Figure 1. The Unfolded Protein Response.
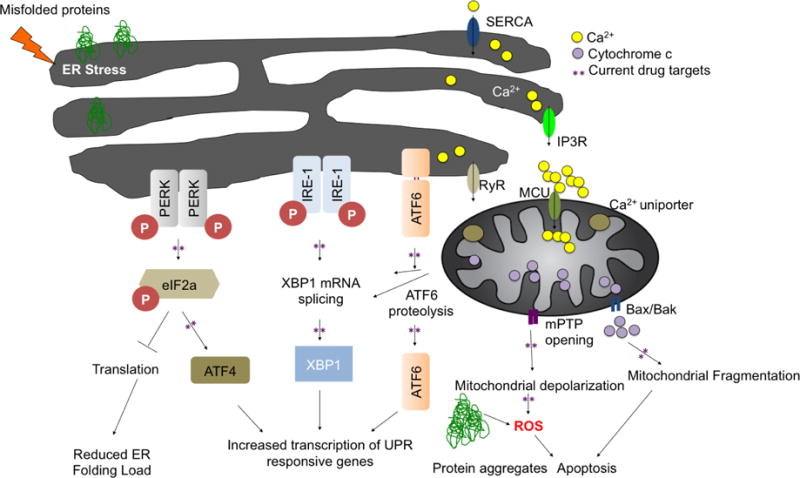
The unfolded protein response consists of three independent signaling pathways that work in parallel and are activated upon accumulation of misfolded proteins inside the ER. ER-stress sensors: RNA-activated protein kinase R (PKR)-like ER kinase (PERK), inositol requiring enzyme 1 (IRE1) and activating transcription factor 6 (ATF6) are membrane resident proteins. The intracellular systems involved in regulating Ca2+ include IP3R: inositol-1,4,5-trisphosphate receptor, RyR: ryanodine receptor, SERCA: ER Ca2+-ATPase at the ER and mPTP: mitochondrial permeability transition pore Bax/Bak and mitochondrial uniporter at the mitochondria. ER stress increases Ca2+ release thus overloading mitochondria, leading to mPTP opening and cytochrome c release. Activation of the UPR leads to an overall translational block and specific activation of ER stress responsive genes, which will increase the protein folding capacity and decrease the protein-folding load in the ER.
