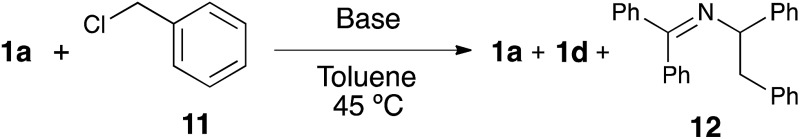
Table 2. The role of the base a .
| ||||
| Entry | Base (equiv.) | 11 (equiv.) | 12 b (%) | Notes |
| 1 | KHMDS (2.0) | 2.0 | 81 | — |
| 2 | NaHMDS (2.0) | 0.10 | 80 | — |
| 3 | KOtBu (2.0) | 0.10 | 60 | Side-products observed |
| 4 | KHMDS (0.5) | — | — | Only 1a and 1d observed |
| 5 | NaHMDS (0.5) | — | 24 | Side-products observed |
| 6 c | KHMDS (0.5) | — | — | Only 1a and 1d observed |
aConditions: benzyl chloride (0.24 mmol, 1.2 equiv., or none), imine 1a (0.2 mmol, 1.0 equiv.), base (0.4 mmol or 0.1 mmol, 2.0 equiv. or 0.5 equiv.), toluene (0.6 mmol), 45 °C, 3 hours.
bCalculated by NMR analysis using dimethyl malonate as an internal standard.
cReaction performed in the presence of 5% [Ni(COD)2]/10% IPr.
