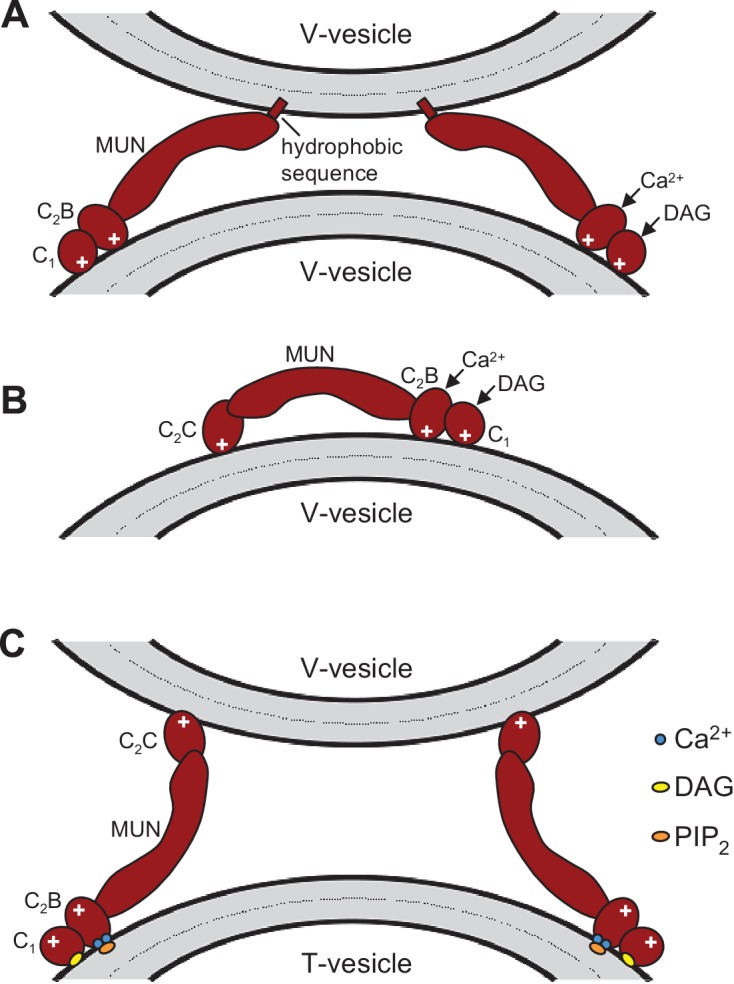
These models serve in part as a basis for the model proposed in
Figure 7 and provide a rationalization for the liposomes clustering activities observed for C
1C
2BMUN and C
1C
2BMUNC
2C. However, it is important to note that there are multiple potential explanations for these activities. The findings that PS is a major determinant of vesicle clustering by C
1C
2BMUN (
Figure 2) without requiring Ca
2+ (
Figure 2—figure supplement 1), but C
1C
2BMUNC
2C requires Ca
2+ to cluster V-liposomes (
Figure 6A,B), suggest that there are multiple membrane binding sites in these large protein fragments that can cooperate in cis to interact with a single membrane or in trans to bind to two membranes. Indeed, the MUN, C
1 and C
2B domains contain several positive patches, the C
1 domain binds to DAG, and the C
2B domain binds to PIP
2 weakly in the absence of Ca
2+ and more strongly in the presence of Ca
2+ (
Shen et al., 2005;
Shin et al., 2010;
Yang et al., 2015). The C
2C domain is likely to have at least one lipid-binding site with moderate affinity that explains the stronger overall liposome clustering activity of C
1C
2BMUNC
2C compared to C
1C
2BMUN (
Figure 6—figure supplement 1) (see discussion). Moreover, the sequence spanning residues 1517–1531 at the C-terminus of C
1C
2BMUN does not form part of the MUN domain structure (
Yang et al. 2015) and contains a highly hydrophobic sequence that could bind to membranes, but this sequence may become structured due to the presence of the C
2C domain in C
1C
2BMUNC
2C, which could render it unable to bind membranes. We speculate that this hydrophobic sequence together with positive patches in the C
1-C
2B region underlie the liposome clustering activity of Ca
2+-free C
1C
2BMUN (
A), while in C
1C
2BMUNC
2C the C
2C domain provides a PS-binding site that cooperates with the C
1-C
2B region to favor binding in cis to the same membrane (
B). Ca
2+ binding to the C
2B domain may favor membrane binding of C
1C
2BMUNC
2C in a different orientation that facilitates interaction of the C
2C domain in trans with another membrane, which would explain why Ca
2+-bound C
1C
2BMUNC
2C can bridge V-liposomes; this orientation could also be favored by binding of the C
2B domain to PIP
2 and of the C
1 domain to DAG in T-liposomes (
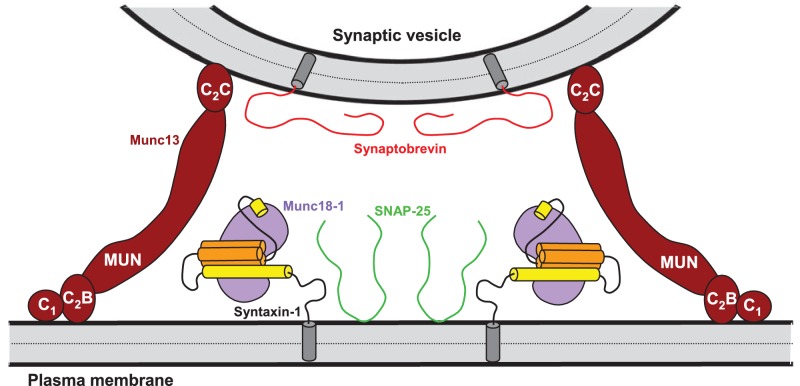
C), leading to the overall notion that the C
1-C
2B region binds to the plasma membrane and the C
2C domain to synaptic vesicle membrane (
Figure 7). Extensive studies will be required to test this and other plausible models compatible with the liposome clustering data.