Figure 1. Different inflammatory responses to LPS/GalN-, HFD/LPS- and CCl4-induced liver injury in mice with impaired macrophage autophagy.
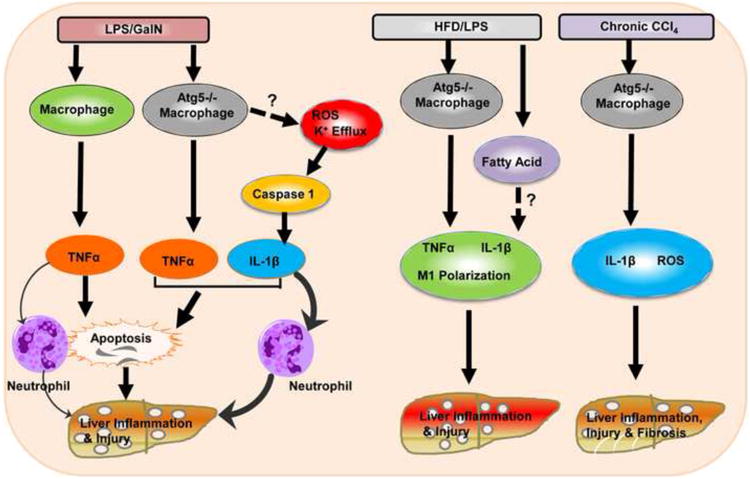
In wild type mice, LPS/GalN activates macrophages to generate TNF-α which induces hepatocyte apoptosis and neutrophil infiltration to the liver resulting in liver injury. In macrophage-specific Atg5 knockout mice, in addition to TNF-α production, LPS/GalN also increased caspase-1-mediated IL-1β production likely via increased ROS and K+ efflux due to the lack of autophagy in macrophages. IL-1β amplifies TNF-α-induced death signals, enhances LPS/GalN-induced apoptosis and induces more neutrophil activation resulting in exacerbated liver injury. In HFD/LPS-challenged macrophage-specific Atg5 knockout mice, in addition to the increased production of TNF-α and IL-1β, autophagy-deficient macrophages also undergo polarization to become more M1 macrophages likely due to the accumulated fatty acids and neutral lipids in the steatotic environment. Macrophage-specific Atg5 knockout mice also have increased IL-1β and ROS production and liver fibrosis after CCl4 administration “?” and dotted-line arrows indicate molecular events that were not investigated in this study.
