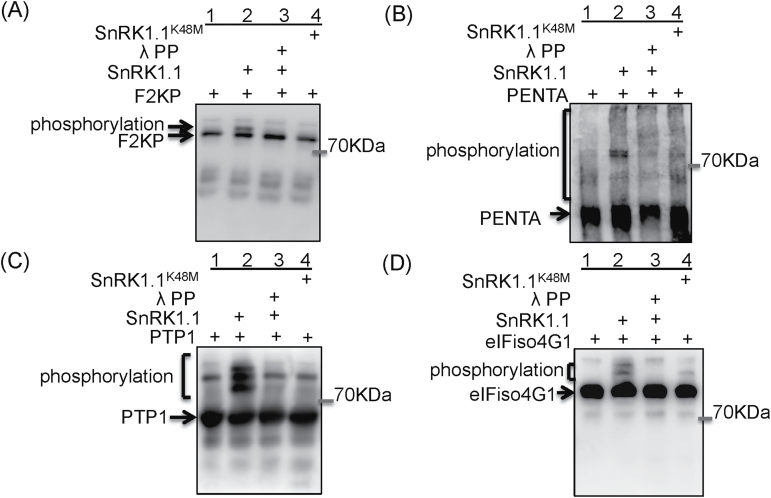
Fig. 4.
In vitro phosphorylation assays of SnRK1. The recombinant proteins fused with the S tag, (A) F2KP, (B) Penta, (C) PTP1, and (D) eIFiso4G1, were mixed with immunoprecipitated active SnRK1.1 (IP-SnRK1.1-S) or inactive SnRK1.1K48M and the phosphorylation of these recombined proteins was examined with Phos-Tag page and S-tag immunoblotting. Phosphorylated protein mobility is retarded in Phos-Tag PAGE and this retardation can be reduced by the addition of λ phosphatase. Lane 1 of each blot was loaded with recombinant protein only, representing the mobility of non-phosphorylated substrate. In the second and fourth lanes of each blot, the four candidates showed mobile retardation in IP-active SnRK1.1 treatment but not in inactive SnRK1.1K48M treatment. In the third lane of each blot, the retardation of the band shift was reduced with λ phosphatase treatment. At least three independent experiments were performed with similar results.