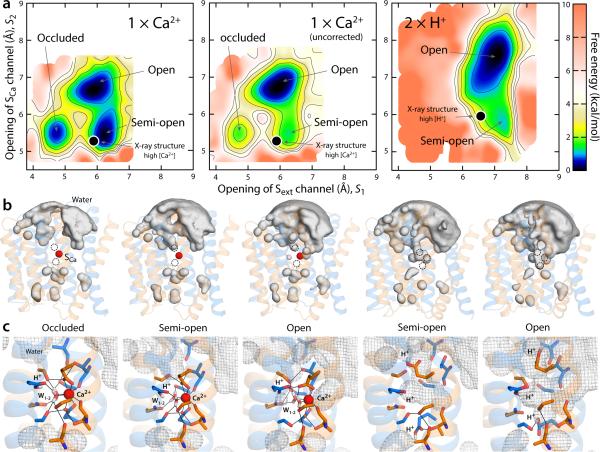
Figure 6.
Thermodynamic basis for the proposed mechanism of substrate control of the alternating-access transition of NCX. (a) Calculated free-energy landscapes for outward-facing NCX_Mj, for the Ca2+ and the fully protonated state. The free energy is plotted as in Fig. 5. For Ca2+, a map is shown in which a correction for the charge-transfer between the ion and the protein is introduced, alongside the uncorrected map (see Supplementary Notes 3-4 and Supplementary Fig. 5-6). The uncorrected map overstabilizes the open state relative to the semi-open and occluded because it also overestimates the cost of dehydration of the ion, once it is bound to the protein (this effect is negligible for Na+). Black circles map the crystal structures obtained at high Ca2+ concentration and at low pH (or high H+) reported in this study. (b) Water-density isosurfaces analogous to those in Fig. 5 are shown for each of the major conformational free-energy minima in the free-energy maps. The Ca2+ ion is shown as a red sphere; the protein is shown as in Fig. 5. (c) Close-up views of the ion-binding region in the same conformational free-energy minima. Key residues involved in Ca2+ and water coordination (W) are highlighted (sticks, black lines). The water-density maps in (b) are shown here as a grey mesh. In the occluded state with Ca2+ bound, helix TM7ab bends in the same way as in the fully occupied Na+ state, as the carbonyl of Ala206 forms a hydrogen-bonding interaction with Ser210.