Abstract
Background
Early identification of stroke is crucial to maximize early management benefits in emergency departments. This study aimed to develop and validate a new stroke recognition instrument for differentiating acute stroke from stroke mimics in an emergency setting.
Methods
A prospective observational cohort study among suspected stroke patients presenting to Emergency Department in the Second Affiliated Hospital of Guangzhou Medical University was conducted from May 2012 to March 2013. The symptoms and signs of suspected stroke patients were collected. Logistic regression analysis was used to identify the factors associated with acute stroke. The symptoms and signs closely associated with acute stroke were selected to develop the new stroke scale, Guangzhou Stroke Scale (GZSS). The diagnostic value of GZSS was then compared with ROSIER, FAST and LAPSS. The primary outcome was confirmed stroke by CT within 24 h.
Results
Four hundred and sixteen suspected stroke patients (247 ischemia, 107 hemorrhage, 4 transient ischemic attack, 58 non-stroke) were assessed. A new stroke scale, GZSS (total score from −1 to 8.5), was developed and consisted of nine parameters: vertigo (−1), GCS ≤ 8 (+2), facial paralysis (+1), asymmetric arm weakness (+1), asymmetric leg weakness (+1), speech disturbance (+0.5), visual field defect (+1), systolic blood pressure ≥145 mmHg (+1) and diastolic blood pressure ≥95 mmHg (+1). Among the four scales, the discriminatory value (C-statistic) of GZSS was the best (AUC: 0.871 (p < 0.001) when compared to ROSIER (0.772), LAPSS (0.722) and FAST (0.699). At an optimal cut-off score of >1.5 on a scale from −1 to 8.5, the sensitivity and specificity of GZSS were 83.2 and 74.1 %, whilst the sensitivities and specificities of ROSIER were 77.7 and 70.7 %, FAST were 76.0 and 63.8 %, LAPSS were 56.4 and 87.9 %.
Conclusion
GZSS had better sensitivity than existing stroke scales in Chinese patients with suspected stroke. Further studies should be conducted to confirm its effectiveness in the initial differentiation of acute stroke from stroke mimics.
Keywords: Diagnosis, Stroke, Stroke mimics, ROSIER scale, FAST scale, LAPSS scale, Emergency department, China
Background
Stroke is one of the most common acute and severe diseases presenting to an emergency department (ED) [1]. The early assessment and management of stroke patients should reduce morbidity and mortality [1]. The use of a stroke screening tool to identify the symptoms and signs of suspected stroke and TIA increases diagnostic accuracy of medical staff in pre-hospital and ED [1]. The widely recommended stroke scales in the western world include the Recognition of Stroke in the Emergency Room scale (ROSIER), the Face Arm Speech Test (FAST) and the Los Angeles Prehospital Stroke Screen (LAPSS). ROSIER is a seven-item ranging score from −2 to +5 that includes the clinical history (loss of consciousness, convulsive fit) and neurological signs (face, arm, or leg weakness, speech disturbance, visual field defect). FAST contains three key elements including facial weakness, arm weakness, and speech disturbance. LAPSS consists of four history items, a blood glucose measure, and three examination items designed to detect unilateral motor weakness [2–5]. However, our previous study demonstrated these three stroke scales were not effective for differentiating stroke from stroke mimics in Chinese settings [6, 7]. The reasons may be related to the difference in factors affecting the incidence of stroke subtypes and stroke mimic in different ethnic populations [8]. Therefore, it is necessary to develop a stroke scale suitable to a Chinese emergency setting.
The aims of our study were firstly to identify factors that predict stroke, secondly to develop a new stroke scale in our emergency setting, and thirdly to compare the diagnostic value of the new stroke scale with ROSIER, FAST and LAPSS.
Methods
Study design
A prospective observational study of patients with suspected stroke was conducted from May 2012 to March 2013. Ethical approval was obtained from the Clinical Research Ethics Committee of the 2nd Affiliated Hospital of Guangzhou Medical University. Written consents were also obtained from all patients or the closest available relatives. Patients were informed that they might withdraw from the study at any time.
Study setting
This study was conducted in the emergency department of the second Affiliated Hospital of Guangzhou Medical University (AHGZMU), which serves a population of approximately 1.56 million people in the Hai Zhu district, Guangzhou. It is an academic hospital with 1500 beds affiliated with the Guangzhou Medical University. The ED receives more than 150,000 new patients per annum and serves a local population of approximately 1,550,000 people.
Inclusion and exclusion criteria
Suspected stroke patients ≥18 years old presenting to the ED with symptoms or signs within 7 days were recruited. Patients were excluded if they were <18 years old, had traumatic brain injury, subarachnoid hemorrhage, or unknown diagnoses.
Measurements and data collection
Demographic data, clinical manifestations, risk factors, medical history information and the assessment of ROSIER, FAST and LAPSS were collected [2, 3]. The final diagnosis was made by a specialist stroke physician after assessment and review of clinical symptoms and brain imaging findings (CT or MR). All the patients were divided into stroke or non-stroke groups based on the final diagnosis. Glasgow Coma Scale (GCS) was used to assess the severity of coma (the motor score was applied to the non-affected limb) [9]. All the patients’ scores in this study were assessed by an emergency doctor who has obtained the certification of National Institute of Health Stroke Scale.
Definitions
Stroke was defined as a focal or global neurological deficit with symptoms lasting for 24 h or resulting in death before 24 h, which was thought to be due to a vascular cause after investigation [3]. TIA was defined as clinical syndromes characterized by an acute loss of focal cerebral or monocular function with symptoms lasting less than 24 h and thought to be caused by in adequate blood supply as a result of thrombosis or embolism [3]. Vertigo is defined as the illusion of movement in space [10].
Statistical analyses
Categorical variables were compared using Chi-square analysis, whilst continuous variables were compared using independent t-tests. Univariate analysis was initially used on all variables, and results were presented as ORs with 95 % CIs. Variables that were identified as significant from the univariate analysis (p < 0.05) were entered into logistic regression models to identify independent factors for differentiation of stroke from stroke mimics. The backward stepwise regression analyses were used to construct the models. Significant predictive variables generated in the first model were selected for the final model. The receiver operating characteristics (ROC) curve analysis was utilized to determine the optimal cut-off value of GZSS for discriminating between patients with stroke and stroke mimic. Diagnostic performances of the new stroke scale, ROSIER, FAST and LAPSS were also compared using ROC analysis. The sensitivities, specificities, positive and negative predictive values (PPV and NPV), positive and negative likelihood ratios (LR+ and LR-), and diagnostic accuracy were calculated. Statistical significance was set at p < 0.05. All analyses were performed using SPSS v17.0 (SPSS Inc, IL, USA) and Medcalc v9.5 (MedCalc Software, Mariakerke, Belgium).
Results
Patient characteristics
Four hundred and sixteen patients were assessed between May 2012 and March 2013. There were 358 (86.1 %) stroke cases, including 247 (69.0 %) ischemic stroke, 107 (29.9 %) intracerebral hemorrhage, 4 (1.1 %) TIA, and 58 (13.9 %) non-stroke cases (Fig. 1). Compared with non-stroke group, patients with stroke had higher systolic blood pressure (SBP), diastolic blood pressure (DBP) and incidence of several symptoms and signs including asymmetric facial weakness, asymmetric arm weakness, asymmetric leg weakness, speech disturbance, visual field defect, gaze palsy, loss of consciousness or syncope and pathologic reflex (Table 1). However, non-stroke patients had higher incidence of vertigo, nausea and vomiting. The most common stroke mimics were cervical spondylosis, seizure, peripheral vertigo, which together composed 68.9 % of non-stroke cases (Table 2). Among the 61 cases of stroke patients with GCS ≤8, there were 47 patients with intracerebral hemorrhage (77 %) and 14 patients with ischemia stroke (23 %), including 8 cases with total anterior circulation stroke and middle cerebral artery occlusion, 3 cases with partial anterior circulation stroke and stroke past history, 3 cases with posterior circulation stroke located in brain stem (Table 3).
Fig. 1.
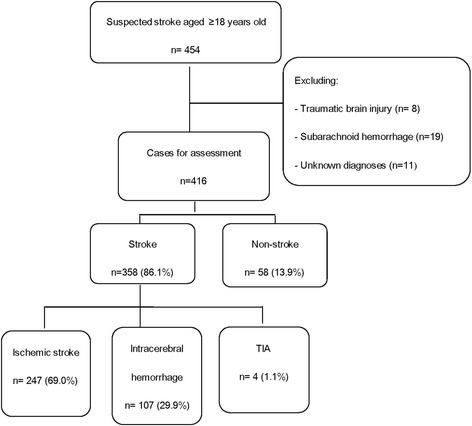
Flowchart of stroke patient recruitment
Table 1.
Baseline demographic characteristics of stroke and non-stroke patients (n = 416)
| Variables | Stroke n = 358 | Non-stroke n = 58 | P |
|---|---|---|---|
| Age | 69.2 ± 13.8 | 70.6 ± 11.4 | 0.397 |
| Male, n (%) | 210 (58.7) | 37 (63.8) | 0.460 |
| SBP, mmHg | 168.8 ± 31.9 | 147.8 ± 27.6 | <0.001* |
| DBP, mmHg | 91.8 ± 20.7 | 80.8 ± 14.3 | <0.001* |
| bpm | 80.9 ± 18.1 | 80.0 ± 13.3 | 0.742 |
| Medical history, n (%) | |||
| Smoker | 146 (40.8) | 20 (34.5) | 0.339 |
| Hypertension | 202 (56.4) | 34 (58.6) | 0.992 |
| Diabetes | 58 (16.2) | 14 (24.1) | 0.157 |
| Hyperlipidemia | 40 (11.2) | 9 (15.5) | 0.407 |
| Ischemic heart disease | 36 (10.1) | 8 (13.8) | 0.434 |
| Atrial fibrillation | 28 (7.8) | 1 (1.7) | 0.148 |
| Symptoms & signs, n (%) | |||
| Asymmetric facial weakness | 181 (50.6) | 8 (13.8) | <0.001* |
| Asymmetric arm weakness | 217 (60.6) | 7 (12.1) | <0.001* |
| Asymmetric leg weakness | 228 (63.7) | 9 (15.5) | <0.001* |
| Speech disturbance | 195 (54.5) | 17 (29.3) | <0.001* |
| Visual field defect | 68 (19.0) | 1 (1.7) | 0.001* |
| gaze palsy | 115 (32.1) | 5 (8.6) | <0.001* |
| Sensory deficits | 78 (21.8) | 8 (13.8) | 0.163 |
| Loss of consciousness or syncope | 119 (33.2) | 10 (17.2) | 0.015* |
| Seizure activity | 21 (5.9) | 6 (10.3) | 0.319 |
| Pathologic reflex | 102 (28.5) | 8 (13.8) | 0.019* |
| Meningeal irritation | 22 (6.1) | 0 (0) | 0.102 |
| Vertigo | 26 (7.3) | 19 (32.8) | <0.001* |
| Headache | 25 (7.0) | 3 (5.2) | 0.816 |
| Nausea | 59 (16.5) | 26 (44.8) | <0.001* |
| Vomiting | 51 (14.2) | 23 (39.7) | <0.001* |
| Score | |||
| GCS > 8, n (%) | 297 (83) | 56 (96.6) | 0.007* |
| GCS ≤ 8, n (%) | 61 (17) | 2 (3.4) | 0.007* |
| GCS, median (IQR) | 15 (12–15) | 15 (15–15) | <0.001* |
*Statistically significant difference was observed between two groups
Table 2.
Diagnoses of stroke and non-stroke patients (n = 416)
| Diagnoses | N (%) |
|---|---|
| Stroke classification | 358 (100.0) |
| Total anterior circulation stroke | 41 (11.5) |
| Partial anterior circulation stroke | 97 (27.1) |
| Lacunar stroke | 88 (24.6) |
| Posterior circulation stroke | 21 (5.7) |
| Intracerebral Hemorrhage | 107 (29.9) |
| Transient Ischemic attack | 4 (1.1) |
| Non-stroke | 58 (100.0) |
| cervical spondylosis | 28 (48.3) |
| Seizure | 6 (10.3) |
| Peripheral vertigo | 6 (10.3) |
| Parkinson’s disease | 2 (3.5) |
| Vasovagal syncope | 2 (3.5) |
| Othera | 14 (24.1) |
aOther diagnoses: hyponatremia and hypokalemia (n = 1), hypoglycemic coma (n = 1), alcoholic cirrhosis (n = 1), overdose of clozapine (n = 1), chronic obstructive pulmonary disease (n = 1), cerebral arteriosclerosis (n = 1), Hashimoto’s encephalopathy (n = 1), vascular headache (n = 1), medulla oblongata and cervical vertebrae (C1-C2) focus (n = 1), scrub typhus (n = 1), central nervous system infection (n = 1), periodic paralysis (n = 1), facial neuritis (n = 1), left middle cerebral artery (M1) following stenting (n = 1)
Table 3.
The subtype of stroke patients with GCS ≤ 8
| Diagnoses | N (%) |
|---|---|
| Intracerebral Hemorrhage | 47 (77.0) |
| Ischemia stroke | 14 (23.0) |
| Total anterior circulation stroke | 8 |
| Partial anterior circulation stroke | 3 |
| Posterior circulation stroke | 3 |
| Total | 61 (100.0) |
Development of the new stroke scale
Logistic regression analysis of clinical symptoms and signs for stroke and non-stroke patients are shown in Table 4. GCS ≤8 was recognized as the highest prevalence, following by visual field defect, asymmetric arm weakness, asymmetric leg weakness, SBP ≥145 mmHg, DBP ≥95 mmHg, speech disturbance and vertigo. As shown in Table 5, the items of the new stroke scale with total score from −1 to 8.5 included vertigo (−1), GCS ≤ 8 (+2), Asymmetric facial weakness (+1), asymmetric arm weakness (+1), asymmetric leg weakness (+1), speech disturbance (+0.5), visual field defect (+1), SBP ≥145 mmHg (+1), DBP ≥95 mmHg (+1). We developed the new scale based on P value of Multiple Logistic regression and odds ratio (Tables 4 and 5).
Table 4.
Factors associated with stroke using logistic regression analysis
| Variables | Univariate regression | Multivariate regression | |||
|---|---|---|---|---|---|
| N (%) | OR (95 % CI) | P | OR (95 % CI) | P | |
| Female | 169 (40.6) | 1 | |||
| Male | 247 (59.4) | 0.81 (0.45–1.43) | 0.461 | ||
| Age | 0.99 (0.97–1.01) | 0.457 | |||
| GCS > 8 | 353 (84.9) | 1 | |||
| GCS ≤ 8 | 63 (15.1) | 5.75 (1.37–24.20) | 0.017* | 5.70 (1.26–25.80) | 0.024* |
| Seizure activity | |||||
| NO | 389 (93.5) | 1 | |||
| YES | 27 (6.5) | 0.54 (0.21–0.54) | 0.205 | ||
| Vertigo | |||||
| NO | 371 (89.1) | 1 | |||
| YES | 45 (10.8) | 0.16 (0.08–0.32) | <0.001* | 0.34 (0.15–0.74) | 0.007* |
| Asymmetric facial weakness | |||||
| NO | 227 (54.6) | 1 | |||
| YES | 189 (45.4) | 6.39 (2.95–13.87) | <0.001* | 2.44 (0.97–6.11) | 0.057 |
| Asymmetric arm weakness | |||||
| NO | 192 (46.2) | 1 | |||
| YES | 224 (53.8) | 11.21 (4.95–25.41) | <0.001* | 3.39 (1.04–11.08) | 0.043* |
| Asymmetric leg weakness | |||||
| NO | 179 (43.0) | 1 | |||
| YES | 237 (57.0) | 9.55 (4.54–20.07) | <0.001* | 2.77 (0.88–8.70) | 0.081 |
| Speech disturbance | |||||
| NO | 204 (49.0) | 1 | |||
| YES | 212 (51.0) | 2.89 (1.58–5.27) | 0.001* | 1.29 (0.58–2.84) | 0.524 |
| Visual field defect | |||||
| NO | 347 (83.4) | 1 | |||
| YES | 69 (16.6) | 13.37 (1.82–98.23) | 0.011* | 3.93 (0.48–32.30) | 0.202 |
| Gaze palsy | |||||
| NO | 296 (71.2) | 1 | |||
| YES | 120 (28.8) | 5.02 (1.95–12.89) | 0.001* | 0.75 (0.19–3.08) | 0.693 |
| Pathologic reflex | |||||
| NO | 306 (73.6) | 1 | |||
| YES | 110 (26.4) | 2.49 (1.14–5.44) | 0.022* | 0.73 (0.28–1.91) | 0.516 |
| SBP ≥ 145 mmHg | |||||
| NO | 111 (26.7) | 1 | |||
| YES | 305 (73.3) | 3.99 (2.25–7.07) | <0.001* | 2.55 (1.25–5.24) | 0.011* |
| DBP ≥ 95 mmHg | |||||
| NO | 270 (64.9) | 1 | |||
| YES | 146 (35.1) | 4.62 (2.04–10.48) | <0.001* | 2.29 (0.87–6.03) | 0.093 |
*Statistically significant (p <0.05)
Table 5.
Clinical signs and symptoms for development of the new stroke scale
| Variables | Score | β | OR (95 % CI) | P value |
|---|---|---|---|---|
| Vertigo | −1 | −1.09 | 0.34 (0.15–0.74) | 0.007* |
| GCS ≤ 8 | 2 | 1.74 | 5.70 (1.26–25.80) | 0.024* |
| Asymmetric facial weakness | 1 | 0.89 | 2.44 (0.97–6.11) | 0.057 |
| Asymmetric arm weakness | 1 | 1.22 | 3.39 (1.04–11.08) | 0.043* |
| Asymmetric leg weakness | 1 | 1.02 | 2.77 (0.88–8.70) | 0.081 |
| Speech disturbance | 0.5 | 0.26 | 1.29 (0.58–2.84) | 0.524 |
| Visual field defect | 1 | 1.37 | 3.93 (0.48–32.30) | 0.202 |
| SBP ≥ 145 mmHg | 1 | 0.94 | 2.55 (1.25–5.24) | 0.011* |
| DBP ≥ 95 mmHg | 1 | 0.83 | 2.29 (0.87–6.03) | 0.093 |
*P < 0.05
Stroke is likely if total scores are >1.5
Diagnostic performances of ROSIER, FAST, LAPSS and GZSS
The nine-item scoring system for GZSS was constructed. Comparison of GZSS to ROSIER, FAST and LAPSS was undertaken using ROC analysis (Fig. 2). The area under curve (AUC) of GZSS was 0.871 (95 % CI 83.5–90.2), whilst the AUC of ROSIER was 0.772 (95% CI 72.8–81.1), LAPSS was 0.722 (95 % CI 67.6–74.6) and FAST was 0.699 (95 % CI 65.2–74.3). By pairwise comparison of the AUC of the four scales (adjusted α = 0.008), the comparison was statistically significant (p < 0.001). Among the four scales, the diagnostic value of GZSS was the best (Fig. 2).
Fig. 2.
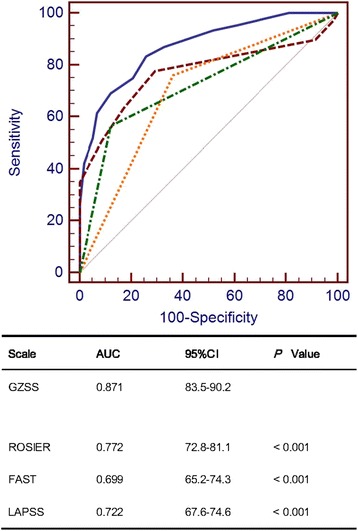
Receiver operating characteristic ROC curves of GZSS, ROSIER, FAST and LAPSS in differentiation of stroke and non-stroke patients
The optimal cut-off score of GZSS was determined to be a total score of 1.5. At this cut-off score, the corresponding sensitivity and specificity were 83.2 % (95 % CI 79.0–87.0) and 74.1 % (95 % CI 61.0–84.7). The sensitivity and specificity of ROSIER scale were 77.7 % (95 % CI 73.0–81.9) and 70.7 % (95 % CI 57.3–81.9) when the optimal cut-off score was 0. The sensitivity and specificity of FAST scale were 76.0 % (95 % CI 71.2–80.3) and 63.8 % (95 % CI 50.1–76.0) when the optimal cut-off score was 0. The sensitivity and specificity of LAPSS scale were 56.4 % (95 % CI 51.1–61.6) and 87.9 % (95 % CI 76.7–95.0) when the optimal cut-off score was 0. The sensitivity of GZSS was the best and was significantly different from the sensitivities of ROSIER scale (p = 0.031), FAST scale (p = 0.004) and LAPSS scale (p < 0.001) (Table 6).
Table 6.
Diagnostic performance of ROSIER, FAST, LAPSS and GZSS
| Scale | Optimal cut-off | Sensitivity (95 % CI) | P | Specificity (95 % CI) | P | PLR (95 % CI) | NLR (95 % CI) | PPV (95 % CI) | NPV (95 % CI) | Diagnostic accuracy (%) |
|---|---|---|---|---|---|---|---|---|---|---|
| ROSIER | 0 | 77.7 (73.0–81.9) | 0.031 | 70.7 (57.3–81.9) | 0.754 | 2.65 (1.8–4.0) | 0.32 (0.2–0.4) | 94.2 (90.9–96.6) | 33.9 (25.5–43.1) | 76.68 |
| FAST | 0 | 76.0 (71.2–80.3) | 0.004 | 63.8 (50.1–76.0) | 0.210 | 2.1 (1.5–3.0) | 0.38 (0.3–0.5) | 92.8 (89.2–95.5) | 30.1 (22.1–39.0) | 74.28 |
| LAPSS | 0 | 56.4 (51.1–61.6) | <0.001 | 87.9 (76.7–95.0) | 0.008 | 4.68 (2.3–9.4) | 0.50 (0.4–0.6) | 96.7 (93.2–98.6) | 24.6 (18.9–31.1) | 60.82 |
| GZSS | 1.5 | 83.2 (79.0–87.0) | Reference | 74.1 (61.0–84.7) | Reference | 3.22 (2.1–5.0) | 0.23 (0.2–0.3) | 95.2 (92.2–97.3) | 41.7 (32.1–51.9) | 81.97 |
ROSIER Recognition of Stroke in the Emergency Room scale, FAST Face Arm Speech Test, LAPSS Los Angeles Pre-Hospital Stroke Screen, GZSS Guangzhou Stroke Scale, PLR positive likelihood ratio, NLR negative likelihood ratio, PPV positive predictive value, NPV negative predictive value
Discussion
This was the first study to develop a new stroke scale in China. In recent years, the incidence of stroke is still rising year by year around the world and its high morbidity can cause serious social and family burden [11–14]. Early recognition and timely treatment of patients with acute stroke by emergency physicians are critical and improve the prognosis of stroke patients [15–22]. Stroke screening scales were recommended by guidelines in pre-hospital and emergency room for rapid triage of suspected patients [1, 3, 23–25].
In this study, we found that there were significant differences in the distribution of stroke subtypes between the patients in our study and the Western patients in the studies using other scales. There was a higher proportion of intracerebral haemorrhage in our study compared with the other Western studies [26–29]. Also, there were less ischaemic stroke (69 versus 76 %) and TIA (1.1 versus 10 %), but more haemorrhagic stroke (29.9 versus 14 %) in our study than in the ROSIER study [2]. These differences in subtype patterns are postulated to be due to differences in genetic, clinical, environmental and lifestyle factors [8, 30, 31]. There were also differences in the proportion of stroke mimics between this study and the other Western studies, Seizures (10.3 %) and syncope (17.2 %) in Guangzhou were less common than in the UK (47 %) [2]. Another UK study found that primary headache disorders and seizures comprised up to 27 % of stroke mimics [32], whilst in Greece [33] the principal stroke mimics were aphasic disturbances (27.3 %) and dizziness or fainting (27.3 %).
We analyzed different clinical features of stroke in our ED and thus developed a new stroke recognition instrument (score range: −1 to 8.5), which consisted of nine items including vertigo (−1), GCS ≤ 8 (+2), facial paralysis (+1), asymmetric upper limb paralysis (+1), asymmetric lower limb paralysis (+1), speech disorders (+0.5), visual field defect (+1), SBP ≥145 mmHg (+1), DBP ≥95 mmHg (+1). The new stroke scale showed good discriminative value in our ED.
In GZSS, five recognition items were the same as in ROSIER scale, FAST score and LAPSS. These items with different odds ratios were included in the new stroke scale, such as asymmetric facial weakness (OR: 6.39), asymmetric arm weakness (OR: 11.21), asymmetric leg weakness (OR: 9.55), speech disturbance (OR: 2.89), visual field defect (OR: 13.37). We assigned the corresponding score to each item of the new scale based on the logistic regression coefficients.
There were some new items added in GZSS based on the analysis. In our previous study, we found that the level of consciousness of patients may affect the diagnostic value of the stroke screening scales [2, 6, 7]. Therefore, we assessed the diagnostic value of GCS in patients with suspected stroke. We found that GCS equal or less than 8 points (OR: 5.75) was associated with the diagnosis of stroke. Stroke patients often have disturbance of consciousness and are unable to cooperate in medical examinations. It would be difficult for coma patients to carry out some of the physical examinations, such as paralysis and speech disorders. In this case, most of the stroke screening scales developed in western countries, such as FAST and LAPSS, cannot be applied to stroke patients with loss of consciousness. Also, if stroke patients present with loss of consciousness and do not have paralysis and speech disorders, the total score of ROSIER scale is equal to or less than 0 and thus means stroke is not likely to occur. This would easily lead to false negative. Therefore, we included GCS in GZSS to compensate the deficiency of the other stroke scales.
Vertigo was another new item in GZSS. Our study suggested vertigo occurred more often in Chinese non-stroke population than Western population. In the western population, the proportion of stroke and non-stroke patients with vertigo were similar (6 and 5 %, respectively) [2]. However, 58.6 % of patients had cervical spondylosis and peripheral vertigo in this study. By logistic regression analysis, the regression coefficient βof vertigo was negative, which suggested that vertigo was a differential symptom between stroke and non-stroke patients.
In addition, our new scale included SBP and DBP. We used ROC analysis to determine the optimal cut-off values of SBP (≥145 mmHg) and DBP (≥95 mmHg). By logistic regression analysis, we found that the OR of SBP and DBP were2.55 and 2.29, respectively. When acute stroke occurs, SBP and DBP are higher than usual. Blood pressure of more than 80 % of patients increased within 24 to 48 h after the onset of cerebral ischemic, and declined gradually in a few days or several weeks. One of the reasons might be due to the regulation disorder of cerebral blood flow in ischemic penumbra [34, 35]. Patients with hemorrhagic stroke experienced increased intracranial pressure, and thus the blood pressure would increase to maintain the normal cerebral blood flow. In this study, the blood pressure was higher in patients with stroke than non-stroke (p < 0.01). Also, 392 patients (94.2 %) including 337 stroke patients (81.0 %) and 55 non-stroke patients (13.2 %) presented to ED within 24 h after symptom onset. Only 5.8 % of patients presented over 24 h after symptom onset. This indicated that the vast majority of patients with suspected stroke in our study were in acute period of cerebral apoplexy. Therefore, blood pressure also played a significant role in the diagnosis of patients with suspected stroke in the emergency department.
A comparison of the new stroke scale with the other three scales (ROSIER scale, FAST scale and LAPSS scale) was conducted in our ED. We found that the new scale had better sensitivity than the other scales. The AUC of GZSS was the largest (AUC = 0.871). At the optimal cut-off score of 1.5, GZSS gave high sensitivity and comparable specificity. It may be more effective to use GZSS to screen Chinese patients with suspected stroke in ED.
Limitations
There were several limitations in this study. First, this study was a single-center study. Our results may not be generalizable to other parts of China, let alone elsewhere in the world. Multicenter studies with larger sample sizes are needed to explore the effectiveness of this new stroke scale. Second, using GCS in GZSS may not be appropriate in all circumstances. A patient who has had a stroke may be aphasic and hemiplegic. They may be fully conscious but only scored E4V1M6 giving them a GCS of 11/15, which is clearly misleading. It may be necessary to break down the GCS into the component parts (i.e. E4V1M6 instead of GCS 9) to get much more information. Third, it would have been stronger to have separate derivation and validation datasets rather than a single dataset. Therefore, further studies with larger sample sizes are required to validate the effectiveness of GZSS and improve its weakness.
Conclusion
GZSS had better sensitivity than the existing stroke scales in Chinese patients with suspected stroke. Further studies are required to validate its usefulness in the initial differentiation of acute stroke from stroke mimics.
Acknowledgements
This research study was supported by the Key Medical Disciplines and Specialties Program of Guangzhou and the Guangzhou City Health Bureau (Grant No.20141A011084).
Funding
This research study was supported by the Key Medical Disciplines and Specialties Program of Guangzhou and the Guangzhou City Health Bureau (Grant No.20141A011084).
Availability of data and materials
The datasets during and/or analysed during the current study available from the corresponding author on reasonable request.
Authors’ contributions
HJ, HM and PL designed the study and undertook the statistical analysis. HM, JM and YL were involved in collecting data. HJ and HM drafted the manuscript. XC and THR contributed to manuscript revision. The corresponding author HJ supervised the whole study. All authors have read and approved the final manuscript.
Competing interests
The authors declare that they have no competing interests.
Consent for publication
Not applicable.
Ethics approval and consent to participate
Ethical approval was obtained from the Clinical Research Ethics Committee of the 2nd Affiliated Hospital of Guangzhou Medical University. Written consents were also obtained from all patients or the closest available relatives. Patients were informed that they might withdraw from the study at any time.
Abbreviations
- AUC
area under the ROC curve
- CT
computed tomography
- DWI
diffusion weighted imaging
- FAST
the face arm speech test
- GCS
Glasgow Coma Scale
- IQR
inter quartile range
- LAPSS
the Los Angeles Prehospital Stroke Screen
- MRI
magnetic resonance imaging
- NIHSS
National Institute of Health stroke scale
- OR
odds ratio
- ROC
receiver operating characteristic
- ROSIER
the Recognition of Stroke in the Emergency Room scale
- TIA
transient ischemic attack
Contributor Information
Haifeng Mao, Email: maomao2010x@163.com.
Peiyi Lin, Email: linpeiyi@163.com.
Junrong Mo, Email: junrongmo@163.com.
Yunmei Li, Email: liyunmeially@163.com.
Xiaohui Chen, Email: cxhgz168@126.com.
Timothy H. Rainer, Email: RainerTH@cardiff.ac.uk
Huilin Jiang, Email: lifisher@126.com.
References
- 1.Jauch EC, Saver JL, Adams HP, Bruno A, Connors JJ, Demaerschalk BM, et al. Guidelines for the early management of patients with acute ischemic stroke: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke. 2013;44:870–947. doi: 10.1161/STR.0b013e318284056a. [DOI] [PubMed] [Google Scholar]
- 2.Nor AM, Davis J, Sen B, Shipsey D, Louw SJ, Dyker AG, et al. The Recognition of Stroke in the Emergency Room (ROSIER) scale: development and validation of a stroke recognition instrument. Lancet Neurol. 2005;4:727–734. doi: 10.1016/S1474-4422(05)70201-5. [DOI] [PubMed] [Google Scholar]
- 3.Harbison J, Hossain O, Jenkinson D, Davis J, Louw SJ, Ford GA. Diagnostic accuracy of stroke referrals from primary care, emergency room physicians, and ambulance staff using the face arm speech test. Stroke. 2003;34:71–76. doi: 10.1161/01.STR.0000044170.46643.5E. [DOI] [PubMed] [Google Scholar]
- 4.Kidwell CS, Starkman S, Eckstein M, Weems K, Saver JL. Identifying stroke in the field. Prospective validation of the Los Angeles prehospital stroke screen (LAPSS) Stroke. 2000;31:71–76. doi: 10.1161/01.STR.31.1.71. [DOI] [PubMed] [Google Scholar]
- 5.Kidwell CS, Saver JL, Schubert GB, Eckstein M, Starkman S. Design and retrospective analysis of the Los Angeles Prehospital Stroke Screen (LAPSS) Prehosp Emerg Care. 1998;2:267–273. doi: 10.1080/10903129808958878. [DOI] [PubMed] [Google Scholar]
- 6.Jiang HL, Chan CP, Leung YK, Li YM, Graham CA, Rainer TH. Evaluation of the Recognition of Stroke in the Emergency Room (ROSIER) scale in Chinese patients in Hong Kong. PLoS One. 2014;9:e109762. doi: 10.1371/journal.pone.0109762. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Chen XH, Mao HF, Mo JR, Lin PY, Tian CW, Zhang Y, et al. Validation of three different stroke screen scales in an Emergency setting. Chinese Journal of Critical Care Medicine. 2013;33:539–542. [Google Scholar]
- 8.Zhang LF, Yang J, Hong Z, Yuan GG, Zhou BF, Zhao LC, et al. Proportion of different subtypes of stroke in China. Stroke. 2003;34:2091–2096. doi: 10.1161/01.STR.0000087149.42294.8C. [DOI] [PubMed] [Google Scholar]
- 9.Ling G. Traumatic brain injury and spinal cord injury. In: Goldman L, Schafer AI, editors. Goldman’s Cecil medicine. 24. Singapore: Elsevier; 2012. p. 2253. [Google Scholar]
- 10.Baloh RW, Jen J. Hearing and equilibrium. In: Goldman L, Schafer AI, editors. Goldman’s Cecil medicine. 24. Singapore: Elsevier; 2012. p. 2465. [Google Scholar]
- 11.Murray CJ, Vos T, Lozano R, Naghavi M, Flaxman AD, Michaud C, et al. Disability-adjusted life years (DALYs) for 291 diseases and injuries in 21 regions, 1990–2010: a systematic analysis for the Global Burden of Disease Study 2010. Lancet. 2012;380:2197–2223. doi: 10.1016/S0140-6736(12)61689-4. [DOI] [PubMed] [Google Scholar]
- 12.Norrving B, Kissela B. The global burden of stroke and need for a continuum of care. Neurology. 2013;80:S5–S12. doi: 10.1212/WNL.0b013e3182762397. [DOI] [PubMed] [Google Scholar]
- 13.Johnston SC, Mendis S, Mathers CD. Global variation in stroke burden and mortality: estimates from monitoring, surveillance, and modelling. Lancet Neurol. 2009;8:345–354. doi: 10.1016/S1474-4422(09)70023-7. [DOI] [PubMed] [Google Scholar]
- 14.Hu XL, Gong XG. The economic burden of ischemic stroke in China. China Healthcare Economy. 2003;22:18–20. [Google Scholar]
- 15.Hacke W, Kaste M, Bluhmki E, Brozman M, Dávalos A, Guidetti D, et al. Thrombolysis with alteplase 3 to 4.5 hours after acute ischemic stroke. N Engl J Med. 2008;359:1317–1329. doi: 10.1056/NEJMoa0804656. [DOI] [PubMed] [Google Scholar]
- 16.Sandercock P, Wardlaw JM, Lindley RI, Dennis M, Cohen G, Murray G, et al. The benefits and harms of intravenous thrombolysis with recombinant tissue plasminogen activator within 6 h of acute ischaemic stroke (the third international stroke trial [IST-3]): a randomised controlled trial. Lancet. 2012;379:2352–2363. doi: 10.1016/S0140-6736(12)60768-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Morgenstern LB, Hemphill JC, 3rd, Anderson C, Becker K, Broderick JP, Connolly ES, Jr, et al. Guidelines for the management of spontaneous intracerebral hemorrhage: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke. 2010;41:2108–2129. doi: 10.1161/STR.0b013e3181ec611b. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Shah KH, Kleckner K, Edlow JA. Short-term prognosis of stroke among patients diagnosed in the emergency department with a transient ischemic attack. Ann Emerg Med. 2008;51:316–323. doi: 10.1016/j.annemergmed.2007.08.016. [DOI] [PubMed] [Google Scholar]
- 19.Hacke W, Donnan G, Fieschi C, Kaste M, von Kummer R, Broderick JP, et al. Association of outcome with early stroke treatment: pooled analysis of ATLANTIS, ECASS, and NINDS rt-PA stroke trials. Lancet. 2004;363:768–774. doi: 10.1016/S0140-6736(04)15692-4. [DOI] [PubMed] [Google Scholar]
- 20.Marler JR, Tilley BC, Lu M, Brott TG, Lyden PC, Grotta JC, et al. Early stroke treatment associated with better outcome: the NINDS rt-PA stroke study. Neurology. 2000;55:1649–1655. doi: 10.1212/WNL.55.11.1649. [DOI] [PubMed] [Google Scholar]
- 21.Moon JS, Janjua N, Ahmed S, Kirmani JF, Harris-Lane P, Jacob M, et al. Prehospital neurologic deterioration in patients with intracerebral hemorrhage. Crit Care Med. 2008;36:172–175. doi: 10.1097/01.CCM.0000297876.62464.6B. [DOI] [PubMed] [Google Scholar]
- 22.Brott T, Broderick J, Kothari R, Barsan W, Tomsick T, Sauerbeck L, et al. Early hemorrhage growth in patients with intracerebral hemorrhage. Stroke. 1997;28:1–5. doi: 10.1161/01.STR.28.1.1. [DOI] [PubMed] [Google Scholar]
- 23.Azzimondi G, Bassein L, Fiorani L, Nonino F, Montaguti U, Celin D, et al. Variables associated with hospital arrival time after stroke: effect of delay on the clinical efficiency of early treatment. Stroke. 1997;28:537–542. doi: 10.1161/01.STR.28.3.537. [DOI] [PubMed] [Google Scholar]
- 24.Ferro JM, Pinto AN, Falcao I, Rodrigues G, Ferreira J, Falcão F, et al. Diagnosis of stroke by the nonneurologist. A validation study. Stroke. 1998;29:1106–1109. doi: 10.1161/01.STR.29.6.1106. [DOI] [PubMed] [Google Scholar]
- 25.Moulin T, Sablot D, Vidry E, Belahsen F, Berger E, Lemounaud P, et al. Impact of emergency room neurologists on patient management and outcome. Eur Neurol. 2003;50:207–214. doi: 10.1159/000073861. [DOI] [PubMed] [Google Scholar]
- 26.Islam MS, Anderson CS, Hankey GJ, Hardie K, Carter K, Broadhurst R, et al. Trends in incidence and outcome of stroke in Perth, Western Australia during 1989 to 2001: the Perth Community Stroke Study. Stroke. 2008;39:776–782. doi: 10.1161/STROKEAHA.107.493643. [DOI] [PubMed] [Google Scholar]
- 27.Béjot Y, Aouba A, de Peretti C, Grimaud O, Aboa-Eboulé C, Chin F, et al. Time trends in hospital-referred stroke and transient ischemic attack: result of a 7-year nationwide survey in France. Cerebrovasc Dis. 2010;30:346–354. doi: 10.1159/000319569. [DOI] [PubMed] [Google Scholar]
- 28.Corso G, Bottacchi E, Giardini G, De la Pierre F, Meloni T, Pesenti Campagnoni M, et al. Community-based study of stroke incidence in the Valley of Aosta, Italy. Neuroepidemiology. 2009;32:186–195. doi: 10.1159/000195688. [DOI] [PubMed] [Google Scholar]
- 29.Tsai CF, Thomas B, Sudlow CL. Epidemiology of stroke and its subtypes in Chinese vs white populations. Neurology. 2013;81:264–272. doi: 10.1212/WNL.0b013e31829bfde3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Jiang B, Wang WZ, Chen H, Hong Z, Yang QD, Wu SP, et al. Incidence and trends of stroke and its subtypes in China:results from three large cities. Stroke. 2006;37:63–68. doi: 10.1161/01.STR.0000194955.34820.78. [DOI] [PubMed] [Google Scholar]
- 31.Feigin VL, Lawes CM, Bennett DA, Anderson CS. Stroke epidemiology: a review of population-based studies of incidence, prevalence, and case-fatality in the late 20th century. Lancet Neurol. 2003;2:43–53. doi: 10.1016/S1474-4422(03)00266-7. [DOI] [PubMed] [Google Scholar]
- 32.Whiteley WN, Wardlaw JM, Dennis MS, Sandercock PA. Clinical scores for the identification of stroke and transient ischaemic attack in the emergency department: a cross-sectional study. J Neurol Neurosurg Psychiatry. 2011;82:1006–1010. doi: 10.1136/jnnp.2010.235010. [DOI] [PubMed] [Google Scholar]
- 33.Hatzitolios A, Savopoulos C, Ntaios G, Papadidaskalou F, Dimitrakoudi E, Kosmidou M, et al. Stroke and conditions that mimic it: a protocol secures a safe early recognition. Hippokratia. 2008;12:98–102. [PMC free article] [PubMed] [Google Scholar]
- 34.Bath P, Chalmers J, Powers W, Beilin L, Davis S, Lenfant C, et al. International Society of Hypertension (ISH): statement on the management of blood pressure in acute stroke. J Hypertens. 2003;21:665–672. doi: 10.1097/00004872-200304000-00003. [DOI] [PubMed] [Google Scholar]
- 35.Blood pressure, cholesterol, and stroke in eastern Asia. Eastern Stroke and Coronary Heart Disease Collaborative Research Group. Lancet. 1998; 352:1801–7. [PubMed]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets during and/or analysed during the current study available from the corresponding author on reasonable request.


