Abstract
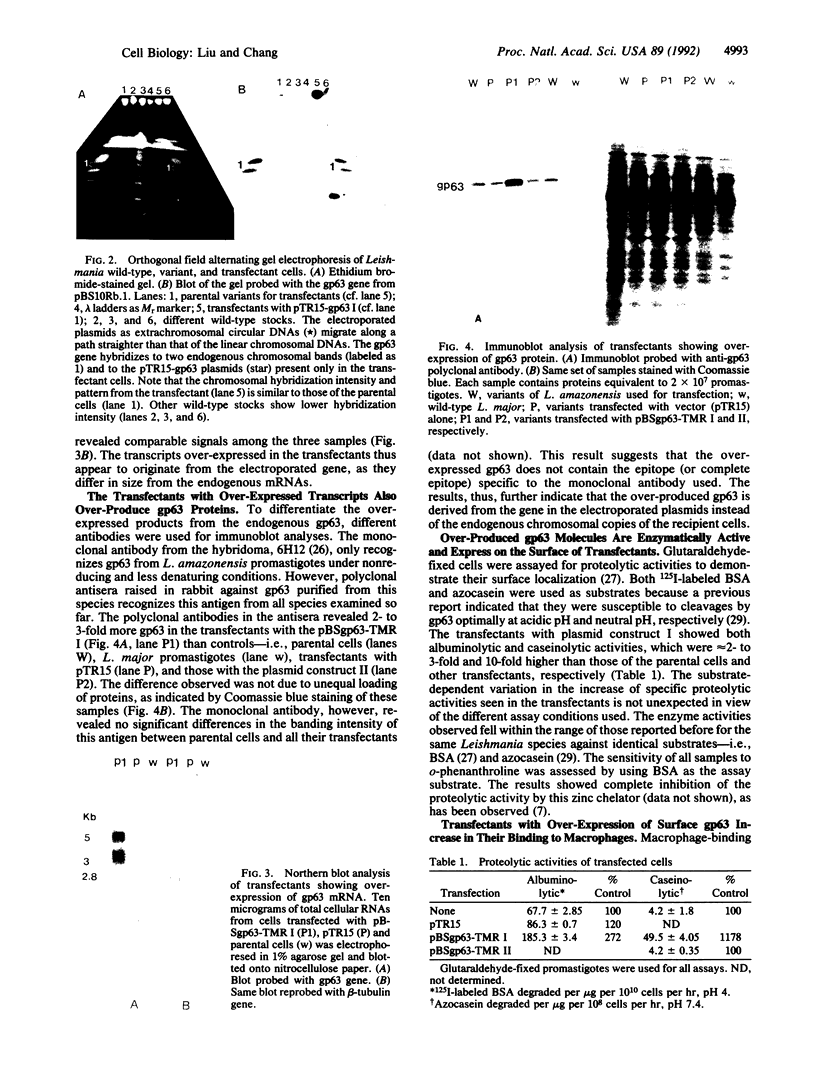
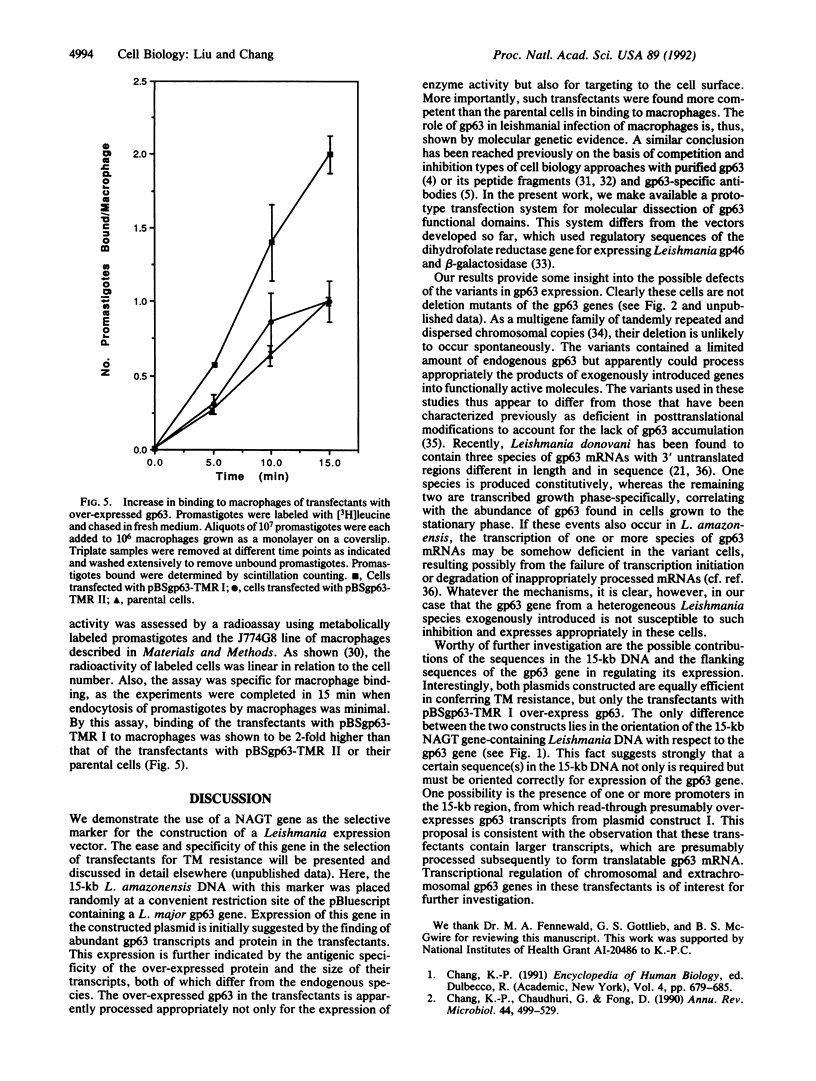
A major surface glycoprotein of 63 kDa (gp63) has been previously identified biochemically and genetically as a zinc proteinase conserved in pathogenic Leishmania spp. The functional significance of this proteinase was analyzed by genetic approaches. A 15-kilobase DNA with a tunicamycin-resistance gene from Leishmania amazonensis was ligated in two different orientations into pBluescript containing a gp63 gene from Leishmania major. These plasmid constructs were used to transfect a variant of L. amazonensis deficient in gp63 expression. Both constructs were found to confer tunicamycin resistance with equal efficiency and remained structurally unchanged in the transfectants. RNA and immunoblot analyses showed over-expression of gp63 in the transfectants with one of the two plasmids constructed. The over-produced products were enzymatically active and expressed on the cell surface. Significantly, the transfectants with over-expressed gp63 increased by 2-fold over controls in their binding to macrophages. Evidence presented thus indicates that the gp63 gene constructed in the plasmid as described and introduced exogenously expresses in the gp63-deficient variants and that the expressed products are functionally active.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bouvier J., Bordier C., Vogel H., Reichelt R., Etges R. Characterization of the promastigote surface protease of Leishmania as a membrane-bound zinc endopeptidase. Mol Biochem Parasitol. 1989 Dec;37(2):235–245. doi: 10.1016/0166-6851(89)90155-2. [DOI] [PubMed] [Google Scholar]
- Bouvier J., Etges R. J., Bordier C. Identification and purification of membrane and soluble forms of the major surface protein of Leishmania promastigotes. J Biol Chem. 1985 Dec 15;260(29):15504–15509. [PubMed] [Google Scholar]
- Bouvier J., Schneider P., Etges R., Bordier C. Peptide substrate specificity of the membrane-bound metalloprotease of Leishmania. Biochemistry. 1990 Oct 30;29(43):10113–10119. doi: 10.1021/bi00495a015. [DOI] [PubMed] [Google Scholar]
- Button L. L., McMaster W. R. Molecular cloning of the major surface antigen of leishmania. J Exp Med. 1988 Feb 1;167(2):724–729. doi: 10.1084/jem.167.2.724. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chang C. S., Chang K. P. Monoclonal antibody affinity purification of a Leishmania membrane glycoprotein and its inhibition of leishmania-macrophage binding. Proc Natl Acad Sci U S A. 1986 Jan;83(1):100–104. doi: 10.1073/pnas.83.1.100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chang K. P., Chaudhuri G., Fong D. Molecular determinants of Leishmania virulence. Annu Rev Microbiol. 1990;44:499–529. doi: 10.1146/annurev.mi.44.100190.002435. [DOI] [PubMed] [Google Scholar]
- Chang K. P. Leishmania donovani-macrophage binding mediated by surface glycoproteins/antigens: characterization in vitro by a radioisotopic assay. Mol Biochem Parasitol. 1981 Nov;4(1-2):67–76. doi: 10.1016/0166-6851(81)90030-x. [DOI] [PubMed] [Google Scholar]
- Chaudhuri G., Chang K. P. Acid protease activity of a major surface membrane glycoprotein (gp63) from Leishmania mexicana promastigotes. Mol Biochem Parasitol. 1988 Jan 1;27(1):43–52. doi: 10.1016/0166-6851(88)90023-0. [DOI] [PubMed] [Google Scholar]
- Chaudhuri G., Chaudhuri M., Pan A., Chang K. P. Surface acid proteinase (gp63) of Leishmania mexicana. A metalloenzyme capable of protecting liposome-encapsulated proteins from phagolysosomal degradation by macrophages. J Biol Chem. 1989 May 5;264(13):7483–7489. [PubMed] [Google Scholar]
- Detke S., Chaudhuri G., Kink J. A., Chang K. P. DNA amplification in tunicamycin-resistant Leishmania mexicana. Multicopies of a single 63-kilobase supercoiled molecule and their expression. J Biol Chem. 1988 Mar 5;263(7):3418–3424. [PubMed] [Google Scholar]
- Etges R., Bouvier J., Bordier C. The major surface protein of Leishmania promastigotes is a protease. J Biol Chem. 1986 Jul 15;261(20):9098–9101. [PubMed] [Google Scholar]
- Etges R., Bouvier J., Bordier C. The major surface protein of Leishmania promastigotes is anchored in the membrane by a myristic acid-labeled phospholipid. EMBO J. 1986 Mar;5(3):597–601. doi: 10.1002/j.1460-2075.1986.tb04252.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fong D., Chang K. P. Surface antigenic change during differentiation of a parasitic protozoan, Leishmania mexicana: Identification by monoclonal antibodies. Proc Natl Acad Sci U S A. 1982 Dec;79(23):7366–7370. doi: 10.1073/pnas.79.23.7366. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hightower R. C., Ruiz-Perez L. M., Wong M. L., Santi D. V. Extrachromosomal elements in the lower eukaryote Leishmania. J Biol Chem. 1988 Nov 15;263(32):16970–16976. [PubMed] [Google Scholar]
- Kapler G. M., Coburn C. M., Beverley S. M. Stable transfection of the human parasite Leishmania major delineates a 30-kilobase region sufficient for extrachromosomal replication and expression. Mol Cell Biol. 1990 Mar;10(3):1084–1094. doi: 10.1128/mcb.10.3.1084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Katakura K., Peng Y., Pithawalla R., Detke S., Chang K. P. Tunicamycin-resistant variants from five species of Leishmania contain amplified DNA in extrachromosomal circles of different sizes with a transcriptionally active homologous region. Mol Biochem Parasitol. 1991 Feb;44(2):233–243. doi: 10.1016/0166-6851(91)90009-u. [DOI] [PubMed] [Google Scholar]
- Kink J. A., Chang K. P. N-glycosylation as a biochemical basis for virulence in Leishmania mexicana amazonensis. Mol Biochem Parasitol. 1988 Jan 15;27(2-3):181–190. doi: 10.1016/0166-6851(88)90037-0. [DOI] [PubMed] [Google Scholar]
- Laban A., Tobin J. F., Curotto de Lafaille M. A., Wirth D. F. Stable expression of the bacterial neor gene in Leishmania enriettii. Nature. 1990 Feb 8;343(6258):572–574. doi: 10.1038/343572a0. [DOI] [PubMed] [Google Scholar]
- Laban A., Wirth D. F. Transfection of Leishmania enriettii and expression of chloramphenicol acetyltransferase gene. Proc Natl Acad Sci U S A. 1989 Dec;86(23):9119–9123. doi: 10.1073/pnas.86.23.9119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LeBowitz J. H., Coburn C. M., McMahon-Pratt D., Beverley S. M. Development of a stable Leishmania expression vector and application to the study of parasite surface antigen genes. Proc Natl Acad Sci U S A. 1990 Dec;87(24):9736–9740. doi: 10.1073/pnas.87.24.9736. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miller R. A., Reed S. G., Parsons M. Leishmania gp63 molecule implicated in cellular adhesion lacks an Arg-Gly-Asp sequence. Mol Biochem Parasitol. 1990 Mar;39(2):267–274. doi: 10.1016/0166-6851(90)90065-t. [DOI] [PubMed] [Google Scholar]
- Miller R. L., Wang A. L., Wang C. C. Identification of Giardia lamblia isolates susceptible and resistant to infection by the double-stranded RNA virus. Exp Parasitol. 1988 Jun;66(1):118–123. doi: 10.1016/0014-4894(88)90056-2. [DOI] [PubMed] [Google Scholar]
- Olafson R. W., Thomas J. R., Ferguson M. A., Dwek R. A., Chaudhuri M., Chang K. P., Rademacher T. W. Structures of the N-linked oligosaccharides of Gp63, the major surface glycoprotein, from Leishmania mexicana amazonensis. J Biol Chem. 1990 Jul 25;265(21):12240–12247. [PubMed] [Google Scholar]
- Ramamoorthy R., Donelson J. E., Paetz K. E., Maybodi M., Roberts S. C., Wilson M. E. Three distinct RNAs for the surface protease gp63 are differentially expressed during development of Leishmania donovani chagasi promastigotes to an infectious form. J Biol Chem. 1992 Jan 25;267(3):1888–1895. [PubMed] [Google Scholar]
- Russell D. G., Talamas-Rohana P., Zelechowski J. Antibodies raised against synthetic peptides from the Arg-Gly-Asp-containing region of the Leishmania surface protein gp63 cross-react with human C3 and interfere with gp63-mediated binding to macrophages. Infect Immun. 1989 Feb;57(2):630–632. doi: 10.1128/iai.57.2.630-632.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Russell D. G., Wilhelm H. The involvement of the major surface glycoprotein (gp63) of Leishmania promastigotes in attachment to macrophages. J Immunol. 1986 Apr 1;136(7):2613–2620. [PubMed] [Google Scholar]
- Russell D. G., Wright S. D. Complement receptor type 3 (CR3) binds to an Arg-Gly-Asp-containing region of the major surface glycoprotein, gp63, of Leishmania promastigotes. J Exp Med. 1988 Jul 1;168(1):279–292. doi: 10.1084/jem.168.1.279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schneider P., Ferguson M. A., McConville M. J., Mehlert A., Homans S. W., Bordier C. Structure of the glycosyl-phosphatidylinositol membrane anchor of the Leishmania major promastigote surface protease. J Biol Chem. 1990 Oct 5;265(28):16955–16964. [PubMed] [Google Scholar]
- Tzinia A. K., Soteriadou K. P. Substrate-dependent pH optima of gp63 purified from seven strains of Leishmania. Mol Biochem Parasitol. 1991 Jul;47(1):83–89. doi: 10.1016/0166-6851(91)90150-5. [DOI] [PubMed] [Google Scholar]
- Webb J. R., Button L. L., McMaster W. R. Heterogeneity of the genes encoding the major surface glycoprotein of Leishmania donovani. Mol Biochem Parasitol. 1991 Oct;48(2):173–184. doi: 10.1016/0166-6851(91)90113-k. [DOI] [PubMed] [Google Scholar]
- Wilson M. E., Hardin K. K., Donelson J. E. Expression of the major surface glycoprotein of Leishmania donovani chagasi in virulent and attenuated promastigotes. J Immunol. 1989 Jul 15;143(2):678–684. [PubMed] [Google Scholar]
- Wilson M. E., Hardin K. K. The major concanavalin A-binding surface glycoprotein of Leishmania donovani chagasi promastigotes is involved in attachment to human macrophages. J Immunol. 1988 Jul 1;141(1):265–272. [PubMed] [Google Scholar]