Abstract
Abstract Avian influenza A viruses exhibit a strong preference for using α2,3‐linked sialic acid as a receptor. Until recently, the presumed lack of this receptor in human airways was believed to constitute an efficient barrier to avian influenza A virus infection of humans. Recent zoonotic outbreaks of avian influenza A virus have triggered researchers to analyse tissue distribution of sialic acid in further detail. Here, we review and extend the current knowledge about sialic acid distribution in human tissues, and discuss viruses with ocular tropism and their preference for α2,3‐linked sialic acid.
Keywords: eye, influenza, receptor, sialic acid, tropism
Introduction
Influenza viruses, members of the family Orthomyxoviridae, are enveloped viruses with segmented, single‐stranded, negative‐sense RNA packaged in virions as ribonucleoproteins. Influenza viruses are further classified into three genera, A, B and C, on the basis of the antigenic diversity of virion proteins such as the matrix protein (M1) and nucleocapsid protein (NP). Further classification is based on the haemagglutinin (HA) and neuraminidase (NA) glycoproteins protruding from the viral envelope of influenza A and B viruses. Human influenza viruses mainly cause acute respiratory disease with high incidence and mortality, usually as epidemics but sometimes as worldwide pandemics. To date, 16 different influenza A virus HA subtypes (H1–H16) and nine different NA subtypes (N1–N9) have been identified in wild waterfowl, which are the major reservoir of all influenza A viruses. All mammalian influenza viruses are believed to originate from avian influenza A viruses, and only three subtypes of influenza A virus, H1N1, H2N2 and H3N2, have adapted to humans since 1918. 1 The causative viruses of the 1957 Asian (H2N2) and 1968 Hong Kong (H3N2) pandemics were found to be reassortants of human and avian influenza A viruses, 2 , 3 whereas the 1918 Spanish (H1N1) influenza A virus was likely to be entirely of avian origin but adapted to humans. 4 When H1N1 reappeared in 1977, H3N2 did not disappear but continued to circulate; therefore, currently, together with their H1N2 reassortant, three subtypes of influenza A virus are associated with epidemics. The ongoing zoonotic outbreaks of H5N1 avian influenza A virus have raised concerns that the situation may develop into a pandemic comparable in magnitude with the Spanish influenza virus pandemic.
Recently it become evident that other pathogenic viruses, including adenovirus type 37 (Ad37, family Adenoviridae), enterovirus type 70 (EV70, family Picornaviridae) and Newcastle disease virus (NDV, family Paramyxoviridae), all with a pronounced tropism for the human eye, share receptor preference with avian influenza A viruses for glycans terminating with α2,3‐linked sialic acid (SAα2,3), 5 , 6 , 7 , 8 , 9 which is distinct from the SAα2,6‐terminated receptor preferred by human influenza A viruses. Here, we review differences in sialic acid preferences of viruses that cause disease in humans and in the distribution of different sialic acids in tissues. Given the ocular tropism of Ad37, EV70 and NDV, and their preference for SAα2,3, we also discuss the human eye as a potential portal of entry for viruses such as the avian influenza A viruses.
Sialic acids, what and where?
Vertebrate cell surface glycans consist mainly of five‐ and six‐carbon monosaccharides. One remarkable exception is the family of sialic acids, whose members have nine‐carbon backbones. 10 Other important features of sialic acids are: (i) the high level of diversity – currently the family comprises over 50 different members, with neuraminic acid considered as the prototype, and includes important variants such as N‐acetylneuraminic acid (Neu5Ac) and N‐glycolylneuraminic acid (Neu5Gc) (Figure 1), (ii) their presence mainly in higher animals and in certain bacteria, and (iii) their location, typically at the termini of glycan chains, and usually linked to galactose via α2,3‐ or α2,6‐glycosidic bonds, or to sialic acid via α2,8‐glycosidic bonds. Not surprisingly, their expression on the surface of cells makes sialic acids easily accessible as attachment molecules for a wide variety of microbial pathogens, including many viruses.
Figure 1.
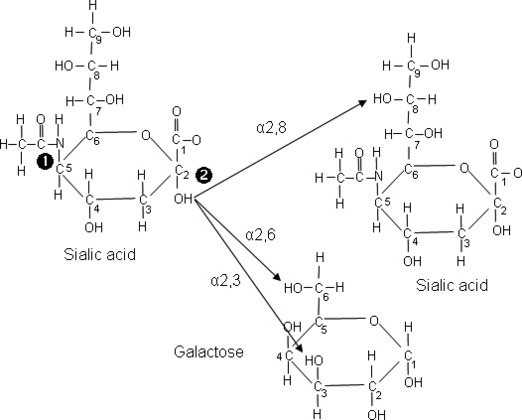
Sialic acid structure and glycosidic bonds to neighbouring saccharides. 1 corresponds to the site of N‐linked acylation (glycolyl or acetyl), and 2 corresponds to the site of α2,3‐, α2,6‐ and α2,8‐glycosidic bonds between sialic acid and galactose or a neighbouring sialic acid sacharride.
Until recently, little was known about the distribution of sialic acid in humans, although the expression of SAα2,6 on the surface of tracheal epithelial cells was demonstrated more than 15 years ago, which, at that time 11 , 12 correlated perfectly with the known receptor preference, tropism and host range of influenza A viruses. Thus, human influenza A viruses bind preferentially to cell surface SAα2,6 and replicate in the respiratory tract, whereas avian influenza A viruses bind preferentially to SAα2,3 and only recently were reported to target the human airway. In this context, it should be noted that humans, but not primates, express SAα2,6 on cells in the upper respiratory tract, 13 which is in agreement with the restricted replication of human influenza A virus in airways of chimpanzees. 14 Another important observation is that Neu5Gc is a frequent constituent of glycans in most birds and mammals, but the gene encoding the hydroxylase enzyme necessary for the conversion of Neu5Ac to Neu5Gc was inactivated in our human ancestors, but not in other primates, approximately 2·7 Ma, 15 possibly as a response to a microbial threat. 16 Some avian influenza A viruses replicate only or mainly in cells that express Neu5Gc. 17 Along with the current attention paid to sialic acid‐binding viruses causing disease in humans, interest in sialic acid biology and its tissue distribution has intensified.
Recent research has more carefully documented the distribution of SAα2,3 and SAα2,6 in human tissues and more particularly in human airways. SAα2,3 is found on alveolar cells, whereas SAα2,6, with a few exceptions, is predominantly found on non‐alveolar cells (Table 1). This correlates reasonably well with the predominant manifestations caused by human influenza A virus infection (rhinitis, pharyngitis, tracheobronchitis, bronchitis and bronchiolitis), indicating replication in the respiratory tract. 18 Importantly, the distribution of SAα2,3 and SAα2,6 also correlates with reports of lower respiratory tract symptoms and the difficulty in detecting H5N1 in nasopharyngeal samples, 19 and with the replication of H5N1 in cells lining the alveoli. 20 Specifically, SAα2,6 predominates on M cells in nasopharyngeal lymphoid tissue 21 and on epithelial cells in the nasal mucosa, 22 paranasal sinuses, 22 pharynx, 22 larynx, 23 trachea, 11 , 12 , 13 , 22 , 23 , 24 trachea/bronchi, 25 , 26 bronchi 22 , 24 , 27 and in bronchioles, 13 , 22 but not on alveolar cells. 24 , 28 Goblet cells in the larynx, 23 trachea 13 , 23 and bronchi 13 , 24 may also contain SAα2,6. Conversely, SAα2,3 is primarily found on alveolar cells 22 , 24 , 28 and on bronchiolar epithelial cells at the junction between bronchioles and alveoli, 22 and has been found on bronchial 27 and tracheal 24 epithelial cells. Epithelial tracheobronchial cells 25 , 26 and epithelial cells in the nasal mucosa 22 may also express SAα2,3, albeit at lower levels. Goblet cells in the larynx, 23 trachea 11 , 12 , 13 , 23 and bronchial 13 epithelium also contain SAα2,3. Nasopharyngeal and bronchial epithelial cells abundantly express SAα2,3 linked via galactose to N‐acetylglucosamine (SAα2,3Galβ1,4GlcNAc), but not SAα2,3 linked via galactose to N‐acetylgalactosamine (SAα2,3Galβ1,3GalNAc). 28 Both trisaccharides, however, are abundantly expressed on alveolar pneumocytes.
Table 1.
Distribution of SAα2,3 or SAα2,6 on cells in human airways and eyes*
| Tissue | Glycan | Reference |
|---|---|---|
| Human airways | ||
| M cells in nasopharyngeal lymphoid tissue | SAα2,6 | 21 |
| Nasal mucosa | SAα2,6 | 22 |
| Paranasal sinuses | SAα2,6 | 22 |
| Pharynx | SAα2,6 | 22 |
| Larynx | SAα2,6 | 23 |
| Trachea | SAα2,6 | 11, 12, 13, 22, 23, 24 |
| Trachea/bronchi | SAα2,6 | 25, 26 |
| Bronchi | SAα2,6 | 22, 24, 27 |
| Bronchioles | SAα2,6 | 13, 22 |
| Laryngeal, tracheal and bronchial** goblet cells | SAα2,6 | 13, 23, 24 |
| Alveoli | SAα2,3 | 22, 24, 28 |
| Alveoli | SAα2,3Gal β1,4GlcNAc | 28 |
| SAα2,3Gal β1,3GalNAc | 28 | |
| Bronchiolar cells junctioning bronchiole and alveoli | SAα2,3 | 22 |
| Bronchi | SAα2,3 | 27 |
| Bronchi | SAα2,3Gal β1,4GlcNAc | 28 |
| Trachea/bronchi** | SAα2,3 | 25, 26 |
| Trachea | SAα2,3 | 24 |
| Nasopharynx | SAα2,3Gal β1,4GlcNAc | 28 |
| Nasal mucosa** | SAα2,3 | 22 |
| Laryngeal, tracheal and bronchial** goblet cells | SAα2,3 | 11, 12, 13, 23 |
| Human eyes | ||
| Conjunctiva | SAα2,3Galβ1,4(Fucα1,3)GlcNAc | 38 |
| SAα2,3Galβ1,3(Fucα1,4)GlcNAc | 39 | |
| Transformed conjunctival cells | SAα2,3(6) | 40, 71 |
*Cells are of epithelial origin if not otherwise stated.
**Expression is stated to be relatively low or on few cells in corresponding reference(s).
The role of ciliated versus non‐ciliated cells as targets for influenza A viruses has also been investigated. In the upper respiratory tract, SAα2,6 seems to be expressed on both ciliated and non‐ciliated cells, whereas SAα2,3 tends to be expressed mainly on ciliated cells. Specifically, SAα2,6 is found not only on non‐ciliated tracheal cells, 24 and non‐ciliated tracheobronchial cells, 25 , 26 but also on ciliated cells in the upper respiratory tract, 28 ciliated tracheal cells, 11 , 12 , 24 ciliated bronchial cells, 24 the ciliated borders of bronchial epithelial cells 13 and to a lesser extent on ciliated tracheobronchial cells. 25 , 26 SAα2,3 has been reported to be expressed on ciliated nasopharyngeal, 28 ciliated tracheal, 24 ciliated tracheobronchial cells 25 , 26 and ciliated bronchial cells, 24 , 28 and also on non‐ciliated cuboidal bronchial cells at the junctions between bronchioles and alveoli. 22 Thus, the distribution of SAα2,3 and SAα2,6 in human cells and tissues (Table 1) generally parallels the cell and tissue tropism of influenza A viruses, 22 , 25 , 26 , 28 , 29 and emphasizes sialic acid’s role as a critical determinant of influenza A virus tropism.
SAα2,3‐binding viruses and ocular tropism
With few exeptions, sialic acid‐binding viruses isolated from humans exhibit a clear preference for two organs, the respiratory tract and the eye (Table 2). SAα2,6‐binding viruses, such as human influenza A and B viruses, coronavirus OC43, polyomavirus JC and human parainfluenza virus 3, are more frequently isolated from the respiratory tract than from the eye. The human eye, on the other hand, apparently constitutes a very efficient replication site for at least three unrelated viruses with specificity for SAα2,3. Ad37 was isolated for the first time in 1976 30 and has been demonstrated to be one of the most frequent agents of epidemic keratoconjunctivitis (EKC). 31 The large amounts of Ad37 detected in eye swabs from EKC patients 32 are consistent with the observations that Ad37 is readily transmitted via direct or indirect contact 33 and that EKC is common in densely populated areas of the world. 34 EV70 was first isolated during a pandemic of a highly contagious form of acute haemorrhagic conjunctivitis (AHC) that originated in Ghana in 1969 35 and subsequently spread through Africa to Asia. EV70 was also responsible for a second pandemic of AHC in the early 1980s 36 that included both the eastern and western hemispheres, and for numerous regional outbreaks. Like Ad37, EV70 also uses SAα2,3 as a cellular receptor. 7 The third virus that binds to SAα2,3 is NDV, 9 a virus of domestic fowl that has been associated with several zoonotic outbreaks of conjunctivitis among poultry and kitchen workers and with case reports of laboratory infection. 37 The ability of these three viruses (Ad37, EV70 and NDV) to utilize the eye as a primary site of replication further emphasizes the potential role of the eye as a gateway for infections caused by other viruses that use SAα2,3 as a receptor. Therefore, it is important to highlight the potential of ocular SA, in general, as a receptor and of ocular SAα2,3 in particular, as a primary target of previously unrecognized importance for human infection by other viruses, including avian influenza A virus.
Table 2.
Viruses with specificity for SAα2,3 or SAα2,6 and their tropism in humans
| Virus | Specificity | Tropism in humans | References |
|---|---|---|---|
| Adenoviridae | |||
| Ad37 | SAα2,3 | Ocular | 6, 31 |
| Picornaviridae | |||
| EV70 | SAα2,3 | Ocular (respiratory, CNS) | 7, 36, 45 |
| CVA24v | SAα2,3/6 | Ocular, respiratory | 40 |
| Reoviridae | |||
| Reo1 | SAα2,3 | Intestinal (ocular, CNS) | 72 |
| Paramyxoviridae | |||
| PIV1 | SA2,3 | Respiratory | 27 |
| PIV3 | SAα2,3/6 | Respiratory(CNS) | 73, 74 |
| PIV5 | SAα2,3 | Respiratory | 75 |
| NDV | SAα2,3 | Ocular (CNS) | 9 |
| Orthomyxoviridae | |||
| Avian influenza A H7 | SAα2,3 | Ocular (respiratory) | 56, 76 |
| Avian influenza A H5 | SAα2,3 | Respiratory, ocular (CNS) | 42, 44, 58, 68 |
| Human influenza A, B | SAα2,6 | Respiratory (ocular, CNS) | 8, 18, 77 |
| Coronaviridae | |||
| OC43 | SAα2,6 | Respiratory (CNS) | 78, 79 |
| Polyomaviridae | |||
| JC | SAα2,6 | Respiratory (lymphoid, renal, CNS) | 80, 81 |
| BK | SAα2,3 | Respiratory, renal (ocular, CNS) | 80, 82, 83 |
Ad37: adenovirus 37; EV70: enterovirus type 70; CVA24v: coxsackievirus A 24 variant; Reo1: reovirus 1; PIV1,3,5: parainfluenzavirus 1, 3, 5; NDV: Newcastle disease virus; OC43, JC, BK: strain OC43, strain JC and strain BK respectively.
It should also be noted that the human eye is an organ in which many other viruses replicate. For example, herpes simplex virus, measles virus, respiratory syncytial virus, echoviruses and species B adenoviruses have all been isolated from the human eye, but use cellular receptors other than SA; therefore, they are not discussed further in this review. For other SAα2,3‐binding viruses with respiratory tropism, such as human parainfluenza viruses and polyomavirus BK, it will also be of interest to identify the cells in which these viruses replicate, and to determine if these cells express SAα2,3.
Sialic acid in the human eye
Relatively little is known about the levels and distribution of sialic acid on cells in the human eye. Sialyl Lewis X (sLex; SAα2,3Galβ1,4(Fucα1,3)GlcNAc) and Sialyl Lewis A (sLea; SAα2,3Galβ1,3(Fucα1,4)GlcNAc) have both been identified on conjunctival epithelial cells using antibodies, 38 , 39 and both SAα2,3 and SAα2,6 have been detected on transformed, normal human conjunctival epithelial cells (NHC) using lectins. 40 Thus, the presence of SAα2,3 on conjunctival cells is in agreement with the ocular tropism of SAα2,3‐binding viruses such as Ad37, EV70 and NDV. This also suggest that human conjunctival cells are susceptible to both human and avian influenza A viruses, which may be of importance when considering potential sites where recombinant influenza A viruses with pandemic potential may be generated. The role of a ‘mixing vessel’ has previously been allotted to the pig, 41 an animal known to express both SAα2,3 and SAα2,6 in the respiratory tract and susceptible to both human and avian influenza A viruses. The presence of both SAα2,3 and SAα2,6 in human conjunctival cells indicates that these cells should not be ignored as candidate mixing vessels.
Avian influenza A virus infections in humans
A major zoonotic outbreak caused by avian influenza A virus occurred in 1997, when at least 18 people in Hong Kong were infected by the H5N1 virus and six of these 18 individuals died, mainly due to respiratory failure. 42 Since 2003, recurrent outbreaks of zoonotic H5N1 virus have been reported from Asia and Africa, where this subtype may now be enzootic. 43 Recently, it has become evident that not only human influenza A virus but also avian influenza A virus infection may give rise to serious CNS manifestations. 44 In this context, it is interesting that several viruses that replicate in the human eye (including EV70 and NDV) have also been associated with CNS complications, 45 suggesting that the CNS is an accessible secondary target for SAα2,3‐binding viruses that replicate in the human eye.
Is the eye a potential portal of entry for avian influenza A viruses?
Multiple reports describe the eye as an intial site of replication in humans for measles virus, 46 respiratory syncytial virus, 47 and CVA24v, 48 and in experimentally infected animals for ebola virus, 49 human influenza A virus, 50 and importantly, avian influenza A virus including H5N1, 51 followed by further transmission to non‐ocular sites, including airways. Conjunctivitis caused by H7 subtypes of avian influenza A virus has been reported sporadically over the years, 52 , 53 , 54 , 55 and both before and after the zoonotic outbreak of H7N7 in the Netherlands in 2003. 56 During this outbreak, one person died of respiratory failure; however, conjunctivitis was the major manifestation observed. It was recently demonstrated that virus isolated from this outbreak was capable of direct binding to human conjunctival tissue. 57 H7N7 influenza virus replication in the eyes of patients was high, and similar to the level of replication of cocirculating human influenza H3N2 virus in patients with respiratory tract infection. 56 While it seems clear that the human eye is the initial and main site of replication for some H7 viruses, this is not obvious for other avian influenza A viruses. While avian H5N1 viruses replicate in the respiratory tract, mainly in alveolar cells 20 that express SAα2,3, 22 the potential for H5N1 influenza A virus replication in the eye needs further attention. H5N1 was reported to be associated with conjunctivitis during the outbreaks in Hong Kong in 1997 42 and in Turkey in 2005–2006. 58 Importantly, 14/37 (37%) of Egyptian patients with H5N1 exhibited conjunctivitis. 59 Additional information indicates that the eye may be an initial site of replication for H5N1 virus in humans. For example, a Belgian veterinarian who investigated two H5N1‐positive Thai eagles developed conjunctivitis 2 days after examination of the eagles, but no respiratory symptoms. 60 This raises the question whether H5N1 may use the human eye as a portal of entry preceeding transmission to the respiratory tract. 5 This possibility is supported by the relatively longer incubation period in humans for H5N1 avian influenza A virus (normally 4–7 days) 58 than for human H3N2 (normally 1–4 days), 18 avian H7N3 (1–3 days) 61 and avian H7N7 (1 day), 62 , 63 as well as the low frequency of virus detection and the relatively low viral load in nasopharyngeal samples from zoonotic H5N1 patients. 19 Also consistent with this idea is that one of the Turkish patients who developed respiratory symptoms of zoonotic H5N1 in the 2005–2006 outbreak presented with conjunctivitis. 58 , 64 Recent work in animal models has confirmed that avian H7 viruses that replicate in mouse eyes (but not H5 or human H3 viruses) cause lethal disease following ocular infection of mice. 65 At present, it is unclear why H5 viruses are much less frequently associated with conjunctivitis in humans than are H7 viruses. Glycan arrays can be used to identify sialylated glycans recognized by influenza virus and other viruses. 66 , 67 , 68 A recent study using glycan arrays 66 showed differences in the sialic acid‐binding specificity of influenza virus H7 isolates. European and North American isolates associated with conjunctivitis exhibited a strong preference for binding SAα2,3; however, the pattern of binding for the European isolates more closely paralleled binding of classic avain influenza viruses, while the North American isolates showed reduced binding to glycans containing SAα2,3 and increased binding to glycans containing SAα2,6. Virus binding was skewed even more towards glycans containing SAα2,6 for North American H7 isolates that were not associated with conjunctivitis. Glycan array analysis will be an important tool for unravelling the complex inter‐relationship between sialic acid‐binding specificity and influenza virus tropism and pathogenicity. However, for identification of the specific sialic acid‐containing cellular receptors used by influenza A viruses, additional experimental approaches are required.
The possibility exists that influenza virus replication in the human eye may occur asymptomatically. It has been suggested that asymptomatic replication occurred frequently during the zoonotic outbreak of H7N7 in the Netherlands in 2003 69 and during the zoonotic outbreaks of H5N2 in Japan in 2005 (http://www.mhlw.go.jp/english/topics/influenza/avian02.html) and H5N1 in South Korea in 2006 (http://www.promedmail.org/pls/otn/f?p=2400:1202:857450566246460::NO::F2400_P1202_CHECK_DISPLAY,F2400_P1202_PUB_MAIL_ID:X,34431). Whether or not patients or their relatives were asked about a history of conjunctivitis during outbreaks of H5N1 has not been reported, and there is no evidence that ocular samples from infected patients or from individuals exposed to H5N1 were tested for the presence of virus. Moreover, human‐to‐human transmission of avian H5N1 influenza A virus is inefficient, 70 which is compatible with virus replication that is limited to the lower respiratory tract, 20 and there are no reported cases of human‐to‐human transmission by small‐particle aerosols. 70 However, influenza infection in the human eye represents a potential source for transmission between humans, but not necessarily a signal of an imminent pandemic. Mild or subclinical ocular infections may explain the prolonged incubation time before respiratory symptoms appear in zoonotic H5N1 cases, and raise the concern that unrecognized cases with mild or inapparent respiratory infection is common.
Conclusions
Not long ago, differences in the receptor specificity of human and avian influenza A viruses and a presumed lack of sialic acid receptors for avian influenza A viruses in humans was believed to constitute an almost absolute barrier to human infection by avian influenza viruses. Recent zonootic outbreaks of avian influenza A viruses triggered research on sialic acid distribution in both humans and birds, leading to evidence that this early picture was invalid. It is now clear that SAα2,3 is expressed on both alveolar and conjunctival cells in humans, which are important targets for viruses such as avian influenza A virus. From these findings it can be concluded that sialic acid is a critical determinant of influenza A virus tropism and that the human eye is a potential portal of entry for emerging sialic acid‐binding viruses. However, further investigation of sialic acid distribution and more extensive diagnostic efforts, which should include ocular sampling, are required in order to better understand the tropism and transmission of zoonotic influenza A viruses.
References
- 1. Webster RG, Bean WJ, Gorman OT, Chambers TM, Kawaoka Y. Evolution and ecology of influenza A viruses. Microbiol Rev 1992; 56:152–179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Kawaoka Y, Krauss S, Webster RG. Avian‐to‐human transmission of the PB1 gene of influenza A viruses in the 1957 and 1968 pandemics. J Virol 1989; 63:4603–4608. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Scholtissek C, Rohde W, Von Hoyningen V, Rott R. On the origin of the human influenza virus subtypes H2N2 and H3N2. Virology 1978; 87:13–20. [DOI] [PubMed] [Google Scholar]
- 4. Taubenberger JK, Reid AH, Lourens RM et al. Characterization of the 1918 influenza virus polymerase genes. Nature 2005; 437:889–893. [DOI] [PubMed] [Google Scholar]
- 5. Olofsson S, Kumlin U, Dimock K, Arnberg N. Avian influenza and sialic acid receptors: more than meets the eye? Lancet Infect Dis 2005; 5:184–188. [DOI] [PubMed] [Google Scholar]
- 6. Arnberg N, Edlund K, Kidd AH, Wadell G. Adenovirus type 37 uses sialic acid as a cellular receptor. J Virol 2000; 74:42–48. [PMC free article] [PubMed] [Google Scholar]
- 7. Nokhbeh MR, Hazra S, Alexander DA et al. Enterovirus 70 binds to different glycoconjugates containing α2,3‐linked sialic acid on different cell lines. J Virol 2005; 79:7087–7094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Rogers GN, Paulson JC. Receptor determinants of human and animal influenza virus isolates: differences in receptor specificity of the H3 hemagglutinin based on species of origin. Virology 1983; 127:361–373. [DOI] [PubMed] [Google Scholar]
- 9. Suzuki Y, Suzuki T, Matsunaga M, Matsumoto M. Gangliosides as paramyxovirus receptor structural requirement of sialo‐oligosaccharides in receptors for hemagglutinating virus of Japan (Sendai virus) and Newcastle disease virus. J Biochem 1985; 97:1189–1199. [DOI] [PubMed] [Google Scholar]
- 10. Angata T, Varki A. Chemical diversity in the sialic acids and related alpha‐keto acids: an evolutionary perspective. Chem Rev 2002; 102:439–469. [DOI] [PubMed] [Google Scholar]
- 11. Baum LG, Paulson JC. Sialyloligosaccharides of the respiratory epithelium in the selection of human influenza virus receptor specificity. Acta Histochem Suppl 1990; 40:35–38. [PubMed] [Google Scholar]
- 12. Couceiro JN, Paulson JC, Baum LG. Influenza virus strains selectively recognize sialyloligosaccharides on human respiratory epithelium; the role of the host cell in selection of hemagglutinin receptor specificity. Virus Res 1993; 29:155–165. [DOI] [PubMed] [Google Scholar]
- 13. Gagneux P, Cheriyan M, Hurtado‐Ziola N et al. Human‐specific regulation of alpha2–6 linked sialic acids. J Biol Chem 2003; 278:48245–48250. [DOI] [PubMed] [Google Scholar]
- 14. Subbarao K, Webster RG, Kawaoka Y, Murphy BR. Are there alternative avian influenza viruses for generation of stable attenuated avian‐human influenza A reassortant viruses? Virus Res 1995; 39:105–118. [DOI] [PubMed] [Google Scholar]
- 15. Chou HH, Hayakawa T, Diaz S et al. Inactivation of CMP‐N‐acetylneuraminic acid hydroxylase occurred prior to brain expansion during human evolution. Proc Natl Acad Sci 2002; 99:11736–11741. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Van Blerkom LM. Role of viruses in human evolution. Am J Phys Anthropol 2003; 37:14–46. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Ito T, Suzuki Y, Suzuki T et al. Recognition of N‐glycolylneuraminic acid linked to galactose by the alpha2,3 linkage is associated with intestinal replication of influenza A virus in ducks. J Virol 2000; 74:9300–9305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Wright PF, Neumann G, Kawaoka Y. Orthomyxoviruses; in Knipe DM, Howley PM. (ed): Fields Virology. Philadelphia: Lippincott Williams & Wilkins, 2007; 1691–1740. [Google Scholar]
- 19. Peiris JS, Yu WC, Leung CW et al. Re‐emergence of fatal human influenza A subtype H5N1 disease. Lancet 2004; 363:617–619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Uiprasertkul M, Puthavathana P, Sangsiriwut K et al. Influenza A H5N1 replication sites in humans. Emerg Infect Dis 2005; 11:1036–1041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Fujimura Y, Takeda M, Ikai H et al. The role of M cells of human nasopharyngeal lymphoid tissue in influenza virus sampling. Virchows Arch 2004; 444:36–42. [DOI] [PubMed] [Google Scholar]
- 22. Shinya K, Ebina M, Yamada S et al. Avian flu: influenza virus receptors in the human airway. Nature 2006; 440:435–436. [DOI] [PubMed] [Google Scholar]
- 23. Paulsen FP, Tschernig T, Debertin AS et al. Similarities and differences in lectin cytochemistry of laryngeal and tracheal epithelium and subepithelial seromucous glands in cases of sudden infant death and controls. Thorax 2001; 56:223–227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Ibricevic A, Pekosz A, Walter MJ et al. Influenza virus receptor specificity and cell tropism in mouse and human airway epithelial cells. J Virol 2006; 80:7469–7480. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Matrosovich MN, Matrosovich TY, Gray T, Roberts NA, Klenk H‐D. Human and avian influenza viruses target different cell types in cultures of human airway epithelium. Proc Natl Acad Sci 2004; 101:4620–4624. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Thompson CI, Barclay WS, Zambon MC, Pickles RJ. Infection of human airway epithelium by human and avian strains of influenza a virus. J Virol 2006; 80:8060–8068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Kogure T, Suzuki T, Takahashi T et al. Human trachea primary epithelial cells express both sialyl(alpha2‐3)Gal receptor for human parainfluenza virus type 1 and avian influenza viruses, and sialyl(alpha2‐6)Gal receptor for human influenza viruses. Glycoconj J 2006; 23:101–106. [DOI] [PubMed] [Google Scholar]
- 28. Nicholls JM, Chan MC, Chan WY et al. Tropism of avian influenza A (H5N1) in the upper and lower respiratory tract. Nat Med 2007; 13:147–149. [DOI] [PubMed] [Google Scholar]
- 29. Van Riel D, Munster VJ, De Wit E et al. Human and Avian Influenza Viruses Target Different Cells in the Lower Respiratory Tract of Humans and Other Mammals. Am J Pathol 2007; 171:1215–1223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. De Jong JC, Wigand R, Wadell G et al. Adenovirus 37: identification and characterization of a medically important new adenovirus type of subgroup D. J Med Virol 1981; 7:105–118. [DOI] [PubMed] [Google Scholar]
- 31. Gordon YJ, Aoki K, Kinchington PR. Adenovirus keratoconjunctivitis; in Pepose JS, Holland GN, Wilhelmus KR. (ed): Ocular Infection and Immunity. St Louis, MO: Mosby, 1996; 877–894. [Google Scholar]
- 32. Heim A, Ebnet C, Harste G, Pring‐Akerblom P. Rapid and quantitative detection of human adenovirus DNA by real‐time PCR. J Med Virol 2003; 70:228–239. [DOI] [PubMed] [Google Scholar]
- 33. Azar MJ, Dhaliwal DK, Bower KS, Kowalski RP, Gordon YJ. Possible consequences of shaking hands with your patients with epidemic keratoconjunctivitis. Am J Ophthalmol 1996; 121:711–712. [DOI] [PubMed] [Google Scholar]
- 34. Aoki K, Tagawa Y. A twenty‐one year surveillance of adenoviral conjunctivitis in Sapporo, Japan. Int Ophthalmol Clin 2002; 42:49–54. [DOI] [PubMed] [Google Scholar]
- 35. Hierholzer JC, Pallansch MA. Acute hemorrhagic conjunctivitis in the western hemisphere (1980–1987); in Uchida Y, Ishii K, Miyamura K, Yamazaki S. (ed): Acute Hemorrhagic Conjunctivitis Etiology, Epidemiology, and Clinical Manifestations. New York: S. Karger AG, 1989; 49–56. [Google Scholar]
- 36. Ishii K. Acute hemorrhagic conjunctivitis in the eastern hemisphere; in Uchida Y, Ishii K, Miyamura K, Yamazaki Y. (ed): Acute Hemorrhagic Conjunctivitis. Etiology, Epidemiology and Clinical Manifestations. New York: S. Karger AG, 1989; 11–33. [Google Scholar]
- 37. Howitt BF, K BL, Kissling RE. Presence of neutralizing antib odies of newcastle disease virus in human sera. Am J Public Health 1948; 38:1263–1272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Creuzot‐Garcher C, Guerzider V, Assem M et al. Alteration of sialyl Lewis epitope expression in pterygium. Invest Ophthalmol Vis Sci 1999; 40:1631–1636. [PubMed] [Google Scholar]
- 39. Terraciano AJ, Wang N, Schuman JS et al. Sialyl Lewis X, Lewis X, and N‐acetyllactosamine expression on normal and glaucomatous eyes. Curr Eye Res 1999; 18:73–78. [DOI] [PubMed] [Google Scholar]
- 40. Nilsson EC, Jamshidi F, Johansson SM, Oberste MS, Arnberg N. Sialic Acid is a cellular receptor for coxsackievirus a24 variant, an emerging virus with pandemic potential. J Virol 2008; 82:3061–3068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Scholtissek C, Burger H, Kistner O, Shortridge KF. The nucleoprotein as a possible major factor in determining host specificity of influenza H3N2 viruses. Virology 1985; 147:287–294. [DOI] [PubMed] [Google Scholar]
- 42. Chan PKS. Outbreak of avian influenza A (H5N1) virus infection in Hong Kong in 1997. Clin Infect Dis 2002; 34:58–64. [DOI] [PubMed] [Google Scholar]
- 43. Li KS, Guan Y, Wang J et al. Genesis of a highly pathogenic and potentially pandemic H5N1 influenza virus in eastern Asia. Nature 2004; 430:209–213. [DOI] [PubMed] [Google Scholar]
- 44. De Jong MD, Bach VC, Phan TQ et al. Fatal avian influenza A (H5N1) in a child presenting with diarrhea followed by coma. N Engl J Med 2005; 352:686–691. [DOI] [PubMed] [Google Scholar]
- 45. Wright PW, Strauss GH, Langford MP. Acute hemorrhagic conjunctivitis. Am Fam Physician 1992; 45:173–178. [PubMed] [Google Scholar]
- 46. McChesney MB, Miller MC, Rota PA et al. Experimental measles I. Pathogenesis in the normal and the immunized host. Virology 1997; 233:74–84. [DOI] [PubMed] [Google Scholar]
- 47. Bitko V, Musiyenko A, Barik S. Viral infection of the lungs through the eye. J Virol 2007; 81:783–790. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. Langford MP, Stanton GJ, Barber JC, Baron S. Early‐appearing antiviral activity in human tears during a case of picornavirus epidemic conjunctivitis. J Infect Dis 1979; 139:653–658. [DOI] [PubMed] [Google Scholar]
- 49. Jaax NK, Davis KJ, Geisbert TJ et al. Lethal experimental infection of rhesus monkeys with Ebola‐Zaire (Mayinga) virus by the oral and conjunctival route of exposure. Arch Pathol Lab Med 1996; 120:140–155. [PubMed] [Google Scholar]
- 50. Tannock GA, Paul JA, Barry RD. Immunization against influenza by the ocular route. Vaccine 1985; 3:277–280. [DOI] [PubMed] [Google Scholar]
- 51. Klopfleisch R, Werner O, Mundt E, Harder T, Teifke JP. Neurotropism of highly pathogenic avian influenza virus A/chicken/Indonesia/2003 (H5N1) in experimentally infected pigeons (Columbia livia f. domestica). Vet Pathol 2006; 43:463–470. [DOI] [PubMed] [Google Scholar]
- 52. CDC . CfDCaP update: influenza activity – United States and worldwide, 2003–04 season, and composition of the 2004–05 influenza vaccine. MMWR Morb Mortal Wkly Rep. 2004; 53:547–552. [PubMed] [Google Scholar]
- 53. Marshall M. Avian influenza H7N2, human – United Kingdom (Wales) (08) 2007. Available at: http://www.promedmail.org/pls/otn/f?p=2400:1202:857450566246460::NO::F2400_P1202_CHECK_DISPLAY,F2400_P1202_PUB_MAIL_ID:X,37732.
- 54. Nguyen‐Van‐Tam JS, Nair P, Acheson P, et al. Outbreak of low pathogenicity H7N3 avian influenza in UK, including associated case of human conjunctivitis Euro Surveill 2006; 11:2952 Available at http://www.eurosurveillance.org/ViewArticle.aspx?ArticleId=2952. [DOI] [PubMed] [Google Scholar]
- 55. Webster RG, Geraci J, Petursson G, Skirnisson K. Conjunctivitis in human beings caused by influenza A virus of seals. N Engl J Med 1981; 304:911. [DOI] [PubMed] [Google Scholar]
- 56. Fouchier RA, Schneeberger PM, Rozendaal FW et al. Avian influenza A virus (H7N7) associated with human conjunctivitis and a fatal case of acute respiratory distress syndrome. Proc Natl Acad Sci 2004; 101:1356–1361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57. Munster VJ, De Wit E, Van Riel D et al. The molecular basis of the pathogenicity of the Dutch highly pathogenic human influenza A H7N7 viruses. J Infect Dis 2007; 196:258–265. [DOI] [PubMed] [Google Scholar]
- 58. Oner AF, Bay A, Arslan S et al. Avian influenza A (H5N1) infection in eastern Turkey in 2006. N Engl J Med 2006; 355:2179–2185. [DOI] [PubMed] [Google Scholar]
- 59. Writing committee of the second world health organization consultation on clinical aspects of human infection with avian influenza A (H5N1) virus . Update on avian influenza A (H5N1) virus infection in humans. N Engl J Med 2008; 358:261–273. [DOI] [PubMed] [Google Scholar]
- 60. Van Borm S, Thomas I, Hanquet G et al. Highly pathogenic H5N1 influenza virus in smuggled Thai eagles, Belgium. Emerg Infect Dis 2005; 11:702–705. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61. Tweed SA, Skowronski DM, David TS et al. Human illness from avian influenza H7N3, British Columbia. Emerg Inf Dis 2004; 10:2196–2199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62. Kurtz J, Manvell RJ, Banks J. Avian influenza virus isolated from a woman with conjunctivitis. Lancet 1996; 348:901–902. [DOI] [PubMed] [Google Scholar]
- 63. Taylor HR, Turner AJ. A case report of fowl plague keratoconjunctivitis. Br J Ophthalmol 1977; 61:86–88. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64. Human cases of influenza A(H5N1) infection in eastern Turkey, December 2005 to January 2006, Wkly Epidemiol Rec, 2006; 81:410–416. [PubMed] [Google Scholar]
- 65. Belser JA, Lu X, Maines TR et al. Pathogenesis of avian influenza (H7) virus infection in mice and ferrets: enhanced virulence of Eurasian H7N7 viruses isolated from humans. J Virol 2007; 81:11139–11147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66. Belser JA, Blixt O, Chen LM et al. Contemporary North American influenza H7 viruses possess human receptor specificity: Implications for virus transmissibility. Proc Natl Acad Sci USA 2008; 105:7558–7563. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67. Stevens J, Blixt O, Paulson JC, Wilson IA. Glycan microarray technologies: tools to survey host specificity of influenza viruses. Nat Rev Microbiol 2006; 4:857–864. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68. Stevens J, Blixt O, Tumpey TM et al. Structure and receptor specificity of the hemagglutinin from an H5N1 influenza virus. Science 2006; 312:404–410. [DOI] [PubMed] [Google Scholar]
- 69. Enserink M. Bird flu infected 1000, Dutch researchers say. Science 2004; 306:590. [DOI] [PubMed] [Google Scholar]
- 70. Beigel JH, Farrar J, Han AM et al. Avian influenza A (H5N1) infection in humans. N Engl J Med 2005; 353:1374–1385. [DOI] [PubMed] [Google Scholar]
- 71. Diebold Y, Calonge M, De Salamanca AE et al. Characterization of a spontaneously immortalized cell line (IOBA‐NHC) from normal human conjunctiva. Invest Ophthalmol Vis Sci 2003; 44:4263–4274. [DOI] [PubMed] [Google Scholar]
- 72. Schiff LA, Nibert ML, Tyler KL. Orthoreoviruses and their replication; in Knipe DM, Howley PM. (ed): Fields Virology. Philadelphia, PA: Lippincott Williams & Wilkins, 2007; 1853–1916. [Google Scholar]
- 73. Arisoy ES, Demmler GJ, Thakar S, Doerr C. Meningitis due to parainfluenza virus type 3: report of two cases and review. Clin Infect Dis 1993; 17:995–997. [DOI] [PubMed] [Google Scholar]
- 74. Zhang L, Bukreyev A, Thompson CI et al. Infection of ciliated cells by human parainfluenza virus type 3 in an in vitro model of human airway epithelium. J Virol 2005; 79:1113–1124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75. Yuan P, Thompson TB, Wurzburg BA et al. Structural studies of the parainfluenza virus 5 hemagglutinin‐neuraminidase tetramer in complex with its receptor, sialyllactose. Structure 2005; 13:803–815. [DOI] [PubMed] [Google Scholar]
- 76. Russell RJ, Stevens DJ, Haire LF, Gamblin SJ, Skehel JJ. Avian and human receptor binding by hemagglutinins of influenza A viruses. Glycoconj J 2006; 23:85–92. [DOI] [PubMed] [Google Scholar]
- 77. Ando Y, Iwasaki T, Terao K, Nishimura H, Tamura S. Conjunctivitis following accidental exposure to influenza B virus/Shangdong/07/97. J Infect Dis 2001; 42:223–224. [DOI] [PubMed] [Google Scholar]
- 78. Kunkel F, Herrler G. Structural and functional analysis of the surface protein of human coronavirus OC43. Virology 1993; 195:195–202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79. Arbour N, Day R, Newcombe J, Talbot PJ. Neuroinvasion by human respiratory coronaviruses. J Virol 2000; 74:8913–8921. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80. Imperiale M, Major EO. Polyomaviruses; in Knipe DM, Howley PM.(ed): Fields Virology. Philadelphia, PA: Lippincott Williams & Wilkins, 2007; 2263–2298. [Google Scholar]
- 81. Liu CK, Wei G, Atwood WJ. Infection of glial cells by the human polyomavirus JC is mediated by an N‐linked glycoprotein containing terminal alpha(2–6)‐linked sialic acids. J Virol 1998; 72:4643–4649. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82. Reploeg MD, Storch GA, Clifford DB. BK virus: a clinical review. Clin Infect Dis 2001; 33:191–202. [DOI] [PubMed] [Google Scholar]
- 83. Dugan AS, Eash S, Atwood WJ. An N‐linked glycoprotein with alpha(2,3)‐linked sialic acid is a receptor for BK virus. J Virol 2005; 79:14442–14445. [DOI] [PMC free article] [PubMed] [Google Scholar]


