Abstract
Aims/hypothesis
Haptoglobin (Hp) 2-2 genotype has been shown to increase coronary artery disease (CAD) risk in numerous type 2 diabetes studies but in only one type 1 diabetes cohort. We assessed the association of Hp 2-2 with incident CAD over 26 years of follow-up in 1,303 Caucasian participants of the Diabetes Control and Complications Trial/Epidemiology of Diabetes Interventions and Complications (DCCT/EDIC) study.
Methods
DCCT randomized volunteers with type 1 diabetes to intensive versus conventional therapy within two cohorts: ‘primary prevention’ with 1–5 years diabetes duration and ‘secondary intervention’ with 1–15 years diabetes duration and early retinopathy, with or without albuminuria, but no advanced complications. CAD was defined as myocardial infarction (MI) or death judged to be from CAD, silent MI, angina, coronary revascularization, or congestive heart failure due to CAD.
Results
In the entire DCCT cohort, Hp 2-2 was not significantly associated with incident CAD or MI. However, in pre-specified exploratory subgroup analyses, an increased MI risk was suggested in the secondary cohort for those with Hp 2-2.
Conclusions/Interpretation
The analysis does not statistically confirm an overall association between Hp 2-2 and incident CAD, however, some suggestions of associations were observed in secondary analyses.
Introduction
Individuals with type 1 diabetes are at increased risk for cardiovascular complications even though, with the exception of hyperglycemia, traditional risk factor profiles are similar to, or even better than, those in the general population (1, 2). In 2005, the Diabetes Control and Complications Trial/Epidemiology of Diabetes Interventions and Complications (DCCT/EDIC) study demonstrated that intensive therapy yields substantial cardiovascular benefit in this patient population (3). Unfortunately, despite the reduction in CVD risk resulting from intensive diabetes management, aimed at achieving glycemic control as close to the non-diabetic range as possible, cardiovascular event rates in type 1 diabetes continue to be reported as being higher than that observed in the general population (4), suggesting that other factors, including genetic, may also play a role. One such genetic factor may comprise a copy number repeat polymorphism in the haptoglobin (Hp) gene (rs72294371).
Hp is a plasma glycoprotein considered to have anti-inflammatory and anti-oxidative properties (5). As inflammation and oxidation are also implicated in the development of atherosclerosis, it is possible that Hp may play an important role in determining susceptibility to early atherosclerotic disease. In humans, Hp is polymorphic with two major alleles which differ by the presence or absence of 1.7 kb partial intragenic duplication, and three genotypes denoted Hp 1-1, 2-1 and 2-2 (6). Structural and functional differences exist between the two Hp protein allele products, with the Hp 2-2 protein having less effective anti-inflammatory/anti-oxidative properties than Hp 1-1 (7–12).
These functional differences are thought to underlie the strong, consistent, findings of increased cardiovascular disease risk, particularly myocardial infarction, among Hp 2-2 genotype carriers as compared to non-Hp 2-2 genotype carriers in cohort studies of type 2 diabetes (13–16). In type 1 diabetes, the Pittsburgh Epidemiology of Diabetes Complications (EDC) study has been the only thus far to evaluate the role of the Hp genotype in the development of CAD, providing similar evidence for an increased risk with the Hp 2-2 genotype (17). In EDC, the Hp 2-2 genotype was also a strong predictor of declining renal function and progression to end-stage renal disease (18), findings which were recently confirmed in the DCCT/EDIC study (19), as well as of mortality attributed to cardiovascular or renal causes (20).
In the present investigation we sought to: 1) confirm the association between Hp genotype and CAD incidence previously demonstrated in type 1 diabetes mellitus (DM) in the EDC study cohort in the DCCT/EDIC cohort; and 2) determine whether the associations between Hp genotype and CAD and myocardial infarction are modified by either the original DCCT treatment group (intensive or conventional) or cohort (primary prevention or secondary intervention) in the DCCT/EDIC cohort.
Research Design and Methods
Detailed descriptions of the methods of the DCCT and EDIC follow-up study have been published previously (19, 21–23). The DCCT, a randomized controlled clinical trial conducted between 1983 and 1993, compared the effects of an intensive diabetes treatment regimen (n=730) with those of conventional therapy (n=711) on the development and progression of microvascular complications. The primary prevention cohort included 726 subjects with diabetes for 1–5 years, no retinopathy, and <40 mg albuminuria per 24 h, and the secondary intervention cohort included 715 participants who had diabetes for 1–15 years with mild to moderate non-proliferative retinopathy and urinary albumin excretion rate <200 mg/d. At baseline, eligibility criteria excluded patients with a history of cardiovascular disease, hypertension or hypercholesterolemia (22).
Of the 1441 patients with type 1 diabetes, who were 13 to less than 40 years old at the time of randomization, 1422 completed the DCCT; the mean follow-up was 6.5 years. During the DCCT, intensive (INT) and conventional (CONV) therapies achieved mean HbA1c levels of 7% and 9% respectively (21). During a bridge period between the DCCT and EDIC, the original conventional group patients were instructed in intensive therapy and ongoing diabetes care was transferred back to the subject’s local providers (23). In 1994, 1394 subjects of the surviving cohort agreed to participate in the long term EDIC observational follow-up study. All participants were encouraged to follow an intensive therapy regimen, and HbA1c has been similar in both groups within 5 years after the initiation of EDIC. This report includes data obtained through April 31, 2010 from 1303 Caucasian participants. Individuals who were determined to be admixed between Caucasian and other ethnic groups by population genetic approaches, using the software Eigenstrat (24), seeding with genotype data from the 3 major populations genotyped in HapMap phase II (25) (n=136), were excluded because of their small number and racial variation in Hp genotypes.
Study Procedures
Glycated hemoglobin was measured quarterly during DCCT and annually during EDIC (23, 26). Renal function (serum creatinine) was measured annually throughout DCCT and EDIC (27). Serum creatinine levels, age, sex and race were used to estimate glomerular filtration rate by the Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI) equation (28). Urinary albumin excretion rate was measured annually during DCCT and every other year during EDIC from a clinic-based, four hour timed, urine collection. Microalbuminuria and macroalbuminuria were defined as ≥30 mg in a 24-hour period at two consecutive study visits and ≥300 mg in a 24- hour period at a single visit, respectively (27).
Coronary artery disease (CAD) events were determined on the basis of clinical history and ECG recordings obtained annually or from death records. All hospitalized events and death records were reviewed by the Mortality and Morbidity Committee (MMC) to determine the presence of a CAD event defined as any of the following: fatal myocardial infarction (MI) or death judged to be from CAD by the MMC, non-fatal MI based on ECG finding and cardiac enzyme level from the hospital records, silent MI based on significant annual serial changes in the ECG read by a central reading center, angina confirmed by ischemic changes with exercise tolerance testing or by clinically significant obstruction on coronary angiography, coronary revascularization, or congestive heart failure due to CAD.
Method for Hp typing
Serum samples from the 1303 subjects in this study were typed for Hp genotype by polyacrylamide gel electrophoresis (PAGE) and ELISA with no knowledge of patient identity. Two samples could not be Hp typed using either method. There was a difference in the Hp type obtained by PAGE and ELISA for five samples and for the analyses performed herein, the results obtained by PAGE were used. In 12 samples where an Hp type could not be obtained by PAGE, the Hp type used in this analysis was obtained by ELISA. Both the PAGE (29) and ELISA methods have been described in detail previously (19).
Statistical analysis
Due to the relatively small size of the cohort and resulting inadequate power to assess effects of the three individual genotypes, we pre-specified that the combined Hp 1-1 and Hp 2-1 group (or non-Hp 2-2) would be compared to the high risk Hp 2-2 genotype for all analyses. Clinical characteristics between the Hp 2-2 and non-Hp 2-2 groups were compared using the Wilcoxon rank sums test for continuous variables and the Chi-Square test for categorical variables. The cumulative incidence of CAD events between the Hp 2-2 and non-Hp 2-2 groups was estimated by the Kaplan-Meier method (30) and differences between groups were tested using the log-rank test (30). Cox proportional hazards models (30–31) were used to assess the effect of Hp type on the risk of CAD and MI/silent MI events without (unadjusted) and with (adjusted) other covariates to estimate the hazard ratio (HR) between Hp types, and group differences were assessed using the Wald test. The models also were adjusted for population stratification using three principal components of ancestry previously derived for DCCT/EDIC (32–33). The models were evaluated by diabetes treatment (INT vs. CON) and study cohort (primary prevention and secondary intervention) separately, were tested for interaction between Hp and treatment group or study cohort, and were tested for three-way interactions among Hp, treatment group, and study cohort.
All analyses were performed using the SAS software (version 9.3; SAS Institute, Cary, NC) and R package (34). A two-sided p≤0.05 was considered nominally statistically significant.
Results
Among the 1303 Caucasian subjects, 169 (13%) were Hp genotype 1-1, 618 (47%) were Hp 2-1, and 516 (40%) were Hp 2-2, conforming to Hardy-Weinberg equilibrium (p=0.92). Baseline associations comparing Hp 2-2 vs. non-Hp 2-2 study participants are presented in Table 1. There were no significant differences in baseline characteristics between Hp 2-2 and non-Hp 2-2.
Table 1.
Baseline Characteristics by Hp genotype
| HP 1-1 or HP 2-1 (N=787) |
HP 2-2 (N=516) |
p-value* | |
|---|---|---|---|
| Intensive treatment group | 399 (51) | 237 (46) | 0.092 |
| Age (years) | 27 ± 7 | 27 ± 7 | 0.667 |
| BMI (kg/m2) | 23.4 ± 2.8 | 23.4 ± 2.8 | 0.730 |
| Duration of type 1 diabetes in months | 66 ± 49 | 70 ± 51 | 0.259 |
| Female | 379 (48) | 230 (45) | 0.205 |
| Baseline HbA1c (%) | 8.8 ± 1.5 | 8.9 ± 1.6 | 0.104 |
| Baseline HbA1c (mmol/mol) | 72.7 ± 16.9 | 74.3 ± 17.6 | 0.104 |
| Primary cohort | 402 (51) | 249 (48) | 0.319 |
| Clinical neuropathy = yes | 48 (6) | 32 (6) | 0.958 |
| Smoker = yes | 145 (18) | 101 (20) | 0.604 |
| LDL (mg/dL) | 108 ± 29 | 111 ± 30 | 0.082 |
| HDL (mg/dL) | 50 ± 12 | 50 ± 12 | 0.652 |
| Triglycerides (mg/dL) | 82 ± 49 | 82 ± 48 | 0.682 |
| Cholesterol (mg/dL) | 175 ± 33 | 178 ± 35 | 0.132 |
| Blood pressure (mm Hg) | |||
| Systolic | 114 ± 12 | 115 ± 11 | 0.171 |
| Diastolic | 73 ± 9 | 72 ± 9 | 0.990 |
| Estimated Glomerular Filtration Rate (mL/min/1.73m2) |
125.5 ± 14.0 | 126.0 ± 13.3 | 0.441 |
| Albumin excretion rate (mg/24 h) | 15.8 ± 17.3 | 15.9 ± 20.8 | 0.862 |
Data are N (%) and mean ± SD.
Wilcoxon for continuous variables and chi-square test for categorical variables.
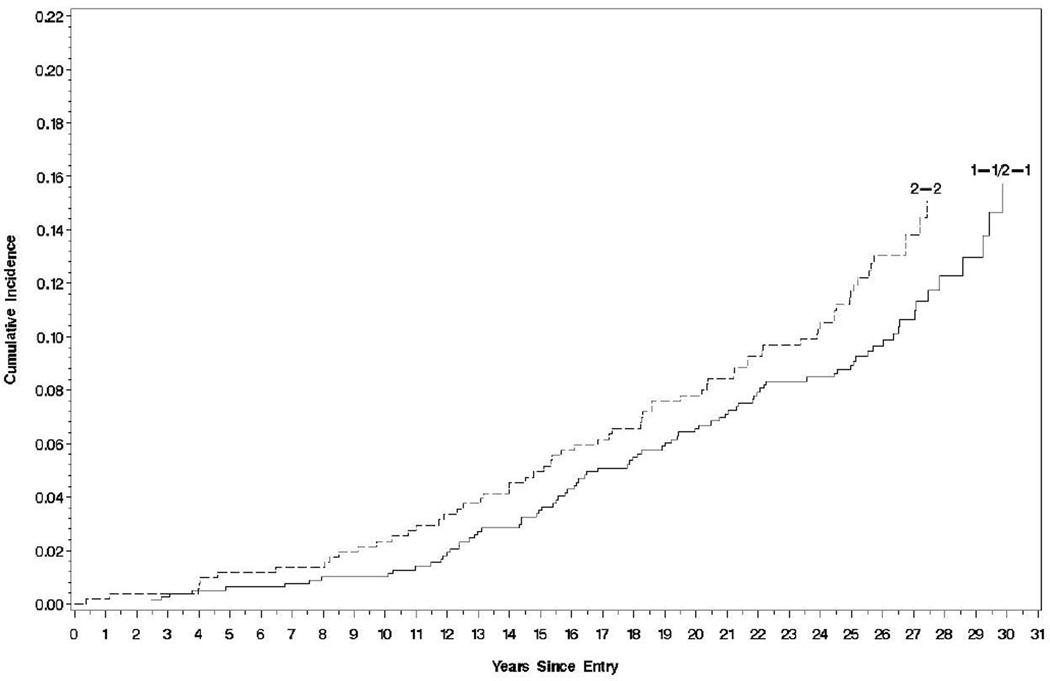
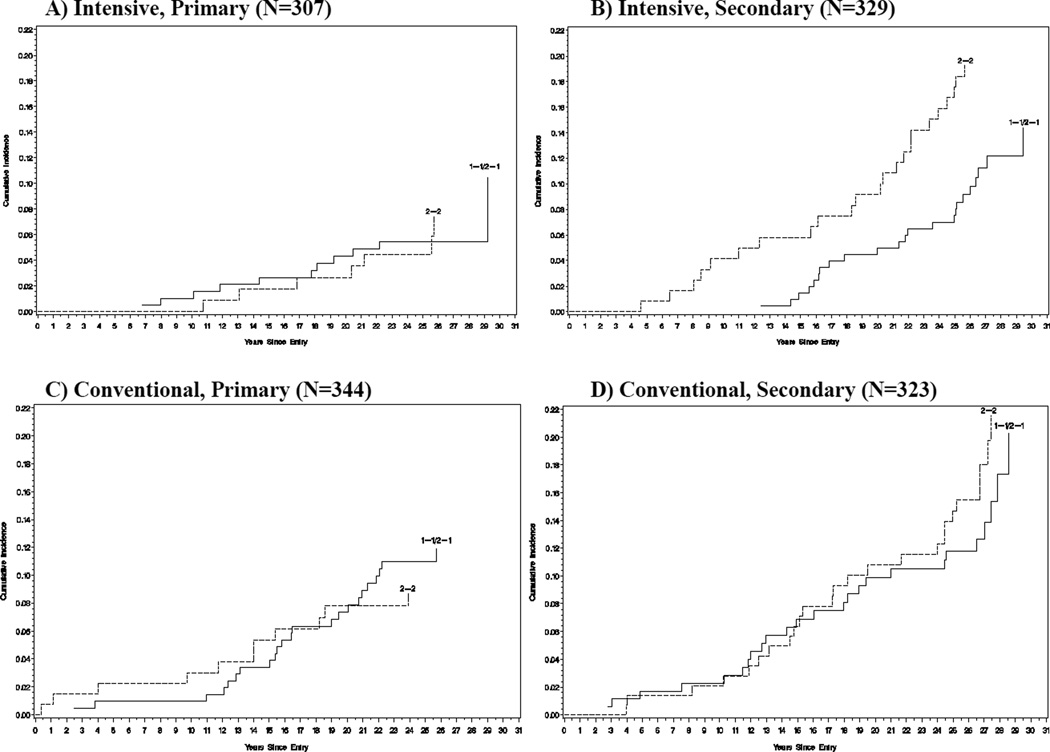
Table 2 shows the overall number and proportion of participants with the Hp 2-2 versus non Hp 2-2 genotype who experienced any CAD event (n=149) or MI/silent MI (n=89) during a mean of 26 years of follow-up in the DCCT/EDIC. Figure 1 shows a small but consistent difference between the time to the first CAD event among Hp 2-2 carriers and non-Hp 2-2 genotype carriers that was, however, non-significant (log-rank test p=0.205). Figure 2 explores this difference in time to the first CAD event further within treatment groups and study cohorts. Little difference was seen within the conventional treatment group for CAD in either the primary (p=0.49) or secondary cohort (p=0.53). However, in the intensive group, a borderline significant difference in CAD risk was seen between Hp genotypes in the secondary cohort (p=0.049 by the log-rank test), but not in the primary cohort (p=0.99). Thus, two-way interaction terms between the Hp genotype and either treatment group or study cohort on CAD were non-significant (p=0.17 for both interactions, Table 2), while a three-way interaction among Hp genotype, treatment group, and study cohort was nominally significant (p=0.049, Table 2), suggesting that the association between Hp and CAD incidence differed within subgroups of intensive vs. conventional therapy and primary vs. secondary cohort.
Table 2.
Cox Proportional Hazards models of CAD and of MI/silent MI for Haptoglobin 2-2 genotype versus Haptoglobin 1-1/2-1 genotype, both unadjusted and adjusted for other factors.
| A. Overall | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| CAD | |||||||||
| Unadjusted |
Adjusted for covariates* and treatment group |
Adjusted for covariates* and study cohort |
Adjusted for covariates* and treatment group and lipid-lowering medication† |
||||||
| N(%) |
Hazard Ratio (95% C.I.) |
p-value |
Hazard Ratio (95% C.I.) |
p-value |
Hazard Ratio (95% C.I.) |
p-value |
Hazard Ratio (95% C.I.) |
p-value | |
| Total | 149(11.4) | ||||||||
| HP 1-1/HP 2-1 | 83(10.6) | 1 Reference | 1 Reference | 1 Reference | 1 Reference | ||||
| HP 2-2 | 66(12.8) | 1.23(0.89, 1.70) | 0.205 | 1.17(0.85, 1.63) | 0.338 | 1.20(0.86, 1.66) | 0.282 | 1.18(0.85, 1.63) | 0.335 |
| HP Interaction by group or cohort |
0.169 | 0.169 | |||||||
| Group by Cohort by HP 3 way interaction |
0.049 | ||||||||
| MI/Silent MI | |||||||||
| Unadjusted |
Adjusted for covariates* and treatment group |
Adjusted for covariates* and study cohort |
Adjusted for covariates* and treatment group and lipid-lowering medication† |
||||||
| N(%) |
Hazard Ratio (95% C.I.) |
p-value |
Hazard Ratio (95% C.I.) |
p-value |
Hazard Ratio (95% C.I.) |
p-value |
Hazard Ratio (95% C.I.) |
p-value | |
| Total | 89(6.8) | ||||||||
| HP 1-1/HP 2-1 | 45(5.7) | 1 Reference | 1 Reference | 1 Reference | 1 Reference | ||||
| HP 2-2 | 44(8.5) | 1.51(0.99, 2.28) | 0.054 | 1.42(0.93, 2.16) | 0.106 | 1.43(0.94, 2.18) | 0.093 | 1.43(0.94, 2.17) | 0.099 |
| HP Interaction by group or cohort |
0.519 | 0.077 | |||||||
| Group by Cohort by HP 3 way interaction |
0.073 | ||||||||
| B. by Treatment Group | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| CAD | ||||||||||
| Intensive Treatment Group | Conventional Treatment Group | |||||||||
| Unadjusted | Adjusted for covariates* | Unadjusted | Adjusted for covariates* | |||||||
| N(%) |
Hazard Ratio (95% C.I.) |
p-value |
Hazard Ratio (95% C.I.) |
p-value | N(%) |
Hazard Ratio (95% C.I.) |
p-value |
Hazard Ratio (95% C.I.) |
p-value | |
| N | 64(10.1) | 85(12.7) | ||||||||
| HP 1-1 /HP 2-1 |
34(8.5) | 1 Reference | 1 Reference | 49(12.6) | 1 Reference | 1 Reference | ||||
| HP 2-2 | 30(12.7) | 1.48(0.91, 2.42) | 0.115 | 1.43(0.88, 2.36) | 0.152 | 36(12.9) | 1.05(0.68, 1.61) | 0.831 | 0.99(0.63, 1.54) | 0.947 |
| MI/Silent MI | ||||||||||
| Intensive Treatment Group | Conventional Treatment Group | |||||||||
| Unadjusted | Adjusted for covariates* | Unadjusted | Adjusted for covariates* | |||||||
| N(%) |
Hazard Ratio (95% C.I.) |
p-value |
Hazard Ratio (95% C.I.) |
p-value | N(%) |
Hazard Ratio (95% C.I.) |
p-value |
Hazard Ratio (95% C.I.) |
p-value | |
| N | 38(6.0) | 51(7.6) | ||||||||
| HP 1-1 /HP 2-1 |
19(4.8) | 1 Reference | 1 Reference | 26(6.7) | 1 Reference | 1 Reference | ||||
| HP 2-2 | 19(8.0) | 1.67(0.88, 3.15) | 0.116 | 1.62(0.85, 3.10) | 0.143 | 25(9.0) | 1.37(0.79, 2.38) | 0.258 | 1.25(0.71, 2.20) | 0.433 |
| C. by Study Cohort | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| CAD | ||||||||||
| Primary Prevention Cohort | Secondary Prevention Cohort | |||||||||
| Unadjusted | Adjusted for covariates* | Unadjusted | Adjusted for covariates* | |||||||
| N(%) |
Hazard Ratio (95% C.I.) |
p-value |
Hazard Ratio (95% C.I.) |
p-value | N(%) |
Hazard Ratio (95% C.I.) |
p-value |
Hazard Ratio (95% C.I.) |
p-value | |
| N | 52(8.0) | 97(14.9) | ||||||||
| HP 1-1 /HP 2-1 |
34(8.5) | 1 Reference | 1 Reference | 49(12.7) | 1 Reference | 1 Reference | ||||
| HP 2-2 | 18(7.2) | 0.86(0.48, 1.52) | 0.594 | 0.83(0.46, 1.49) | 0.526 | 48(18.0) | 1.47(0.99, 2.18) | 0.059 | 1.45(0.97, 2.18) | 0.070 |
| MI/Silent MI | ||||||||||
| Primary Prevention Cohort | Secondary Prevention Cohort | |||||||||
| Unadjusted | Adjusted for covariates* | Unadjusted | Adjusted for covariates* | |||||||
| N(%) |
Hazard Ratio (95% C.I.) |
p-value |
Hazard Ratio (95% C.I.) |
p-value | N(%) |
Hazard Ratio (95% C.I.) |
p-value |
Hazard Ratio (95% C.I.) |
p-value | |
| N | 30(4.6) | 59(9.0) | ||||||||
| HP 1-1 /HP 2-1 |
20(5.0) | 1 Reference | 1 Reference | 25(6.5) | 1 Reference | 1 Reference | ||||
| HP 2-2 | 10(4.0) | 0.80(0.38, 1.72) | 0.572 | 0.78(0.36, 1.70) | 0.529 | 34(12.7) | 2.02(1.20, 3.38) | 0.008 | 1.91(1.13, 3.22) | 0.016 |
| D. by Treatment Group and Study Cohort (Unadjusted) | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| CAD | ||||||||||||
| Intensive Treatment Group | Conventional Treatment Group | |||||||||||
|
Primary Prevention Cohort, Unadjusted |
Secondary Prevention Cohort, Unadjusted |
Primary Prevention Cohort, Unadjusted |
Secondary Prevention Cohort, Unadjusted |
|||||||||
| N(%) |
Hazard Ratio (95% C.I.) |
p-value | N(%) |
Hazard Ratio (95% C.I.) |
p-value | N(%) |
Hazard Ratio (95% C.I.) |
p-value | N(%) |
Hazard Ratio (95% C.I.) |
p-value | |
| N | 18(5.9) | 46(14.0) | 34(9.9) | 51(15.8) | ||||||||
| HP 1-1 /HP 2-1 |
11(5.7) | 1 Reference | 23(11.1) | 1 Reference | 23(11.0) | 1 Reference | 26(14.6) | 1 Reference | ||||
| HP 2-2 | 7(6.1) | 1.01(0.39, 2.59) | 0.992 | 23(18.9) | 1.78(1.00, 3.17) | 0.052 | 11(8.2) | 0.78(0.38, 1.59) | 0.487 | 25(17.2) | 1.19(0.69, 2.07) | 0.528 |
| MI/Silent MI | ||||||||||||
| Intensive Treatment Group | Conventional Treatment Group | |||||||||||
|
Primary Prevention Cohort, Unadjusted |
Secondary Prevention Cohort, Unadjusted |
Primary Prevention Cohort, Unadjusted |
Secondary Prevention Cohort, Unadjusted |
|||||||||
| N(%) |
Hazard Ratio (95% C.I.) |
p-value | N(%) |
Hazard Ratio (95% C.I.) |
p-value | N(%) |
Hazard Ratio (95% C.I.) |
p-value | N(%) |
Hazard Ratio (95% C.I.) |
p-value | |
| N | 11(3.6) | 27(8.2) | 19(5.5) | 32(9.9) | ||||||||
| HP 1-1 /HP 2-1 |
8(4.2) | 1 Reference | 11(5.3) | 1 Reference | 12(5.7) | 1 Reference | 14(7.9) | 1 Reference | ||||
| HP 2-2 | 3(2.6) | 0.59(0.16, 2.22) | 0.435 | 16(13.1) | 2.56(1.19, 5.53) | 0.017 | 7(5.2) | 0.95(0.37, 2.41) | 0.912 | 18(12.4) | 1.60(0.79, 3.21) | 0.189 |
Unadjusted PH models assessed the difference in HP 2-2 versus 1-1 and 2-1 combined without adjustment for other factors. Baseline covariates employed in adjusted models were gender, DCCT baseline age, duration, BMI, HbA1c, LDL, systolic blood pressure, smoking, triglycerides, albumin excretion rate, glomerular filtration rate, and three principal components of ancestry.
Lipid lowering medication is a time-dependent covariate.
Figure 1.
Time to the first CAD event by haptoglobin genotype Hp 2-2 vs. non-HP 2-2 (Log-rank test p-value, p=0.205).
Figure 2.
Time to the first CAD event by haptoglobin genotype Hp 2-2 (dashed lines) vs. non-Hp 2-2 (solid lines), stratified by intensive vs. conventional treatment group and primary vs. secondary cohort. Number of observations and log-rank test p-value are: A) Intensive and Primary, N=307, p=0.992. B) Intensive and Secondary, N=329, p=0.049. C) Conventional and Primary, N=344, p=0.486. D) Conventional and Secondary, N=323, p=0.528.
A borderline non-significant difference between Hp genotypes was observed for MI/silent MI alone (p=0.054) overall (Table 2), while the unadjusted HR was significantly increased for Hp 2-2 compared to non-Hp 2-2 in the secondary cohort (HR=2.02, CI: 1.28, 3.38, p=0.008). A two-way interaction between the Hp genotype and treatment group was not significant for the outcome of MI/silent MI (p=0.52), while the two-way interaction between Hp and study cohort as well as the three-way interaction among Hp genotype, treatment group, and study cohort were borderline non-significant (p=0.08 and p=0.07, respectively), suggesting that the effect of Hp on the incidence of MI/Silent MI may differ in those in the primary vs. secondary cohort as well as within treatment/cohort subgroups.
Adjusted Cox Proportional Hazards models are also shown in Table 2. There was little effect when adjusting for key covariates, which include albumin excretion rate and estimated glomerular filtration rate, and/or treatment group or cohort in the overall CAD analyses. Similarly, the associations observed within the cohort and treatment subgroups remained essentially unaltered after covariate adjustment. For the outcome of MI/silent MI, in the entire study population, adjustment for covariates reduced the HR for Hp 2-2 vs. non-Hp 2-2 from 1.51 (0.99, 2.28) to 1.42 (0.93, 2.16) while in the secondary cohort, the HR for MI/silent MI, remained significantly elevated at 1.91 (1.13, 3.22 p=0.016).
Discussion
The Pittsburgh EDC study demonstrated a significantly increased risk of CAD events in those carrying the Hp 2-2 vs. the Hp 1-1 genotype, with the fully adjusted HR of 2.21 (95% CI.1.05, 4.65, p=0.04) (17). However, in this analysis of the Caucasian DCCT/EDIC cohort, we failed to show a significantly increased risk of CAD events in those carrying the Hp 2-2 genotype. The proportion of cases by specific Hp genotype in DCCT/EDIC was too small to permit separate analyses by the three Hp genotypes as was done in EDC, where the proportion developing events was nearly three times as high (30% vs. 11% for CAD). However, the risk of CAD appeared somewhat increased in those Hp 2-2 DCCT/EDIC participants randomized to intensive therapy (HR 1.48; 0.91, 2.42) and in those recruited into the secondary prevention cohort (HR 1.47; 0.99, 2.18), where the majority of events occurred. The Hp 2-2 effect also appeared stronger for MI (HR 1.51; 0.99, 2.28) than for total CAD, particularly for those in the secondary intervention cohort where a highly significant association was evident (HR 2.02; 1.20, 3.38 p=0.008).
Clearly the overall results lack statistical significance and are not confirmatory of the Hp 2-2 association for all CAD. However, it should be noted that in the EDC cohort, the Hp 2-2 genotype was associated with a significantly increased 36% in the incidence of CAD in type 1 individuals, somewhat lower than the 47% borderline significant increase seen in the current report for the DCCT/EDIC secondary cohort. Furthermore, given the overall lower event rate in DCCT/EDIC (11% vs. 30%), the current analysis had only 45% power to detect a statistically significant increase in CAD in participants with the Hp 2-2 genotype similar to the magnitude seen in EDC.
A critical difference between the EDC and DCCT/EDIC cohorts is the diabetes duration at baseline, which was 19 years in EDC compared to only 6 years in DCCT/EDIC. Participants in DCCT/EDIC are only now reaching the duration of diabetes that resulted in the association between Hp genotype and CAD incidence previously demonstrated in the EDC type 1 diabetes mellitus study. A stronger association was observed in the secondary DCCT/EDIC cohort, albeit only in the intensive treatment group, in which participants had a longer duration of diabetes and more advanced disease, and were thus more akin to the EDC population. This perhaps lends credence as to why the analysis of the entire DCCT/EDIC population failed to confirm the Hp 2-2 association with CAD. In addition, it should be noted that, in contrast to EDC, the DCCT/EDIC study excluded those at high risk of CAD at baseline.
The current findings concerning treatment group differences are of interest and require further exploration. The risk of CAD associated with Hp 2-2 seen in the intensively treated group (HR 1.48 (0.91–2.42), p=0.11) appears greater than that in the conventionally treated group (HR 1.05; 0.68, 1.61, p=0.83). Though this difference is clearly not significant, there was a nominally significant decrease in the time to a first CAD event in Hp 2-2 vs. non-Hp 2-2 (p=0.049 by log-rank analysis) in the intensively treated secondary cohort. A number of potential explanations of these findings arise.
First, it maybe that the weaker (relative to glycemic) Hp 2-2 effect can be observed only in those on intensive therapy, in whom the glycemic related risk is reduced. This would primarily be seen, as observed, in the secondary cohort where the majority of events occurred. Another potential explanation is that intensive treatment had no effect on the event rate in persons at genetically high risk by virtue of having Hp 2-2 (12.7% with intensive vs.12.9% with conventional treatment) but did have a borderline significant effect in non-Hp 2-2 individuals (8.5% with intensive vs. with 12.6% conventional treatment, p=0.06).
It may also be possible that intensive treatment initiated very early on in the natural history of type 1 diabetes (as in the primary prevention cohort) provides greater benefits to those genetically susceptible to complication development (i.e. Hp 2-2). In contrast, starting intensive therapy post manifestation of early complications (as in the secondary intervention cohort) may not benefit these individuals as much, thus allowing the increased risk associated with Hp 2-2 to become apparent. Nevertheless, as the current study was not sufficiently powered to detect an interaction between Hp genotype and treatment group (study power of less than 20%), these results need confirmation and no conclusions concerning differential effects of diabetes therapy by Hp genotype can be drawn at this time.
Finally, the finding that the relationship between the Hp genotype and CAD is stronger when assessing myocardial infarction as the outcome rather than using a broader definition of CAD is supported by mechanistic data, including evidence that Hp genotype stimulates opposing responses to intraplaque hemorrhage (35), a recognized event immediately prior to plaque rupture and myocardial infarction (36). These differences suggest that the likelihood of plaque rupture and myocardial infarction is higher in Hp 2-2, a hypothesis which is supported by studies in Hp 2-2 transgenic mice.
A limitation of these analyses is the small number of incident events, especially within the primary cohort and the intensively treated group, severely limiting our power to detect statistically significant associations. Nevertheless, the effect size observed for an Hp – CAD association in the secondary cohort, which more closely resembles the EDC population, was of the same direction and of greater magnitude than that previously reported within the EDC study. The small event number further affected our ability to conduct multivariable analyses when stratifying by both study cohort and treatment group (Table 2D). However, as no differences in baseline risk factors were observed by Hp genotype (Table 1), it is not clear that such adjustment would greatly alter study findings.
In conclusion, this DCCT/EDIC analysis does not statistically confirm an overall association between Hp 2-2 and incident CAD. However, subgroup analyses in the DCCT/EDIC study suggest that increased CAD risk in the Hp 2-2 vs. non-Hp 2-2 groups may be stronger in those recruited into the secondary cohort, those randomized to intensive therapy and for the harder outcome of myocardial infarction. These findings should be explored in other cohorts.
Highlights.
We investigated the association between the Hp 2-2 genotype and CAD incidence in the DCCT/EDIC cohort.
Hp 2-2 was not significantly associated with incident CAD.
An increased MI risk was suggested in the secondary cohort for those with Hp 2-2.
Acknowledgments
Industry contributors have had no role in the DCCT/EDIC study but have provided free or discounted supplies or equipment to support participants’ adherence to the study: Abbott Diabetes Care (Alameda, CA), Animas (Westchester, PA), Bayer Diabetes Care (North America Headquarters, Tarrytown, NY), Becton Dickinson (Franklin Lakes, NJ), Eli Lilly (Indianapolis, IN), Extend Nutrition (St. Louis, MO), Insulet Corporation (Bedford, MA), Lifescan (Milpitas, CA), Medtronic Diabetes (Minneapolis, MN), Nipro Home Diagnostics (Ft. Lauderdale, FL), Nova Diabetes Care (Billerica, MA), Omron (Shelton, CT), Perrigo Diabetes Care (Allegan, MI), Roche Diabetes Care (Indianapolis, IN), and Sanofi-Aventis (Bridgewater NJ).
The authors acknowledge the data processing and technical assistance of Wanyu Hsu at the Biostatistics Center, the George Washington University.
Funding/Support: The DCCT/EDIC has been supported by cooperative agreement grants (1982–1993, 2012–2017), and contracts (1982–2012) with the Division of Diabetes Endocrinology and Metabolic Diseases of the National Institute of Diabetes and Digestive and Kidney Disease (current grant numbers U01 DK094176 and U01 DK094157), and through support by the National Eye Institute, the National Institute of Neurologic Disorders and Stroke, the General Clinical Research Centers Program (1993–2007), and Clinical Translational Science Center Program (2006-present), Bethesda, Maryland, USA.
Haptoglobin genotyping was supported by NIH RO1DK085226-03, Israel Science Foundation and the Rappaport Institute for Medical Sciences.
Conflict of Interest Disclosures: TJO consultant (Advisory Board) for Eli Lilly Inc. and Profil Institute for Scientific Research. APL’s institution, Israel Institute of Technology, Haifa, Israel, owns a patent which claims that haptoglobin genotype can predict risk of diabetes complications.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
Trial Registration: clinicaltrials.gov NCT00360815 and NCT00360893.
Author Contributions: Dr Lachin had full access to all of the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
T.J.O. researched the data, assisted with analysis and wrote the manuscript; J-Y.C.B. analyzed the data and wrote the manuscript; T.C. assisted with analysis and wrote the manuscript; P.C. reviewed/edited the manuscript; M.L.V. reviewed/edited the manuscript; A.P.L. researched the data, assisted with analysis and wrote the manuscript; J.M.L. researched the data, assisted with analysis and reviewed/edited the manuscript.
References
- 1.Dabelea D, Kinney G, Snell-Bergeon JK, Hokanson JE, Eckel RH, Ehrlich J, Garg S, Hamman RF, Rewers M Coronary Artery Calcification in Type 1 Diabetes Study. Effect of type 1 diabetes on the gender difference in coronary artery calcification: a role for insulin resistance? The Coronary Artery Calcification in Type 1 Diabetes (CACTI) Study. Diabetes. 2003;52:2833–2839. doi: 10.2337/diabetes.52.11.2833. Erratum in: Diabetes 2004;53:2177. [DOI] [PubMed] [Google Scholar]
- 2.Guy J, Ogden L, Wadwa RP, Hamman RF, Mayer-Davis EJ, Liese AD, D'Agostino R, Jr, Marcovina S, Dabelea D. Lipid and lipoprotein profiles in youth with and without type 1 diabetes: the SEARCH for Diabetes in Youth case-control study. Diabetes Care. 2009;3:416–420. doi: 10.2337/dc08-1775. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Nathan DM, Cleary PA, Backlund JY, Genuth SM, Lachin JM, Orchard TJ, Raskin P, Zinman B Diabetes Control and Complications Trial/Epidemiology of Diabetes Interventions and Complications (DCCT/EDIC) Study Research Group. Intensive diabetes treatment and cardiovascular disease in patients with type 1 diabetes. N Engl J Med. 2005;353:2643–2653. doi: 10.1056/NEJMoa052187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Secrest AM, Becker DJ, Kelsey SF, Laporte RE, Orchard TJ. Cause-specific mortality trends in a large population-based cohort with long-standing childhood-onset type 1 diabetes. Diabetes. 2010 Dec;59(12):3216–3222. doi: 10.2337/db10-0862. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Langlois MR, Delanghe JR. Biological and clinical significance of haptoglobin polymorphism in humans. Clin Chem. 1996;42:1589–1600. [PubMed] [Google Scholar]
- 6.Wejman JC, Hovsepian D, Wall JS, Hainfeld JF, Greer J. Structure and assembly of haptoglobin polymers by electron microscopy. J Mol Biol. 1984;174:343–368. doi: 10.1016/0022-2836(84)90342-5. [DOI] [PubMed] [Google Scholar]
- 7.Kristiansen M, Graversen JH, Jacobsen C, Sonne O, Hoffman HJ, Law SK, Moestrup SK. Identification of the haemoglobin scavenger receptor. Nature. 2001;409(6817):198–201. doi: 10.1038/35051594. [DOI] [PubMed] [Google Scholar]
- 8.Asleh R, Marsh S, Shilkrut M, Binah O, Guetta J, Lejbkowicz F, Enav B, Shehadeh N, Kanter Y, Lache O, Cohen O, Levy NS, Levy AP. Genetically determined heterogeneity in hemoglobin scavenging and susceptibility to diabetic cardiovascular disease. Circ Res. 2003;92:1193–1200. doi: 10.1161/01.RES.0000076889.23082.F1. [DOI] [PubMed] [Google Scholar]
- 9.Asleh R, Guetta J, Kalet-Litman S, Miller-Lotan R, Levy AP. Haptoglobin genotype and diabetes dependent differences in iron mediated oxidative stress in vitro and in vivo. Circ Res. 2005;96:435–441. doi: 10.1161/01.RES.0000156653.05853.b9. [DOI] [PubMed] [Google Scholar]
- 10.Asleh R, Miller-Lotan R, Aviram M, Hayek T, Yulish M, Levy JE, Miller B, Blum S, Milman U, Shapira C, Levy AP. Haptoglobin genotype is a regulator of reverse cholesterol transport in diabetes in vitro and in vivo. Circ Res. 2006;99:1419–1425. doi: 10.1161/01.RES.0000251741.65179.56. [DOI] [PubMed] [Google Scholar]
- 11.Levy AP, Purushothaman KR, Levy NS, Purushothaman M, Strauss M, Asleh R, Marsh S, Cohen O, Moestrup SK, Moller HJ, Zias EA, Benhayon D, Fuster V, Moreno PR. Downregulation of the hemoglobin scavenger receptor in individuals with diabetes and the Hp 2-2 genotype: implications for the response to intraplaque hemorrhage and plaque vulnerability. Circ Res. 2007;101:106–110. doi: 10.1161/CIRCRESAHA.107.149435. [DOI] [PubMed] [Google Scholar]
- 12.Asleh R, Blum S, Kalet-Litman S, Alshiek J, Miller-Lotan R, Asaf R, Rock W, Aviram M, Milman U, Shapira C, Abassi Z, Levy AP. Correction of HDL dysfunction in individuals with diabetes and the haptoglobin 2-2 genotype. Diabetes. 2008;57:2794–2800. doi: 10.2337/db08-0450. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Levy AP, Hochberg I, Jablonski K, Resnick HE, Lee ET, Best L, Howard BV Strong Heart Study. Haptoglobin phenotype is an independent risk factor for cardiovascular disease in individuals with diabetes: The Strong Heart Study. J Am Coll Cardiol. 2002;40(11):1984–1990. doi: 10.1016/s0735-1097(02)02534-2. [DOI] [PubMed] [Google Scholar]
- 14.Suleiman M, Aronson D, Asleh R, Kapeliovich MR, Roguin A, Meisel SR, Shochat M, Sulieman A, Reisner SA, Markiewicz W, Hammerman H, Lotan R, Levy NS, Levy AP. Haptoglobin polymorphism predicts 30-day mortality and heart failure in patients with diabetes and acute myocardial infarction. Diabetes. 2005;54:2802–2806. doi: 10.2337/diabetes.54.9.2802. [DOI] [PubMed] [Google Scholar]
- 15.Roguin A, Koch W, Kastrati A, Aronson D, Schomig A, Levy AP. Haptoglobin genotype is predictive of major adverse cardiac events in the 1-year period after percutaneous transluminal coronary angioplasty in individuals with diabetes. Diabetes Care. 2003;26:2628–2631. doi: 10.2337/diacare.26.9.2628. [DOI] [PubMed] [Google Scholar]
- 16.Ijäs P, Saksi J, Soinne L, Tuimala J, Jauhiainen M, Jula A, Kähönen M, Kesäniemi YA, Kovanen PT, Kaste M, Lindsberg PJ. Haptoglobin 2 allele associates with unstable carotid plaque and major cardiovascular events. Atherosclerosis. 2013 Oct;230(2):228–234. doi: 10.1016/j.atherosclerosis.2013.07.008. [DOI] [PubMed] [Google Scholar]
- 17.Costacou T, Ferrell R, Orchard TJ. Haptoglobin genotype: a determinant of cardiovascular complication risk in type 1 diabetes. Diabetes. 2008;57:1702.42–1706.42. doi: 10.2337/db08-0095. [DOI] [PubMed] [Google Scholar]
- 18.Costacou T, Ferrell RE, Ellis D, Orchard TJ. Haptoglobin genotype and renal function decline in type 1 diabetes. Diabetes. 2009;58:2904–2909. doi: 10.2337/db09-0874. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Orchard TJ, Sun W, Cleary PA, Genuth SM, Lachin JM, McGee P, Paterson AD, Raskin P, Anbinder Y, Levy AP DCCT/EDIC Research Group. Haptoglobin genotype and the rate of renal function decline in the Diabetes Control and Complications Trial/Epidemiology of Diabetes Interventions and Complications study. Diabetes. 2013;62(9):3218–3223. doi: 10.2337/db13-0256. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Costacou T, Orchard TJ. The Haptoglobin genotype predicts cardio-renal mortality in type 1 diabetes. J Diabetes Complications. 2016;30(2):221–226. doi: 10.1016/j.jdiacomp.2015.11.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.The DCCT/EDIC Research Group. Intensive diabetes therapy and glomerular filtration rate in type 1 diabetes. NEJM. 2011;365:2366–2376. doi: 10.1056/NEJMoa1111732. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.The Diabetes Control and Complications Trial (DCCT) Design and methodologic considerations for the feasibility phase. The DCCT Research Group. Diabetes. 1986;35(5):530–545. [PubMed] [Google Scholar]
- 23.EDIC Research Group. Epidemiology of Diabetes Interventions and Complications (EDIC): Design and implementation of a long-term follow-up of the Diabetes Control and Complications Trial Cohort. Diabetes Care. 1999;22:99–111. doi: 10.2337/diacare.22.1.99. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Patterson N, Price AL, Reich D. Population Structure and Eigenanalysis. PLoS Genet. 2006;2(12):e190. doi: 10.1371/journal.pgen.0020190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Frazer KA, Ballinger DG, Cox DR, Hinds DA, Stuve LL, Gibbs RA, Belmont JW, Boudreau A, et al. A second generation human haplotype map of over 3.1 million SNPs. Nature. 2007;449:851–861. doi: 10.1038/nature06258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Goldstein DE, Soeldner S, Cleary PA, Nathan DM. Feasibility of centralized measurements of glycated hemoglobin in the Diabetes Control and Complications Trial (DCCT): A multicenter trial. Clin Chem. 1987;33:2267–2271. [PubMed] [Google Scholar]
- 27.EDIC Research Group. Effect of intensive therapy on the development and progression of diabetic nephropathy in the Diabetes Control and Complications Trial. idney Int. 1995;47:1703–1720. doi: 10.1038/ki.1995.236. [DOI] [PubMed] [Google Scholar]
- 28.Levy AS, Stevens LA, Schmid CH, et al. A new equation to estimate glomerular filtration rate. Ann Intern Med. 2009;150:604–612. doi: 10.7326/0003-4819-150-9-200905050-00006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Hochberg I, Roguin A, Nikolsky E, Chanderashekhar PV, Cohen S, Levy AP. Haptoglobin phenotype and coronary artery collaterals in diabetic patients. Atherosclerosis. 2002;161:441–446. doi: 10.1016/s0021-9150(01)00657-8. [DOI] [PubMed] [Google Scholar]
- 30.Lachin JM. Biostatistical Methods: The Assessment of Relative Risks. Second. John Wiley and Sons; 2011. [Google Scholar]
- 31.Kalbfleisch JD, Prentice RL. The Statistical Analysis of Failure Time Data. 2nd. New York: John Wiley & Sons; 2002. [Google Scholar]
- 32.Paterson AD, Lopes-Virella MF, Waggott D, Boright AP, Hosseini SM, Carter RE, Shen E, Mirea L, Bharaj B, Sun L, Bull SB The Diabetes Control and Complications Trial/Epidemiology of Diabetes Interventions and Complications Research Group. Genome-wide association identifies the ABO blood group as a major locus associated with serum levels of soluble E-selectin. Atherosclerosis, Thrombosis, and Vascular Biology. 2009;29:1958–1967. doi: 10.1161/ATVBAHA.109.192971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Paterson AD, Waggott D, Boright AP, Hosseini SM, Shen E, Sylvestre MP, Wong I, Bharaj B, Cleary PA, Lachin JM, MAGIC, Below JE, Nicolae D, Cox NJ, Canty AJ, Sun L, Bull SB The Diabetes Control and Complications Trial/Epidemiology of Diabetes Interventions and Complications Research Group. A genome-wide association study identifies a novel major locus for glycemic control in type 1 diabetes, as measured by both A1C and glucose. Diabetes. 2010;59:539–549. doi: 10.2337/db09-0653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.R Core Team. R: A language and environment for statistical computing. Vienna, Austria: R Foundation for Statistical Computing; 2013. [Google Scholar]
- 35.Guetta J, Strauss M, Levy NS, Fahoum L, Levy AP. Haptoglobin genotype modulates the balance of Th1/Th2 cytokines produced by macrophages exposed to free hemoglobin. Atherosclerosis. 2007;91:48–53. doi: 10.1016/j.atherosclerosis.2006.04.032. [DOI] [PubMed] [Google Scholar]
- 36.Levy AP, Moreno PR. Intraplaque hemorrhage. Curr Mol Med. 2006;6:479–488. doi: 10.2174/156652406778018626. [DOI] [PubMed] [Google Scholar]