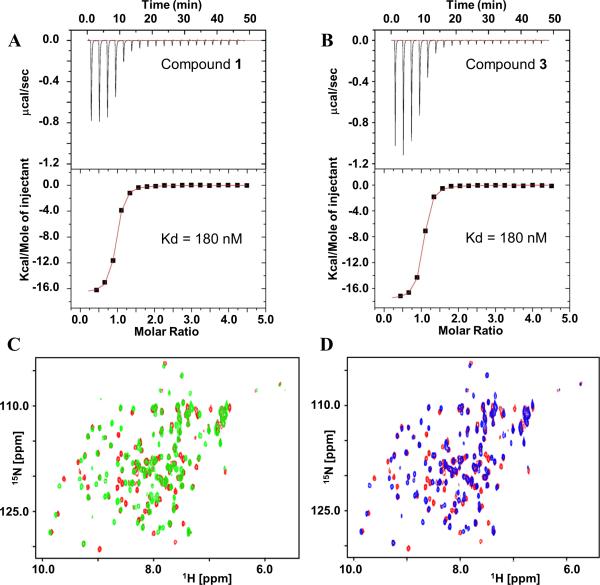
Figure 3. Comparative binding data of compounds 1 and 3.
Isothermal titration calorimetry data of binding to YopH-NT for A) compound 1 (Kd = 180 nM, ΔH = −16.5 Kcal/mole, −TΔS= 7.4 Kcal/mole, n = 0.87) and B) compound 3 (Kd = 180 nM, ΔH = −17.6 Kcal/mole, −TΔS= 8.43 Kcal/mole, n =0.96). C) Overlay of [1H,15N] HSQC spectra of YopH-NT (50 μM) in free form (red) and in presence of compound 1 (green, 150 μM). D) Overlay of [1H,15N] HSQC spectra of YopH-NT (50 μM) in free form (red) and in presence of compound 3 (blue, 150 μM). Both compounds produce very similar perturbations to YopH-NT suggesting a similar binding mode.