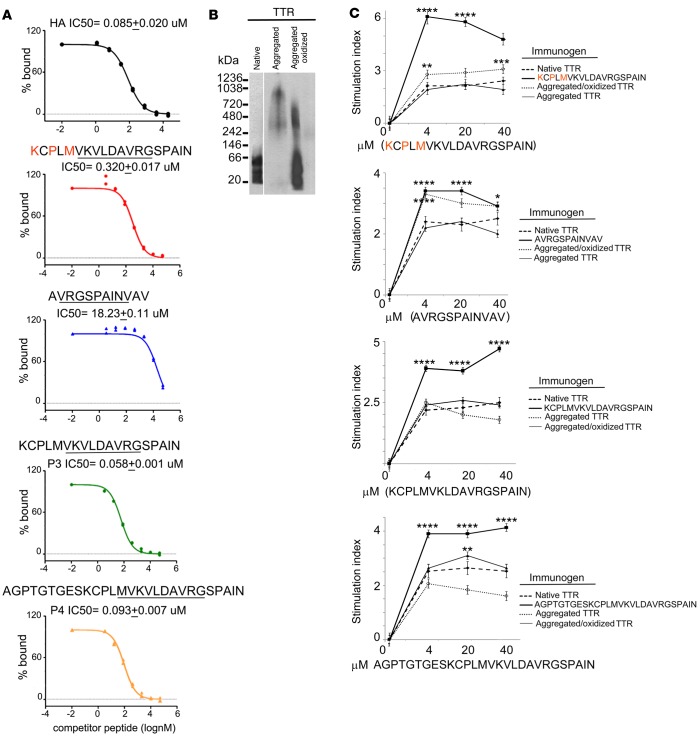
Figure 8. HLA-DR1–binding affinity and proliferative response to transthyretin peptides following immunization of HLA-DR1 mice with native, aggregated, or aggregated/oxidized transthyretin.
(A) Inhibition binding curves (percentage bound test peptide vs. test peptide concentration [nM]) for transthyretin (TTR) peptides. Binding to HLA-DR1 molecules measured at 72 hours (1 out of 3 representative experiments is shown). Shown at the top of the graphs are the peptide sequences as well as the calculated values for binding (IC50 [μM]). Binding of the immunodominant viral epitope HA peptide is shown as control. (B) Native gel showing native TTR (monomer at 15 kDa and tetramer at 60 kDa); aggregated TTR, following exposure to low pH; and aggregated and oxidized TTR, following oxidation by Fenton reaction. One out of three gels is shown. (C) T cell–proliferative responses to increasing concentrations of the reported TTR peptides following immunization with native or aggregated or aggregated/oxidized TTR. HLA-DR1 mice were immunized as reported in Methods, popliteal and axillary nodes were harvested 3 weeks later, and T cells were rechallenged in vitro with increasing concentrations of the reported immunogens. Data are reported as stimulation index (BrDu incorporation following antigen stimulation over BrDu incorporation in absence of specific antigen). Data were compiled from 3 separate immunizations (n = 6). Experiments to evaluate T cell proliferation in response to antigen were run in quadruplicate for each antigen concentration tested. Mean ± SD. Data were analyzed by 1-way ANOVA (P < 0.005) and Tukey test. Asterisks indicate statistical significance, calculated at each concentration, between the peptide stimulation index and the native TTR or between the native TTR and the unfolded/carbonylated TTR. *P < 0.05; **P < 0.01; ***P < 0.005; ****P < 0.001.