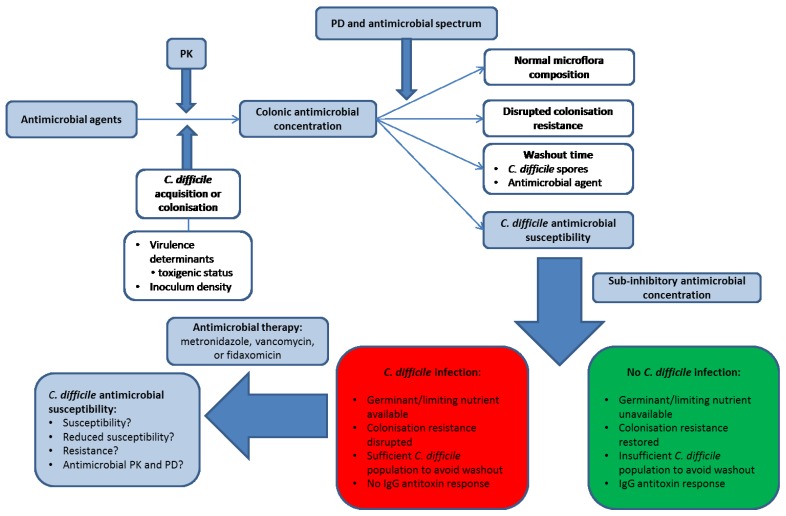
Figure 1.
Factors involved in the development of Clostridium difficile infection (CDI). Blue shaded boxes involve antimicrobial agents. PK, pharmacokinetics; PD, pharmacodynamics. Antimicrobial agents differ in their pharmacokinetics (PK) and therefore in the luminal concentration that is present (and bioactive) in the human colon. In order for CDI to develop, a patient must either acquire toxigenic C. difficile from an exogenous source or be colonised with an endogenous strain. The antimicrobial agent disrupts the indigenous microflora to an extent governed by its antimicrobial spectrum of activity and pharmacodynamic (PD) profile, and a steady state concentration of antimicrobial agent may be reached by the end of dosing. The antimicrobial agent (and C. difficile spores) may then be washed out of the colon to a sub-inhibitory concentration, the speed of which depends on the steady-state drug concentration in the colonic lumen and the colonic transit time of the patient. The observation and timing of CDI depends on host immunological factors, C. difficile-specific factors (antimicrobial susceptibility to the inciting agent and the inoculum density of spores), and the rate and extent of recovery in the indigenous gut microflora. C. difficile spores do not outgrow in the presence of supra-inhibitory concentrations of antimicrobial agents, but once concentrations are sub-inhibitory then CDI may develop assuming the nutritional environment is conducive for spore germination and outgrowth (this may indicate prolonged disruption of colonisation resistance) and also that there is no IgG antitoxin response in the patient. Antimicrobial therapy for CDI may then be initiated by clinicians, the initial success of which depends on the PK/PD profile of the therapeutic agent and the susceptibility of the C. difficile strain. Whether recurrent CDI is subsequently observed may then depend on the degree to which the CDI therapy has negatively impacted on the indigenous gut microflora, the concentration of C. difficile spores remaining in the colon of the patient, whether the antimicrobial agent may persist in the colon or potentially adhere to C. difficile spores, and whether an IgG antitoxin response is observed.