Sir, Cutaneous squamous cell carcinoma (CSCC) is the second most common malignant neoplasm of the human skin. The incidence of CSCC is increasing, representing a major medical and economic problem. CSCC is characterized by a marked propensity for invasion and metastatic capacity. The degree of cellular differentiation, tumor thickness, location, and other features have prognostic value. Identification of the pathogenic mechanisms for CSCC could facilitate the treatment and prevention of this cancer.
There is increasing evidence that the ADAMs (a disintegrin and metalloprotease) are differentially expressed in malignant tumors and may, therefore, participate in the pathogenesis of carcinomas. Among proteases, 2 ADAMs (ADAM10 and ADAM17) have been of special interest, due to their characteristics of releasing and activating several ligands for the epidermal growth factor receptor (EGFR)/human EGFR (HER) family of receptors.1 HB-EGF, important substrate of ADAM10 and 17, plays a key role in the transactivation of the EGFR by G protein-coupled receptors.2 Recent studies have indicated that HB-EGF gene expression is significantly elevated in variety of human cancers and its expression level is much higher than those of the other EGFR ligands.3 Interestingly, it was postulated that HB-EGF might promote tumor growth of CSCC.4 In this study, we investigated for the first time the expression and localization of ADAM10 and 17 in different histologic subtypes of CSCC, using immunohistochemical analysis.
The study was approved by the ethical committee of the Catholic University of Korea (DC12TIG10010). Formalin-fixed, paraffin-embedded specimens were used. CSCC can be divided into 3 histological subtypes, according to differentiation. The following histological specimens were examined: CSCC (n = 26) [histological subtypes: well differentiated (n=12); differentiated cells are greater than 75%, moderately differentiated (n = 7); differentiated cells are 25–75%, poorly differentiated (n = 7); differentiated cells are less than 25%]. Immunohistochemical analysis was performed using specific polyclonal antibodies and a streptavidin-peroxidase technique with Dako Kit (DAKO REAL detection system alkaline phosphatase/RED rabbit/mouse, Dako, Cat.No. K5005). The primary antibody to the C-terminus of ADAM10 was from Santa Cruz (Heidelberg, Germany), the ADAM17 antibody from eBioscience (Malden, Netherland). The antibodies were used in the following dilutions: ADAM10 (1:50), and ADAM17 (1:100). Microscopic analysis was performed by 2 independent observers (S. O. and J. R.). The degree of expression was graded semi-quantitatively as follows: −, negative (0%); +, focal (1–20%); ++, moderate (21–50%); and +++, diffuse (>50 %). The ADAM 10, 17 immunoreactivity was also assessed with respect to localization (membranous, cytoplasmic and nuclear). Mann-Whitney test was performed to compare ADM10 and ADAM17 expression between normal and CSCC epidermis, respectively. Kruskal-Wallis test was done to determine the overall difference among groups in terms of ADAM10 and ADAM17, respectively. Post hoc Mann-Whitney test was subsequently performed to reveal difference between each group and normal epidermis after Bonferroni’s correction.
Both ADAM10 (P = 0.045) and ADAM17 (P = 0.012) was shown to be significantly differently expressed between normal and CSCC epidermis, respectively. Regarding ADAM10, the overall difference was demonstrated among groups (P < 0.017), and poorly differentiated CSCC was shown to be different from normal epidermis (P = 0.014). In the present study, no overall difference was shown among groups as for ADAM17 (P = 0.093). In addition, ADAM10 immunoreactivity is not increased in the cytoplasm of well differentiated CSCC tumor islands as compared to the normal epidermis (P = 0.368) (Fig. 1B). However, ADAM10 immunoreactivity is increased in the peripheral margin of tumor islands in well differentiated CSCC as compared to the central portion of tumor islands (Fig. 1A). ADAM10 immunoreactivity is increased in the cytoplasm of moderately differentiated CSCC tumor islands as compared to the normal epidermis (P = 0.021) (Fig. 1B). ADAM10 immunoreactivity is increased in the cytoplasm of poorly differentiated CSCC as compared to the normal epidermis (P = 0.014) (Table 1). Cytoplasmic and nuclear immunoreactivity of ADAM17 is not increased in well differentiated CSCC as compared to the normal epidermis (P = 0.063) (Fig. 1D). Cytoplasmic and nuclear ADAM17 immunoreactivity is increased in the cells of moderately differentiated CSCC (P = 0.015) (Fig. 1E) and poorly differentiated CSCC (P = 0.012) (Fig. 1F) as compared to the normal epidermis (Table 1).
Figure 1.
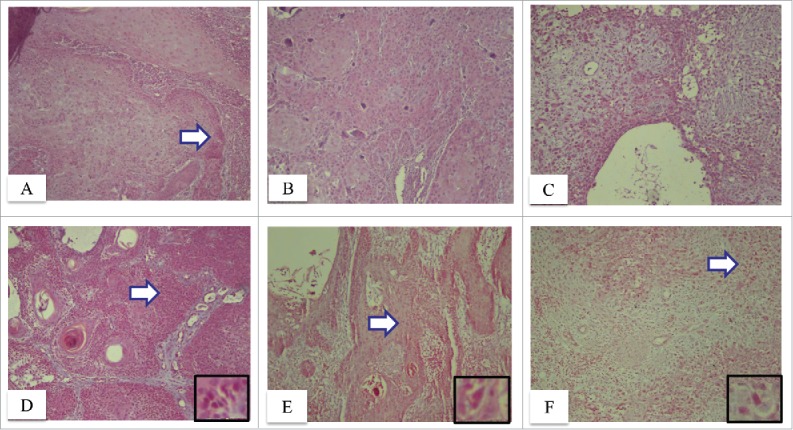
Immunolocalization of ADAM10, ADAM17 in cutaneous squamous cell carcinomas. Note the increased cytoplasmic immunoreactivity of ADAM10 in well differentiated CSCC. Note that ADAM10 immunoreactivity is increased in the peripheral margin of tumor islands in well differentiated CSCC as compared to the central portion of tumor islands (arrow) (A, x100). Note the increased ADAM10 immunoreactivity in the cytoplasm of moderately differentiated CSCC tumor islands (B, x100). Note that ADAM10 immunoreactivity is increased in the cytoplasm of poorly differentiated CSCC (C, x100). Note the increased cytoplasmic and nuclear immunoreactivity of ADAM17 in well differentiated CSCC (D, ×100). The arrow highlights the area magnified in the inset (D, x400). Note the increased cytoplasmic and nuclear ADAM17 immunoreactivity in the cells of moderately differentiated CSCC (E, x100). The arrow highlights the area magnified in the inset (E, x400). Note that ADAM17 immunoreactivity is increased in the nucleus and cytoplasm of poorly differentiated CSCC (F, x100). The arrow highlights the area magnified in the inset (F, x400).
Table 1.
Immunohistochemical expression of ADAM 10, 17 in cutaneous squamous cell carcinoma.
| ADAM 10 |
|||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Immunoreactivity |
|||||||||||
| Variables | 0 | + | ++ | +++ | P-value | Location in the cell | |||||
| Normal epidermis (n = 5) | 2 | 3 | 0 | 0 | Cytoplasm | ||||||
| Differentiation | Well differentiated CSCC (n = 12) | 4 | 4 | 4 | 0 | *0.368 | Cytoplasm | ||||
| Moderately differentiated CSCC (n = 7) | 0 | 3 | 4 | 0 | *0.021 | Cytoplasm | |||||
| Poorly differentiated CSCC (n = 7) | 0 | 2 | 2 | 3 | *0.014 | Cytoplasm | |||||
| Total (n = 26) | 4 | 9 | 10 | 3 | *.0045 | – | |||||
| **0.017 | |||||||||||
| ADAM 17 | |||||||||||
| Immunoreactivity | |||||||||||
| Variables | 0 | + | ++ | +++ | P-value | Location in the cell | |||||
| Normal epidermis (n = 5) | 1 | 4 | 0 | 0 | Cytoplasm | ||||||
| Differentiation | Well differentiated CSCC (n = 12) | 2 | 2 | 4 | 4 | *0.063 | Cytoplasm & Nucleus | ||||
| Moderately differentiated CSCC (n = 7) | 0 | 2 | 5 | 0 | * 0.015 | Cytoplasm & Nucleus | |||||
| Poorly differentiated CSCC (n = 7) | 0 | 2 | 4 | 1 | * 0.017 | Cytoplasm & Nucleus | |||||
| Total (n = 26) | 2 | 6 | 13 | 5 | * 0.012 | – | |||||
| **0.093 | |||||||||||
a; ADAM 10, 17 positivity: +, weakly positive (1–20%); ++, moderately positive (21–50%), and +++, strongly positive (>50 %)
compared with normal epidermis
compared among 3 differentiated CSCC
In the present study, we found the increased ADAM10/17 expression in CSCC. It is well known that ADAM10/17 are overexpressed and play important roles in variety of cancers.5 Therefore it is possible to suspect that ADAM10/17 may play a role in the pathogenesis of CSCC. In addition, we detected increased ADAM10 expression in the peripheral portion as compared to the central portion of CSCC tumor islands. Substrates of ADAM10 include L1 adhesion molecule6 and, CD44.7 They have been characterized as potential inducers of tumor cell migration. Therefore, it is possible to suspect that ADAM10 may play a role in the invasion of CSCC.
In our study, overexpression of ADAM10 was observed in the poorly differentiated CSCC and we found increased ADAM10 expression along the differentiation of CSCC. It has been known that ADAM10 expression was increased in the high grade versus low grade cancer, and ADAM10 contributes to the progression of human prostate cancer.8,9 Therefore, ADAM10 overexpression in the poorly differentiated subtype of CSCC indicates that ADAM10 may play a role in the progression of CSCC. Especially, we observed nuclear ADAM17 expression in all 3 differentiated CSCC. Although its function in the nucleus is still unknown, nuclear ADAM17 may play a pathophysiological role in CSCC. However, further studies are warranted.
Main treatment modality of CSCC is surgery. However, at least in metastatic CSCC, surgery could not be an option. Given the number of anti-ADAM10/17 drugs, which are currently in development, inhibitors of ADAM10 and 17 may be useful, as a novel therapeutic modality in the treatment of metastatic CSCC patients or if surgery is contraindicated or unwilling to undergo surgery.
Our results underline that increased ADAM10 and 17, produced by CSCC cancer cells, might play a critical role in CSCC pathogenesis. The expression of ADAM10 was increased along the grade of tumor histology, which suggests its involvement in tumor progression of CSCC. However, different expression pattern of ADAM10 and 17 indicates that these 2 play a different role in the pathogenesis of CSCC.
Disclosure of potential conflicts of interest
No potential conflicts of interest were disclosed.
References
- [1].Sahin U, Weskamp G, Kelly K, Zhou HM, Higashiyama S, Peschon J, Hartmann D, Saftig P, Blobel CP. Distinct roles for ADAM10 and ADAM17 in ectodomain shedding of six EGFR ligands. J Cell Biol 2004; 164:769-79; PMID:14993236; http://dx.doi.org/ 10.1083/jcb.200307137 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [2].Higashiyama S, Nanba D. ADAM-mediated ectodomain shedding of HB-EGF in receptor cross-talk. Biochim Biophys Acta 2005; 1751:110-7; PMID:16054021 [DOI] [PubMed] [Google Scholar]
- [3].Miyamoto S, Yagi H, Yotsumoto F, Kawarabayashi T, Mekada E. Heparin-binding epidermal growth factor-like growth factor as a novel targeting molecule for cancer therapy. Cancer Sci 2006; 97:341-7; PMID:16630129; http://dx.doi.org/ 10.1111/j.1349-7006.2006.00188.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- [4].Kivisaari AK, Kallajoki M, Ala-aho R, McGrath JA, Bauer JW, Königová R, Medvecz M, Beckert W, Grénman R, Kähäri VM. Matrix metalloproteinase-7 activates heparin-binding epidermal growth factor-like growth factor in cutaneous squamous cell carcinoma. Br J Dermatol 2010; 163:726-35. [DOI] [PubMed] [Google Scholar]
- [5].Saftig P, Reiss K. The “A Disintegrin And Metalloproteases” ADAM10 and ADAM17: novel drug targets with therapeutic potential? Eur J Cell Biol 2011; 90:527-35; PMID:21194787; http://dx.doi.org/ 10.1016/j.ejcb.2010.11.005 [DOI] [PubMed] [Google Scholar]
- [6].Mechtersheimer S, Gutwein P, Agmon-Levin N, Stoeck A, Oleszewski M, Riedle S, Postina R, Fahrenholz F, Fogel M, Lemmon V, et al.. Ectodomain shedding of L1 adhesion molecule promotes cell migration by autocrine binding to integrins. J Cell Biol 2001; 155:661-73; PMID:11706054; http://dx.doi.org/ 10.1083/jcb.200101099 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [7].Reiss K, Maretzky T, Ludwig A, Tousseyn T, de Strooper B, Hartmann D, Saftig P. ADAM10 cleavage of N-cadherin and regulation of cell-cell adhesion and beta-catenin nuclear signalling. EMBO J 2005; 24:742-52; PMID:15692570; http://dx.doi.org/ 10.1038/sj.emboj.7600548 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [8].Arima T, Enokida H, Kubo H, Kagara I, Matsuda R, Toki K, Nishimura H, Chiyomaru T, Tatarano S, Idesako T, et al.. Nuclear translocation of ADAM-10 contributes to the pathogenesis and progression of human prostate cancer. Cancer Sci 2007; 98:1720-6; PMID:17727679; http://dx.doi.org/ 10.1111/j.1349-7006.2007.00601.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- [9].McCulloch DR, Akl P, Samaratunga H, Herington AC, Odorico DM. Expression of the disintegrin metalloprotease, ADAM-10, in prostate cancer and its regulation by dihydrotestosterone, insulin-like growth factor I, and epidermal growth factor in the prostate cancer cell model LNCaP. Clin Cancer Res 2004; 10:314-23; PMID:14734484; http://dx.doi.org/ 10.1158/1078-0432.CCR-0846-3 [DOI] [PubMed] [Google Scholar]


