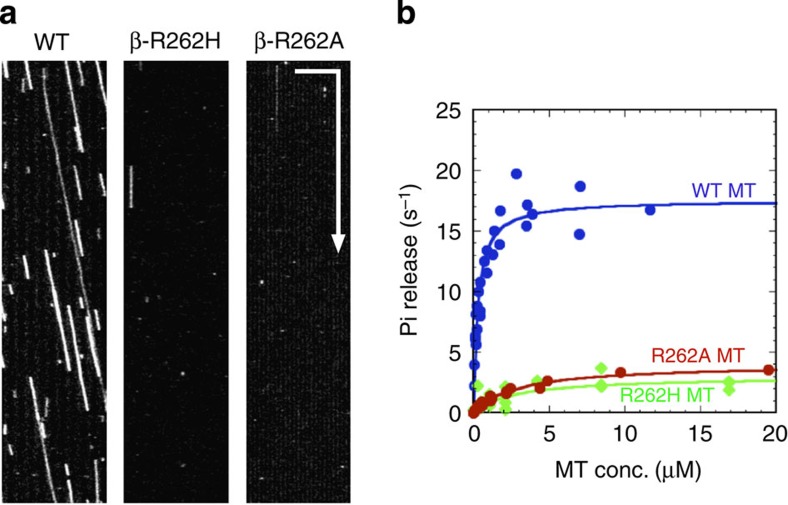
Figure 2. Effect of tubulin mutations β-R262H and β-R262A on single-molecule motility and MT-activated ATPase activity of the KIF5B motor.
(a) Kymographs of kinesin motility. Horizontal bar, 5 μm; vertical arrow, 10 s. (b) The ATPase activity of KIF5B activated by WT (blue circles), R262H (green diamonds) or R262A MTs (red circles). All data obtained from four independent measurements for WT, three measurements for β-R262H and three measurements for β-R262A are plotted in the graph. Smooth curves were the best fit for the Michaelis–Menten equation with kcat and K0.5MT values given in Table 1.