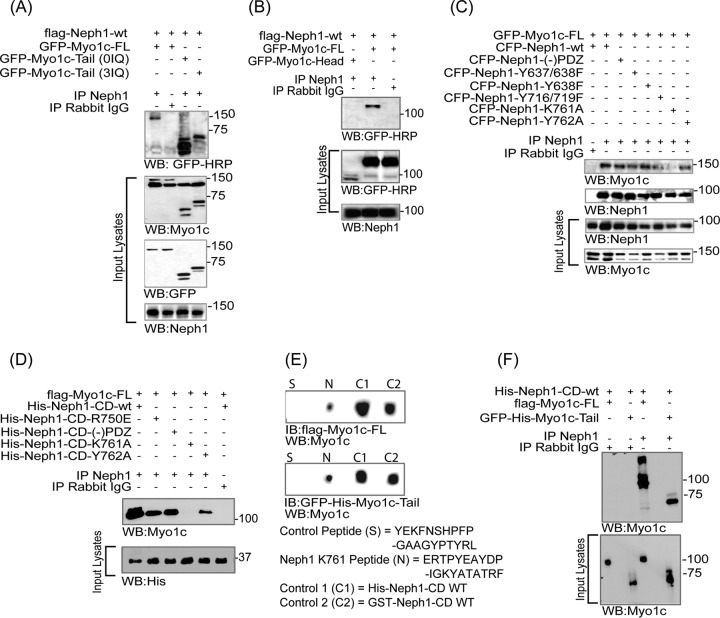
FIG 6.
Neph1 interacts with the IQ domains and C-terminal region of Myo1c. (A and B) COS7 cells were cotransfected with Flag-Neph1-wt and various Myo1c constructs, including GFP-Myo1c-FL, GFP-Myo1c-tail (with no IQ domain), GFP-Myo1c (3IQ domain), and GFP-Myo1c (head), and immunoprecipitated (IP) with Neph1 antibody. Immune complexes were evaluated for binding with Myo1c by Western blotting (WB) with GFP-HRP. Neph1 interacted with GFP-Myo1c-FL and GFP-Myo1c tail with or without the IQ domains but not with the GFP-Myo1c head. (C) In a reciprocal experiment, Neph1 mutants, including the phosphorylation site mutants (CFP-Neph1-Y637/638 and -Y716/719) and the deletion mutant CFP-Neph1-(−)PDZ (without the PDZ binding domain) and the CFP-Neph1-K761 mutant, were cotransfected with GFP-Myo1c-FL in COS7 cells and tested for binding to Myo1c. All the Neph1 mutants interacted with Myo1c except CFP-Neph1-Y761A. (D) The recombinant His-tagged Neph1 (His-Neph1 CD-wt) and its mutants, including His-Neph1-CD-R750E, His-Neph1-CD-(−)PDZ, His-Neph1-CD-K761A, and His-Neph1-CD-Y762A, were separately mixed with the purified Flag-Myo1c-FL protein produced in baculovirus, and pulldown was performed using Neph1 antibody. Western blot analysis using Myo1c antibody showed loss of interaction with Myo1c in the His-Neph1-CD-K761A mutant. (E) A peptide binding experiment showed that similar to purified His-Neph1-CD-wt and GST-Neph1-CD-wt, the peptide containing the K761 region had strong affinity toward Flag-Myo1c-FL and GFP-Myo1c-tail. IB, immunoblotting. (F) Recombinant proteins His-Neph1-CD-wt and either Flag-Myo1c-FL or GFP-His-Myo1c-tail were mixed and pulled down with Neph1 antibody. Western blotting using myo1c antibody was performed to evaluate binding of Neph1 with the Myo1c tail region.