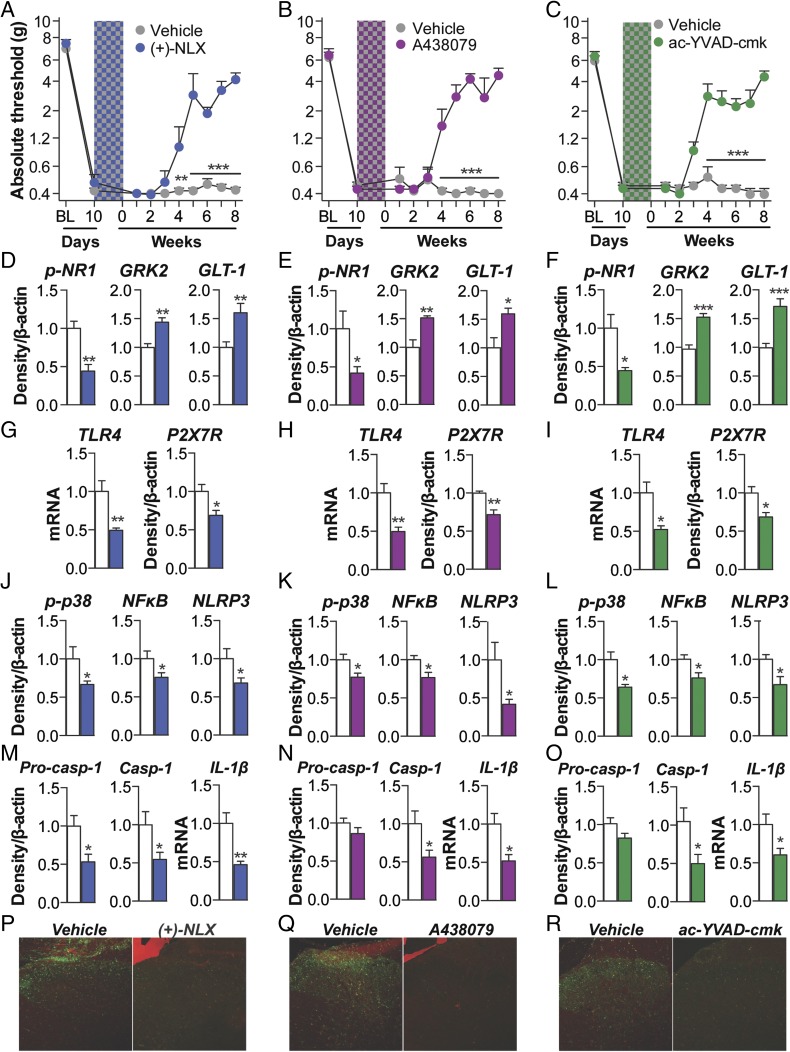
Fig. 5.
Induction of persistent sensitization is dependent on spinal cord inflammasome signaling. (A–C) The TLR4 antagonist (+)-naloxone (blue hatch; 5 d) (A), the P2X7R antagonist A438079 (purple hatch; 5 d) (B), or the caspase-1 inhibitor ac-YVAD-cmk (green hatch; 5 d) (C) was coadministered with morphine (5 d; shaded area) 10 d after CCI surgery, and absolute thresholds for mechanical allodynia were quantified in F344 rats. Ipsilateral lumbar dorsal spinal cords were collected from F344 rats that had undergone CCI surgery, 5 wk after morphine and inhibitor coadministration. (D–F) Respective levels of phospho-NR1, GRK2, and GLT-1 were quantified after treatment with (+)-naloxone (D), A438079 (E), or ac-YVAD-cmk (F). (G–I) Respective levels of P2X7R and TLR4 were quantified after treatment with (g) (+)-naloxone (G), A438079 (H), or ac-YVAD-cmk (I). (J–L) Respective levels of phospho-p38/total ERK ratio, NF-κB (p65 subunit), and NLRP3 were quantified after treatment with (+)-naloxone (J), A438079 (K), or ac-YVAD-cmk (L). (M–O) Respective levels of procaspase-1, caspase-1, and IL-1β were quantified after treatment with (+)-naloxone (M), A438079 (N), or ac-YVAD-cmk (O). (P–R) Reactive lumbar dorsal spinal microglia (Iba1+ and phospho-p38+) after treatment with (+)-naloxone (P), A438079 (Q), ac-YVAD-cmk (R), and respective vehicle controls. (S) Nlrp3 siRNA (7 d, beginning 8 d after CCI; yellow hatched bar) and morphine (5 d, beginning 10 d after CCI; shaded area) were administered, and absolute thresholds for mechanical allodynia were quantified in F344 rats. *P < 0.05; **P < 0.01; ***P < 0.001 (inhibitor vs. control). Data are presented as mean ± SEM; n = 6 or 7 per group.