced stress may thus reflect its ability to increase GST-4 levels. DPDS, C1, C3 and C4 failed to decrease GST-4::GFP fluorescence even at doses that significantly reduced juglone toxicity (Fig. 4). Juglone can induce oxidative stress through various mechanism, such as superoxide (O2−•), hydroxyl radical (•OH) and hydrogen peroxide (H2O2) formation, triggering toxic effects by protein modifications [31], [32], lipid oxidation [33] and nuclear DNA damage [34]. Earlier studies from our group have demonstrated that DPDS analogs were able to protect against thioacetamide-induced oxidative stress in mice [35]. However, further studies are needed to verify the effects of the novel organic selenium compounds via an analogous mechanism in C. elegans.
Organic selenium compounds are known to possess antimicrobial activity [36]. Accordingly, we performed the minimum inhibitory concentration (MIC) of these compounds in E. coli OP50 to ascertain whether they might cause a decrease bacterial viability, and thus interfere in the worms' lifespan. Our results established that neither of the novel organic selenium compounds at concentration tested can diminish the E. coli OP50 quality, which provide further impetus for future studies regarding C. elegans development and lifespan.
In conclusion, we have demonstrated that structural changes made in the C1, C2, C3 and C4 organic selenium compounds prevented juglone-induced stress in C. elegans, mimicking the efficacy of DPDS. Furthermore, our findings indicate that C1, in addition to possessing high efficacy against juglone-invoked stress, also maintained optimal AChE activity and normal defecation cycle in the worm. This effect may reflect the presence of amino group and p-methyl insertions in the C1 molecule. Combined, these findings suggest the novel organic selenium compounds have promising pharmacological properties in attenuating oxidative stress conditions. Future studies could be profitably directed at deciphering their efficacy in other animal models and establishing the molecular mechanisms inherent to this protection.
Conflict of interest
The authors report no financial or other conflict of interest relevant to the subject of this article.
Acknowledgments
This work was carried out with funds from Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq Universal #472669/2011-7 and #475896/2012-2), Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES for providing fellowship to S.T.S. and P.G.) and Fundação de Amparo à Pesquisa do Estado do Rio Grande do Sul (Programa de Apoio a Núcleos emergentes - PRONEM/FAPERGS #11/2029-1, for providing fellowship to B.P.). Prof. Michael Aschner was supported in part by grants from the National Institute of Environmental Health Sciences (NIEHS) R01 ES10563 and R01 ES 07731.
References
- 1.Nogueira C.W., Zeni G., Rocha J.B.T. Organoselenium and organotellurium compounds: toxicology and pharmacology. Chem. Rev. 2004;104(12):6255–6286. doi: 10.1021/cr0406559. [DOI] [PubMed] [Google Scholar]
- 2.Sarma B.K., Mugesh G. Antioxidant activity of the anti-inflammatory compound ebselen: a reversible cyclization pathway via selenenic and seleninic acid intermediates. Chemistry. 2008;14(34):10603–10614. doi: 10.1002/chem.200801258. [DOI] [PubMed] [Google Scholar]
- 3.Nogueira C., Rocha J.T. Toxicology and pharmacology of selenium: emphasis on synthetic organoselenium compounds. Arch. Toxicol. 2011;85(11):1313–1359. doi: 10.1007/s00204-011-0720-3. [DOI] [PubMed] [Google Scholar]
- 4.Mautner H.G. Isologous oxygen, sulfur, and selenium compounds as probes of acetylcholine receptors. J. Gen. Physiol. 1969;54(1):271–277. doi: 10.1085/jgp.54.1.271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Stefanello S.T. Evaluation of in vitro antioxidant effect of new mono and diselenides. Toxicol. In Vitro. 2013;27(5):1433–1439. doi: 10.1016/j.tiv.2013.03.001. [DOI] [PubMed] [Google Scholar]
- 6.Leung M.C.K. Caenorhabditis elegans: an emerging model in biomedical and environmental toxicology. Toxicol. Sci. 2008;106(1):5–28. doi: 10.1093/toxsci/kfn121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Riddle D.L., Blumenthal T., Meyer B.J., Priess J.R. Introduction to C. elegans. In: Riddle D.L., Blumenthal T., Meyer B.J., Priess J.R., editors. C. elegans II. Cold Spring Harbor Laboratory Press; Plainview, New York: 1997. pp. 1–22. [Google Scholar]
- 8.Zamberlan D.C. Diphenyl-diselenide suppresses amyloid-β peptide in Caenorhabditis elegans model of Alzheimer’s disease. Neuroscience. 2014;278(0):40–50. doi: 10.1016/j.neuroscience.2014.07.068. [DOI] [PubMed] [Google Scholar]
- 9.Avila D.S. Organotellurium and organoselenium compounds attenuate Mn-induced toxicity in Caenorhabditis elegans by preventing oxidative stress. Free Radic. Biol. Med. 2012;52(9):1903–1910. doi: 10.1016/j.freeradbiomed.2012.02.044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Wollenhaupt S.G. Seleno- and telluro-xylofuranosides attenuate Mn-induced toxicity in C. elegans via the DAF-16/FOXO pathway. Food Chem. Toxicol. 2014;64:192–199. doi: 10.1016/j.fct.2013.11.030. [DOI] [PubMed] [Google Scholar]
- 11.Salman S.M. CuO nano particles and [bmim]BF4: an application towards the synthesis of chiral [small beta]-seleno amino derivatives via ring opening reaction of aziridines with diorganyl diselenides. RSC Adv. 2012;2(22):8478–8482. [Google Scholar]
- 12.Wood W.B. Cold Spring Harbor Laboratory Press; 1988: 2015. The Nematode Caenorhabditis elegans. [Google Scholar]
- 13.Gubert P. Behavioral and metabolic effects of the atypical antipsychotic ziprasidone on the nematode Caenorhabditis elegans. PLoS One. 2013;8(9):e74780. doi: 10.1371/journal.pone.0074780. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Tsalik E.L., Hobert O. Functional mapping of neurons that control locomotory behavior in Caenorhabditis elegans. J. Neurobiol. 2003;56(2):178–197. doi: 10.1002/neu.10245. [DOI] [PubMed] [Google Scholar]
- 15.Ellman G.L. A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem. Pharmacol. 1961;7(2):88–95. doi: 10.1016/0006-2952(61)90145-9. [DOI] [PubMed] [Google Scholar]
- 16.Cole R.D., Anderson G.L., Williams P.L. The nematode Caenorhabditis elegans as a model of organophosphate-induced mammalian neurotoxicity. Toxicol. Appl. Pharmacol. 2004;194(3):248–256. doi: 10.1016/j.taap.2003.09.013. [DOI] [PubMed] [Google Scholar]
- 17.Bradford M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976;72(1-2):248–254. doi: 10.1006/abio.1976.9999. [DOI] [PubMed] [Google Scholar]
- 18.Clinical Laboratory Standards Institute . CLSI document M7-A7. 7th ed. Wayne [PA]: Clinical and Laboratory Standards Institute; 2006. Methods for dilution antimicrobial susceptibility tests for bacteria that grow aerobically: approved standard. [Google Scholar]
- 19.Brenneisen P., Steinbrenner H., Sies H. Selenium, oxidative stress, and health aspects. Mol. Aspects Med. 2005;26(4-5):256–267. doi: 10.1016/j.mam.2005.07.004. [DOI] [PubMed] [Google Scholar]
- 20.Papp L.V. From selenium to selenoproteins: synthesis, identity, and their role in human health. Antioxid Redox Signal. 2007;9(7):775–806. doi: 10.1089/ars.2007.1528. [DOI] [PubMed] [Google Scholar]
- 21.Mugesh G., du Mont W.W., Sies H. Chemistry of biologically important synthetic organoselenium compounds. Chem. Rev. 2001;101(7):2125–2179. doi: 10.1021/cr000426w. [DOI] [PubMed] [Google Scholar]
- 22.Mugesh G., Singh H.B. Synthetic organoselenium compounds as antioxidants: glutathione peroxidase activity. Chem. Soc. Rev. 2000;29(5):347–357. [Google Scholar]
- 23.Prestes A.S. Antioxidant activity of beta-selenoamines and their capacity to mimic different enzymes. Mol. Cell. Biochem. 2012;365(1-2):85–92. doi: 10.1007/s11010-012-1246-6. [DOI] [PubMed] [Google Scholar]
- 24.Thomas J.H. Genetic analysis of defecation in Caenorhabditis elegans. Genetics. 1990;124(4):855–872. doi: 10.1093/genetics/124.4.855. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Avery L. The genetics of feeding in Caenorhabditis elegans. Genetics. 1993;133(4):897–917. doi: 10.1093/genetics/133.4.897. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Luchese C. Efficacy of diphenyl diselenide against cerebral and pulmonary damage induced by cadmium in mice. Toxicol. Lett. 2007;173(3):181–190. doi: 10.1016/j.toxlet.2007.07.011. [DOI] [PubMed] [Google Scholar]
- 27.Martini F., Brunning C.A., Soares Nogueira S.M.C.W., Zeni G. Inhibitory effect of ebselen on cerebral acetylcholinesterase activity in vitro: kinetics and reversibility of inhibition. Curr. Pharm. Des. 2014;21(7):920–924. doi: 10.2174/1381612820666141014124319. [DOI] [PubMed] [Google Scholar]
- 28.Strayer A. Expression of the small heat-shock protein Hsp-16-2 in Caenorhabditis elegans is suppressed by Ginkgo biloba extract EGb 761. FASEB J. 2003 doi: 10.1096/fj.03-0376fje. [DOI] [PubMed] [Google Scholar]
- 29.de Castro E., Hegi de Castro S., Johnson T.E. Isolation of long-lived mutants in Caenorhabditis elegans using selection for resistance to juglone. Free Radic. Biol. Med. 2004;37(2):139–145. doi: 10.1016/j.freeradbiomed.2004.04.021. [DOI] [PubMed] [Google Scholar]
- 30.Przybysz A.J. Increased age reduces DAF-16 and SKN-1 signaling and the hormetic response of Caenorhabditis elegans to the xenobiotic juglone. Mech. Ageing Dev. 2009;130(6):357–369. doi: 10.1016/j.mad.2009.02.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Inbaraj J.J., Chignell C.F. Cytotoxic action of juglone and plumbagin: a mechanistic study using HaCaT keratinocytes. Chem. Res. Toxicol. 2003;17(1):55–62. doi: 10.1021/tx034132s. [DOI] [PubMed] [Google Scholar]
- 32.Stadtman E. Protein oxidation and aging. Science. 1992;257(5074):1220–1224. doi: 10.1126/science.1355616. [DOI] [PubMed] [Google Scholar]
- 33.Fong K.-L. Evidence that peroxidation of lysosomal membranes is initiated by hydroxyl free radicals produced during flavin enzyme activity. J. Biol. Chem. 1973;248(22):7792–7797. [PubMed] [Google Scholar]
- 34.Bjelland S., Seeberg E. Mutagenicity, toxicity and repair of DNA base damage induced by oxidation. Mutat Res. 2003;531(1-2):37–80. doi: 10.1016/j.mrfmmm.2003.07.002. [DOI] [PubMed] [Google Scholar]
- 35.Stefanello S.T. Effect of diselenide administration in thioacetamide-induced acute neurological and hepatic failure in mice. Toxicol. Res. 2015;4(3):707–717. [Google Scholar]
- 36.Wójtowicz H. Azaanalogues of ebselen as antimicrobial and antiviral agents: synthesis and properties. II Farmaco. 2004;59(11):863–868. doi: 10.1016/j.farmac.2004.07.003. [DOI] [PubMed] [Google Scholar]