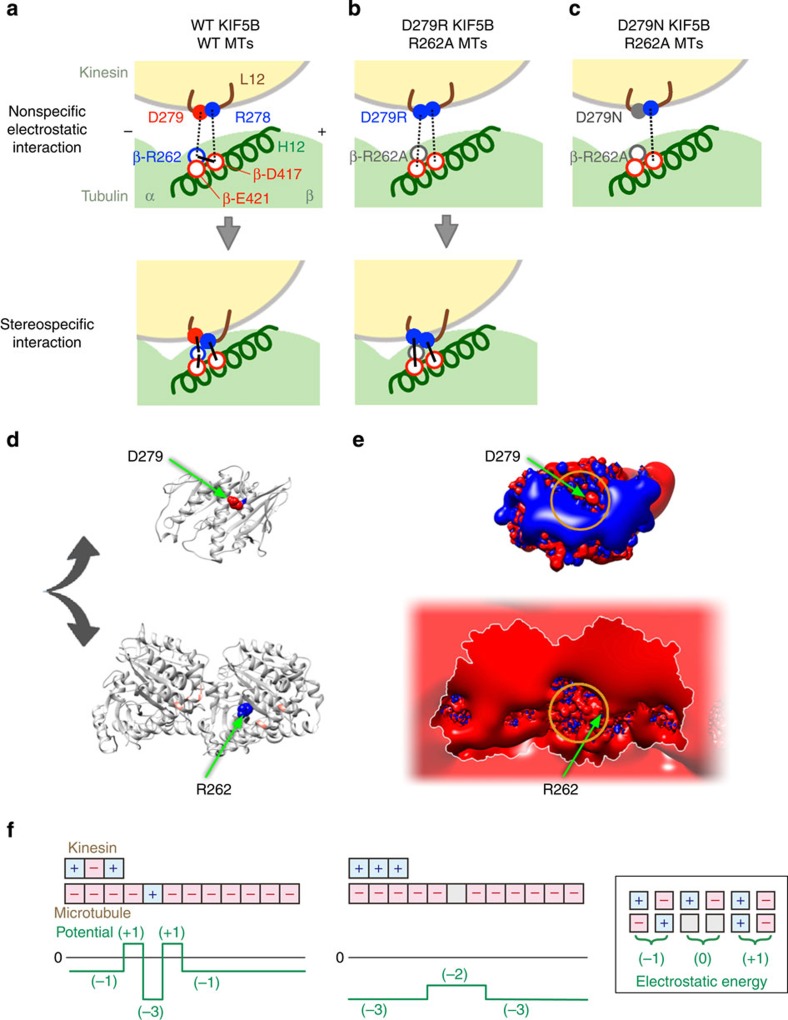
Figure 5. Schematic working model for the electrostatic latch.
(a–c) Possible models for kinesin–MT interactions in the WT KIF5B–WT MT pair (a), D279R KIF5B-β-R262A MT pair (b) and D279N KIF5B-β-R262A MT pair (c). Red-, blue- and grey-filled circles represent basic, acidic and neutral residues in kinesin, respectively, whereas red, blue and grey unfilled circles represent basic, acidic and non-polar residues in tubulin, respectively. Dotted and thick lines indicate electrostatic attraction and the salt bridge, respectively. (d) Location of the residues D279 and R262 shown in the structure of KIF5B (top) and tubulin (bottom), based on the crystal structure of the KIF5B–tubulin complex in the nucleotide-free state20. (e) Three-dimensional isopotential contours for the KIF5B (top) and tubulin (bottom), calculated from the interface shown in d. The values of the contours are −2.5 kT e−1 (red) and 2.5 kT e−1 (blue) for the KIF5B and −26 kT e−1 (red) and 26 kT e−1 (blue) for tubulin, respectively, where k is the Boltzmann constant, T is the temperature and e is the magnitude of the electron charge. The arrows indicate the position of the residue D279 and β-R262. The yellow circles indicate the areas where the direction of the electrostatic field is locally reversed. (f) The schematic representation of the effect of the charge-reversed pair at the kinesin–MT interface on the electrostatic energy of the kinesin–MT interaction. For simplicity, the space has been one-dimensionalized and discretized. (Left) ‘Kinesin charges' (upper three boxes) electrostatically interact with ‘MT charges' (lower array of boxes), where the size of the box is roughly the Debye screening length58. The green line in the graph shows the electrostatic energy of the interaction between kinesin and MT, plotted as a function of the centre position of kinesin along the MT. The energy of each charge pair face-to-face is defined in the inset. In the WT KIF5B–WT MT pair (left), the energy landscape shows a distinct localized dip and peripheral banks, although the average binding energy is reduced as compared with that in the paired mutant D279R KIF5B-β-R262A MT (right).