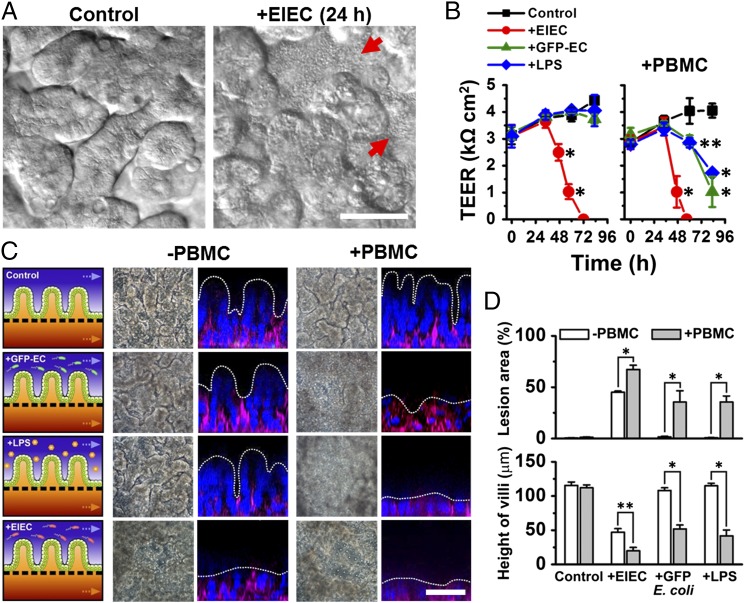
Fig. 2.
Reconstitution of pathological intestinal injury induced by interplay between nonpathogenic or pathogenic enteroinvasive E. coli bacteria or LPS endotoxin with immune cells. (A) DIC images showing that the normal villus morphology of the intestinal epithelium cultured on-chip (Control) is lost within 24 h after EIEC (serotype O124:NM) are added to the apical channel of the chip (+EIEC; red arrows indicate bacterial colonies). (B) Effects of GFP-EC, LPS (15 µg/mL), EIEC, or no addition (Control) on intestinal barrier function (Left). Right shows the TEER profiles in the presence of human PBMCs (+PBMC). GFP-EC, LPS, and EIEC were added to the apical channel (intestinal lumen) at 4, 12, and 35 h, respectively, and PBMCs were subsequently introduced through the lower capillary channel at 44 h after the onset of experiment (0 h) (n = 4). (C) Morphological analysis of intestinal villus damage in response to addition of GFP-EC, LPS, and EIEC in the absence (−PBMC) or the presence of immune components (+PBMC). Schematics (experimental setup), phase contrast images (horizontal view, taken at 57 h after onset), and fluorescence confocal micrographs (vertical cross-sectional views at 83 h after onset) were sequentially displayed. F-actin and nuclei were coded with magenta and blue, respectively. (D) Quantification of intestinal injury evaluated by measuring changes in lesion area (Top; n = 30) and the height of the villi (Bottom; n = 50) in the absence (white) or the presence (gray) of PBMCs. Intestinal villi were grown in the gut-on-a-chip under trickling flow (30 µL/h) with cyclic deformations (10%, 0.15 Hz) during the preculture period for ∼100 h before stimulation (0 h, onset). Asterisks indicate statistical significance compared with the control at the same time point (*P < 0.001, **P < 0.05). (Scale bars, 50 µm.)