The cell membrane is always associated with the underlying actin cytoskeleton. During cytokinesis or apoptosis, increased intracellular pressure or destruction of the actin cytoskeleton leads to detachment of the plasma membrane from the cytoskeleton and protrusions of the plasma membrane are passively formed. These membrane protrusions are termed membrane blebs. Until recently, membrane blebs were not intensively studied. However, concrete evidence that many organisms use dynamic membrane blebs as a tool for locomotion has accumulated.1 Furthermore, recent studies revealed that some types of cancer cells adopt large polarized membrane blebs for migration and invasion under certain conditions.2 This mode of cell migration is called “amoeboid” migration. Epithelial-mesenchymal transition (EMT) was assumed to be the primary cause of cancer cells acquiring an invasive phenotype. However, cancer cells in which transcription factors essential for EMT are knocked out also exhibit invasion and metastasis, suggesting the existence of EMT-independent mechanisms of cancer invasion.3 One such EMT-independent invasion mechanism is bleb-based cell migration. Therefore, understanding the molecular mechanisms associated with membrane blebs is biologically and clinically important. However, understanding of the molecular mechanisms involved in the regulation of membrane blebs is still in its infancy.
The cycle of membrane blebs initiates when the plasma membrane detaches from the underlying cytoskeleton. The actin cortex-free membrane passively protrudes due to the intracellular hydrostatic pressure. At the end of the expansion phase, actin filaments start to locally accumulate. Then, actin filaments gradually cover the entire protruded membrane. Finally, myosin II and myosin light chain are recruited to the actin cortex, and membrane blebs retract due to the contractile activity of the acto-myosin cortex (Fig. 1). These processes seem to be quite simple; however, there are many unsolved issues regarding the dynamic interplay between the actin cortex and plasma membrane. Among them, the central question is how reassembly of the actin cortex is initiated at the protruded membrane.
Figure 1.
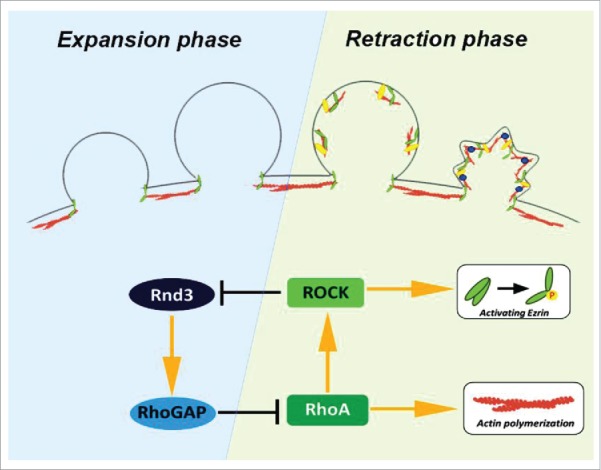
The life cycle of blebs is governed by 2 small GTPases. Rnd3 is preferentially recruited to the actin cortex-free membrane protrusion. During expansion phase, reassembly of actin cortex was actively suppressed by Rnd3-p190BRhoGAP complex and bleb continues to expand passively by the intracellular pressure. Sporadic activation of RhoA is stabilized locally by the phosphorylation of Rnd3 by RhoA-ROCK pathway because phosphorylated Rnd3 is removed from the plasma membrane.
The processes of actin cortex reassembly were first closely examined by Charras et al. in 2006.4 In this seminal paper, they demonstrated that regulators of actin filaments are recruited to the plasma membrane in a staged manner. This study and subsequent studies revealed that epidermal growth factor receptor pathway substrate 8 (Eps8) and ezrin/radixin/moesin (ERM) family proteins rapidly move to the plasma membrane prior to the regrowth of actin filaments at the end of the expansion phase. Eps8 is an actin-capping and actin-bundling protein. ERM family proteins are anchoring proteins that connect the plasma membrane and actin cytoskeleton. ERM family proteins adopt an open form when phosphorylated at Thr567. We recently found that activation of ERM family proteins occurs at the onset of retraction and is essential for the rapid regrowth of the actin cortex.5
Then, what activates the ERM family at the plasma membrane and promotes regrowth of actin filaments only at the retraction phase of membrane blebbing? We recently revealed that two small GTPases are involved in this regulation. One is Rnd3 (also known as RhoE) and the other is RhoA. In the expansion phase, Rnd3 is recruited to the plasma membrane; however, its membrane localization is gradually lost when reassembly of the actin cortex proceeds. On the other hand, activation of RhoA occurs in the retraction phase and coincides with the regrowth of actin filaments. Rnd3 binds to p190RhoGAP and enhance its activity, resulting in the down-regulation of RhoA in the expansion phase. On the contrary, in the retraction phase, RhoA stimulates ROCK (Rho-associated protein kinase) and ROCK phosphorylates Rnd3 at Ser240, which leads to the binding of 14-3-3 protein to Rnd3 and the sequestration of Rnd3 in the cytoplasm.6 Therefore, the interlocked feedback loop between Rnd3 and RhoA is a switch to shift from the expansion phase to the retraction phase. The up-regulation of RhoA activity is necessary not only for the downregulation of Rnd3 activity, but also for the regrowth of actin filaments at the expanded membrane blebs. RhoA activates nucleation of actin filaments by directly binding to formin family proteins, key regulators of actin nucleation, and promotes steady interaction between actin filaments and the plasma membrane by activating ERM family proteins. Previously, it was assumed that the plasma membrane is passively protruded due to the intracellular pressure during the expansion phase. However, our findings suggest that Rnd3 actively suppresses reassembly of the actin cortex to continue the expansion of blebs (Fig. 1).
Then, how Rnd3 is recruited to expanding membrane blebs? Because Rnd3 is recruited to the actin cortex-free membrane in the expansion phase, we examined whether Rnd3 is recruited to the plasma membrane when the actin cytoskeleton is disrupted by treatment with Latrunculin B. Interestingly, localization of Rnd3 at the plasma membrane was enhanced upon treatment with Latrunculin B, suggesting that Rnd3 preferentially localizes to the cortex-free plasma membrane. The underlying molecular mechanisms remain totally unclear; however, there are several possibilities to explain how cells sense the actin cortex-free membrane.
The underlying actin cortex affects many properties of the plasma membrane. Membrane tension reportedly changes during bleb expansion. The changes of membrane tension modulate the activity of small GTPases and mechanosensitive channels and affect membrane localization of the F-BAR domain protein FBP17.7 In addition, loss of the actin cytoskeleton reportedly affects the membrane-associated cytoskeleton and the lateral mobility of lipids and changes lipid bilayer phase behavior. Thus, these changes may be converted to active signals for the recruitment of Rnd3 to the cortex-free membrane. In future studies, it will be very interesting to determine how cells sense the actin cortex-free membrane via mechanical and chemical signals and utilize these signals to repair the actin cortex in order to retract membrane blebs.
Abbreviations
- EMT
epithelial-mesenchymal transition
- ERM
ezrin/radixin/moesin
- Eps8
epidermal growth factor receptor pathway substrate 8
Disclosure of potential conflicts of interest
No potential conflicts of interest were disclosed.
References
- [1].Charras G, Paluch E. Blebs lead the way: how to migrate without lamellipodia. Nat Rev Mol Cell Biol 2008; 9:730-6; PMID:18628785; http://dx.doi.org/ 10.1038/nrm2453 [DOI] [PubMed] [Google Scholar]
- [2].Liu YJ, et al. Confinement and low adhesion induce fast amoeboid migration of slow mesenchymal cells. Cell 2015; 160:659-72; PMID:25679760; http://dx.doi.org/ 10.1016/j.cell.2015.01.007 [DOI] [PubMed] [Google Scholar]
- [3].Zheng X, et al. Epithelial-to-mesenchymal transition is dispensable for metastasis but induces chemoresistance in pancreatic cancer. Nature 2015; 527:525-30; PMID:26560028; http://dx.doi.org/ 10.1038/nature16064 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [4].Charras GT, et al. Reassembly of contractile actin cortex in cell blebs. J Cell Biol 2006; 175:477-90; PMID:17088428; http://dx.doi.org/ 10.1083/jcb.200602085 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [5].Aoki K, et al. A RhoA and Rnd3 cycle regulates actin reassembly during membrane blebbing. Proc Natl Acad Sci USA 2016; 113(13):E1863-71; PMID: 26976596; http://dx.doi.org/23622247 10.1073/pnas.1600968113 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [6].Riou P, et al.. 14-3-3 proteins interact with a hybrid prenyl-phosphorylation motif to inhibit G proteins. Cell 2013; 153:640-53; PMID:23622247; http://dx.doi.org/ 10.1016/j.cell.2013.03.044 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [7].Tsujita K, et al. Feedback regulation between plasma membrane tension and membrane-bending proteins organizes cell polarity during leading edge formation. Nat Cell Biol 2015; 17:749-58; PMID:25938814; http://dx.doi.org/ 10.1038/ncb3162 [DOI] [PubMed] [Google Scholar]


