|
| |
| Drug name (generic) | Empagliflozin |
|
| |
| Phase | IV |
|
| |
| Indication | Indicated as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes mellitus |
|
| |
| Pharmacology description/method of action | Inhibits the renal sodium glucose co-transporter 2 isoform thereby enhancing urinary glucose excretion |
|
| |
| Route of administration | Oral |
|
| |
| Chemical structure | D-Glucitol,1,5-anhydro-1-C-[4-chloro-3-[[4-[[(3S)-tetrahydro-3-furanyl]oxy]phenyl]methyl]phenyl]-,(1S)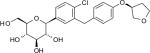
|
|
| |
| Pivotal trials | EMPA-REG MONO (NCT01177813) [4] EMPA-REG MET (NCT01159600) [5] EMPA-REG METSU (NCT01159600) [6] EMPA-REG PIO (NCT01210001) [7] EMPA-REG MDI (NCT01306214) [8] EMPA-REG H2H SU (NCT01167881) [9] |
|
| |

An official website of the United States government
Here's how you know
Official websites use .gov
A
.gov website belongs to an official
government organization in the United States.
Secure .gov websites use HTTPS
A lock (
) or https:// means you've safely
connected to the .gov website. Share sensitive
information only on official, secure websites.