Abstract
The aim of the present study was to examine the effect of glycitin on the regulation of osteoblasts from bone marrow stem cells (BMSCs) through transforming growth factor (TGF)-β or protein kinase B (AKT) signaling pathways. BMSCs were extracted from New Zealand white rabbits and used to analyze the effect of glycitin on BMSCs. BMSCs were cleared using xylene and observed via light microscopy. BMSCs were subsequently induced with glycitin (0.01, 0.5, 1, 5 and 10 µM) for 7 days, and stained with Oil Red O. The mechanism of action of glycitin on BMSCs was investigated, in which contact with collagen type I (Col I), alkaline phosphatase (ALP), TGF-β and AKT was studied. Firstly, BMSCs appeared homogeneously mazarine blue, and which showed that BMSCs were successful extracted. Administration of glycitin increased cell proliferation and promoted osteoblast formation from BMSCs. Furthermore, glycitin activated the gene expression of Col I and ALP in BMSCs. Notably, glycitin suppressed protein expression of TGF-β and AKT in BMSCs. These results indicated that glycitin may regulate osteoblasts through TGF-β or AKT signaling pathways in BMSCs.
Keywords: glycitin, osteoblast, bone marrow stem cells, transforming growth factor-β 1, protein kinase B
Introduction
Osteanagenesis, which is also known as bone tissue engineering, is a novel and multidisciplinary field (1). By employing the fundamental principles of biology, medical science and tissue engineering, it is possible to remodel injured bone tissue or cure bone diseases (2). Resulting from trauma or physiological and pathological bone resorption, bone defects are a global heath issue, the treatment of which can be challenging. Among craniofacial and plastic surgeries, bone defects caused by trauma are a common clinical problem, and osteanagenesis may be beneficial in repairing injuries and improving the quality of life of patients (3). Osteanagenesis is also used to treat and repair bone mass in various bone diseases caused by gender, age and infection, including osteoporosis, osteopenia and tooth loss as a result of periodontitis (4).
Bone formation is a lengthy and strictly regulated process associated with embryonic development, reconstruction of bone tissue and bone fracture repair (5). Classical bone biology theories claim that mature osteoblasts are formed from BMSCs. During bone formation, bone precursor cells are differentiated into mature osteoblasts that can compound and secrete bone matrix (6), and are subsequently mineralized and imbedded into bone matrix. During the embryonic development process, BMSCs participate in bone formation through membranous ossification and entochondrostosis (7). Membranous ossification predominantly occurs in craniofacial bones, parts of the cartilage and mandibles. During the developmental process of these tissues, BMSCs are able to directly differentiate into osteoblasts, whereas entochondrostosis is employed in the development process of torso and limb bones (8). Initially, through accumulation, proliferation and differentiation into chondrocytes, BMSCs begin to form bones (9). Cartilage cells are then gradually divided into hypertrophic chondrocytes. With the mineralization of deep stromas and in-growth of blood vessels, chondrocytes die and are replaced by osteoblasts, and mature bones are formed.
As a bioactivator, soy isoflavone is a type of flavonoid compound and a secondary metabolite formed during the growth of soybeans (10). Although it is extracted from plants, it has the same structure as estrogen; therefore, it is also known as phytoestrogen. Soy isoflavone can occur in various types, including daidzin, daidzein, genistin, genistein, glycitin and glycitein (11). Glycitin is antibacterial, antiviral and estrogenic (10), and has been demonstrated to have preventative effects on alcoholism, cardiovascular and cerebrovascular diseases and some types of cancer (12,13). It can also reduce the number of tumors and alleviate or avoid macteric syndrome caused by a decrease of estrogen (14), and has demonstrated anti-aging effects (15). The aim of the present study was to investigate whether glycitin regulates osteoblast formation from BMSCs through TGF-β or AKT signaling pathways.
Materials and methods
Isolation and culture of primary BMSCs
A total of 24 healthy male New Zealand white rabbits (age, 6 months; weight, 1–1.5 kg) were purchased from the Experimental Animal Center of Jilin City (Jilin, China). Rabbits were housed at 22–24°C (relative humidity, 55–70%) with natural light and air circulation, and were allowed free access to food and water. All animal procedures were conducted in strict accordance with the Animal Ethical Standard, and the present study was approved by the Experimental Animal Center of Beihua University, Jilin Province Ethics Committee (Jilin, China).
BMSCs were isolated from New Zealand white rabbits using the method described previously by Li et al (14). Initially, the femurs and tibias were removed, and flushed bone marrow cells were acquired via Percoll density gradient centrifugation (1.073 g/ml). Flushed bone marrow cells were washed with phosphate-buffered saline (PBS) and seeded into 25-cm2 cell culture flasks. Flushed bone marrow cells were incubated with L-Dulbecco's modified Eagle medium (DMEM) supplemented with 10% fetal bovine serum and 1% penicillin-streptomycin at 37°C in an atmosphere containing 5% CO2 for 48 h, and subsequently incubated with DMEM for 48 h. Cells were detached using 0.25% trypsin and 0.02% EDTA (Merck Millipore, Darmstadt, Germany) and centrifuged at 2,000 × g for 5 min. Suspended cells were gathered, seeded on 6-well plates at 1.5–2×106 cells/well and incubated for two days.
Authenticating BMSCs
BMSCs were fixed using 5% precooled paraformaldehyde for 30 min at 4°C and incubated with hematoxylin (Merck Millipore) for 10 min. BMSCs were washed with water for 10 min, and 95% ethyl alcohol and xylene were used to dehydrate and clear BMSCs, respectively. BMSCs were observed using light microscopy (D5300; Nikon Corp., Tokyo, Japan).
Assessment of primary BMSCs growth
BMSCs (1–2×106 cells or 1–2×104 per well) were cultured in 6- or 96-well culture plates overnight at 37°C in an atmosphere containing 5% CO2. Glycitin (Merck Millipore) was added to the wells at final concentrations of 0.01, 0.5, 1, 5 and 10 µM and cultured for 7 days.
In cells cultured in 6-well culture plates, BMSCs were determined using Oil Red O staining and observed via light microscopy at 510 nm. BMSCs were fixed using 5% precooled paraformaldehyde for 30 min at 4°C and stained with 0.6% (w/v) Oil Red O solution for 15 min at room temperature. Cells stained with Oil Red O were washed with water (3×5 min) to remove unbound dye, and culture dishes were stained with 1 ml isopropyl alcohol for 10 min.
In cells cultured in 96-well culture plates, BMSCs were determined via MTT assay. A total of 20 µl MTT (5 g/l) were added to each well and cultured for 4 h. The supernatant was removed and 200 µl dimethylsulfoxide were added to each well for 15 min. Optical density (OD) was measured using a microplate spectrophotometer (model 680; Bio-Rad Laboratories, Inc., Hercules, CA, USA) at 570 nm. Proliferation rate was calculated using: OD treated / OD control × 100%.
Measurement of collagen type 1 (Col I) and alkaline phosphatase (ALP) using reverse-transcription polymerase chain reaction (RT-PCR)
Total RNA was extracted from BMSCs treated with glycitin (0, 0.5, 1 and 5 µM) using TRIzol reagent (Invitrogen; Thermo Fisher Scientific, Inc., Waltham, MA, USA). Total RNA (1–2 µg) was used to transcribe cDNA using a SYBR PrimeScript RT-PCR kit (Takara Bio, Inc., Otsu, Japan), according to the manufacturer's protocol PCR was performed on an ABI 7500 Real-Time PCR system (Applied Biosystems; Thermo Fisher Scientific, Inc.). PCR thermal cycling was performed as follows: Amplification at 94°C for 1 min, followed by 40 cycles of amplification at 94°C for 30 sec, annealing at 58°C for 45 sec, and extension at 72°C for 30 sec. Primers were designed as follows: Col I, forward 5′-TGACCTCAAGATGTGCCACT-3′ and reverse 5′-GGGAGTTTCCATGAAGCCAC-3′; and β-actin forward 5′-CGTGCGGGACATCAAGGA-3′ and reverse 5′-AGGAAGGAGGGCTGGAACA-3′. Subsequently, 7500 Fast Real-Time PCR system software was used to analyze crossing threshold (Cq) values using the second derivative maximum method (16).
Measurement of ALP activity
BMSCs (1–2×106 cells) were cultured in 6-well plates overnight at 37°C in an atmosphere containing 5% CO2. Glycitin was added to the wells at final concentrations of 0, 0.5, 1 and 5 µM and cultured for 7 days. Cells were washed with ice-cold PBS and lysed via the repeated freeze-thaw method. Supernatant was analyzed using an ALP kit (Sangon Biotech Co., Ltd., Shanghai, China). ALP activity was calculated according to the formula: Treated group / control × 100%.
Western blotting for TGF-β and phosphorylated AKT (p-AKT)
Proteins were extracted from BMSCs treated with glycitin (0, 0.5, 1 and 5 µM) by grinding with protease inhibitors. BMSCs were lysed using RIPA lysis buffer and protein content was measured using a micro-Bradford assay kit (Sangon Biotech Co., Ltd.). Protein samples (50–100 µg) were separated using 10–12% SDS-PAGE and transferred onto a polyvinylidene fluoride membrane. Membranes were incubated with 5% non-fat milk for 2 h at room temperature, and were subsequently incubated with antibodies against TGF-β (1:500; sc-146), p-AKT (1:500; sc-135,650) and β-actin (1:1,000; sc-130,656; all Santa Cruz Biotechnology, Inc., Dallas, TX, USA) at 4°C for 24 h. Membranes were washed three times with Tris-buffered saline with Tween-20 and subsequently incubated with an anti-rabbit secondary antibody (1:5,000; C2247; Applygen Technologies Inc., Beijing, China) at 37°C for 30 min at room temperature and were visualized with enhanced chemiluminescent reagent (ECL Plus; P0018A; Beyotime Institute of Biotechnology, Haimen, China). Proteins were quantified using Image Lab 4.1 (Bio-Rad Laboratories, Inc.).
Statistical analysis
Data are presented as the mean ± standard deviation using SPSS software (version 18.0; SPSS, Inc., Chicago, IL, USA). Statistical differences were analyzed using Student's t-test. P<0.05 was considered to indicate a statistically significant difference.
Results
Authentication of BMSCs
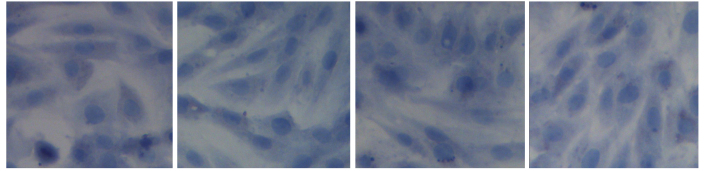
The constitutional formula of glycitin is displayed in Fig. 1. BMSCs appeared homogeneously mazarine blue, which demonstrated that BMSCs were successful extracted, indicating that BMSCs were successfully separated and cultivated (Fig. 2).
Figure 1.
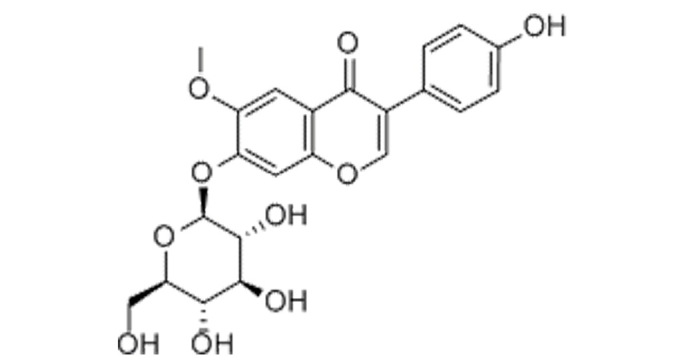
Constitutional formula of glycitin.
Figure 2.
Authentication of bone marrow stem cells, as detected by hematoxylin (magnification, ×400).
Glycitin promotes BMSC proliferation
MTT assay was used to measure the effect of glycitin on the proliferation of BMSCs. As shown in Fig. 3, glycitin increased the proliferation of BMSCs, with statistical significance detected after treatment with 1 and 5 µM glycitin (P=0.0023 and P=0.0004, respectively).
Figure 3.
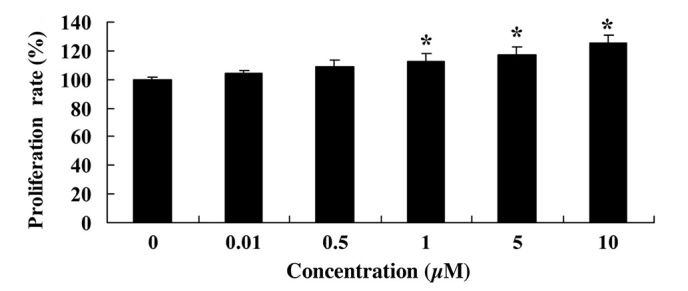
Glycitin promotes the proliferation of bone marrow stem cells. *P<0.01 vs. the 0 µM glycitin-treated group.
Glycitin increases adipogenic differentiation of BMSCs
As shown in Fig. 4, Oil Red O staining was observed in every group. A marked increase in red staining, and therefore osteoblasts, was detected following treatment with 1 and 5 µM glycitin.
Figure 4.
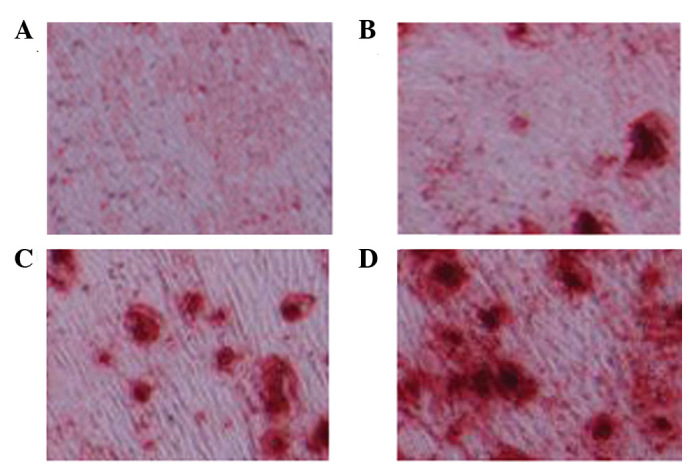
Adipogenic differentiation of bone marrow stem cells, as detected by Oil Red O staining (magnification, ×400). (A) 0, (B) 0.5, (C) 1 and (D) 5 µM glycitin-treated BMSCs.
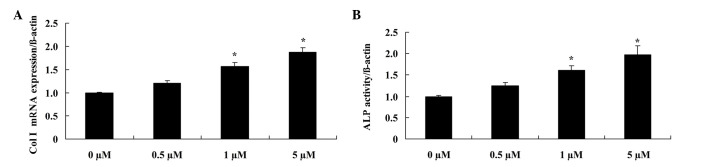
Glycitin increases Col I mRNA expression and ALP activity in BMSCs
In order to elucidate the effect of glycitin on the mRNA expression levels and activity of Col I and ALP, respectively, RT-PCR and ALP kits were used. The results demonstrated that administration of 1 and 5 µM glycitin significantly promoted Col I mRNA expression (P=0.0079 and P=0.0031, respectively) and ALP activity in BMSCs (P=0.0049 and P=0.0023, respectively; Fig. 5).
Figure 5.
Effect of glycitin on Col I and ALP mRNA expression levels in BMSCs. (A) Col I and (B) ALP mRNA expression levels were analyzed in BMSCs after treatment with glycitin. *P<0.01 vs. the 0 µM glycitin-treated group. Col I, collagen type I; ALP, alkaline phosphatase; BMSCs, bone marrow stem cells.
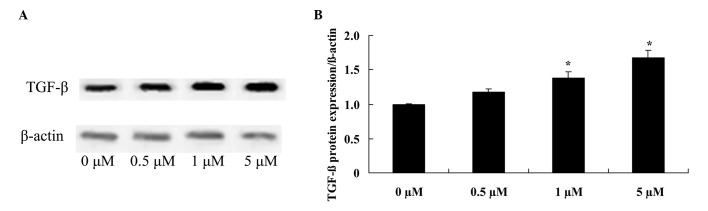
Glycitin increases TGF-β expression levels in BMSCs
To confirm the effect of glycitin on TGF-β signaling in BMSCs, TGF-β protein expression of BMSCs was detected. The results indicated that pretreatment with 1 and 5 µM glycitin significantly enhanced TGF-β protein expression of BMSCs (P=0.0063 and P=0.0021, respectively; Fig. 6).
Figure 6.
Effect of glycitin on TGF-β signaling of BMSCs. The effect of glycitin on TGF-β protein expression levels was assessed by (A) western blotting assays and (B) subsequent statistical analysis of TGF-β protein expression levels in BMSCs. *P<0.01 vs. the 0 µM glycitin-treated group. TGF-β, transforming growth factor beta-1; BMSCs, bone marrow stem cells.
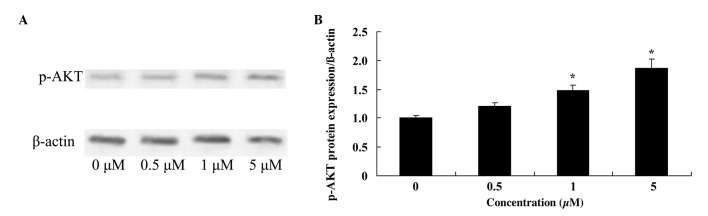
Glycitin increases p-AKT levels in BMSCs
To investigate the effect of glycitin on p-AKT in BMSCs, p-AKT was measured using western blotting. The results demonstrated that 1 and 5 µM glycitin significantly increased the presence of p-AKT in BMSCs (P=0.0071 and P=0.0033, respectively; Fig. 7).
Figure 7.
Effect of glycitin on p-AKT of BMSCs. The effect of glycitin on p-AKT protein expression levels was assessed by (A) western blotting assays and (B) subsequent statistical analysis of p-AKT protein expression levels in BMSCs. *P<0.01 vs. the 0 µM glycitin-treated group. P-AKT, phosphorylated AKT; BMSCs, bone marrow stem cells.
Discussion
Old bones and osteoblasts are absorbed by osteoclasts and new bones are formed (17). During continuous reconstruction, the equilibrium of the re-constructional process is regulated by a complicated signal network consisting of hormones, growth factors, cytokines, chemo-tactic factors and mechanical signals (18). BMSCs are capable of osteogenic differentiation, which indicates the prospect of the clinical application for biotherapy based on BMSCs (19). Osteogenic differentiation of BMSCs, in vitro and in vivo, has been extensively studied (20,21). The results of the present study demonstrated that glycitin promotes cell proliferation, osteoblast induction, and activates Col I mRNA expression and ALP activity of BMSCs. Li et al (14) reported that daidzin, genistin, and glycitin affects osteogenic and adipogenic differentiation.
The TGF-β protein super family includes TGF, activins, inhibin and bone morphogenetic proteins (22). These proteins have important roles in cell proliferation, differentiation, formation of cell matrix, the formation of tissues and organs, embryonic development and immunoregulation. During bone formation, the combination of TGF-β and its ligand may inhibit the formation of osteoclasts and bone absorption, but may additionally facilitate the osteogenesis of osteoblasts (23). Recent research has shown that TGF-β1 may promote the osteogenic differentiation of hMSCs and induce the expression of osteogenesis genes, ALP, collagen type I and osteocalcins (24). Conversely, the addition of TGF-β3 may inhibit the expression of ALP, suggesting that the TGF-β signaling pathway has a different role in the osteogenic differentiation of hMSCs (25). In addition, it was demonstrated that glycitin significantly enhanced T GF-β protein expression levels in BMSCs. Kim et al (26) indicated that glycitin promotes the proliferation and migration of human dermal fibroblast cells through TGF-β signaling.
Through the above data analysis, it was indicated that oxygen deficit and Ang II may induce the phosphorylation of Akt, a downstream modifier of PI3K, which can alter downstream modifiers and lead to changes in cell proliferation (27). Glycitin significantly advanced p-AKT formation in BMSCs. Kim et al (26) indicated that glycitin promotes proliferation and migration of human dermal fibroblast cells through TGF-β and p-AKT signaling.
In conclusion, the results of the present study demonstrated that glycitin promotes cell proliferation and induces osteoblast differentiation in BMSCs. A notable finding was that molecules endowed with activating Col I mRNA expression, ALP activity, and TGF-β and p-AKT signaling participated in the effect of glycitin regulating osteoblasts in BMSCs.
Acknowledgements
This research was supported by the National Natural Science Foundation of China (grant no. 81301,564) and the Army Medical Science Youth Training Project (grant no. 13QNP184), the ‘Twelfth Five-year’ science and technology research project of Jilin Department of Education (grant no. 2015-141) and the Jilin province Department of Project (grant no. 20130624003JC).
References
- 1.Mathews S, Bhonde R, Gupta PK, Totey S. Extracellular matrix protein mediated regulation of the osteoblast differentiation of bone marrow derived human mesenchymal stem cells. Differentiation. 2012;84:185–192. doi: 10.1016/j.diff.2012.05.001. [DOI] [PubMed] [Google Scholar]
- 2.Yamachika E, Tsujigiwa H, Matsubara M, Hirata Y, Kita K, Takabatake K, Mizukawa N, Kaneda Y, Nagatsuka H, Iida S. Basic fibroblast growth factor supports expansion of mouse compact bone-derived mesenchymal stem cells (MSCs) and regeneration of bone from MSC in vivo. J Mol Histol. 2012;43:223–233. doi: 10.1007/s10735-011-9385-8. [DOI] [PubMed] [Google Scholar]
- 3.Staudt ND, Aicher WK, Kalbacher H, Stevanovic S, Carmona AK, Bogyo M, Klein G. Cathepsin X is secreted by human osteoblasts, digests CXCL-12 and impairs adhesion of hematopoietic stem and progenitor cells to osteoblasts. Haematologica. 2010;95:1452–1460. doi: 10.3324/haematol.2009.018671. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Yang C, Liu Y, Li C, Zhang B. Repair of mandibular defects by bone marrow stromal cells expressing the basic fibroblast growth factor transgene combined with multi-pore mineralized Bio-Oss. Mol Med Rep. 2013;7:99–104. doi: 10.3892/mmr.2012.1171. [DOI] [PubMed] [Google Scholar]
- 5.Kim BS, Kang HJ, Park JY, Lee J. Fucoidan promotes osteoblast differentiation via JNK- and ERK-dependent BMP2-Smad 1/5/8 signaling in human mesenchymal stem cells. Exp Mol Med. 2015;47:e128. doi: 10.1038/emm.2014.95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Glynn ER, Londono AS, Zinn SA, Hoagland TA, Govoni KE. Culture conditions for equine bone marrow mesenchymal stem cells and expression of key transcription factors during their differentiation into osteoblasts. J Anim Sci Biotechnol. 2013;4:40. doi: 10.1186/2049-1891-4-40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Lee HW, Kim SY, Kim AY, Lee EJ, Choi JY, Kim JB. Adiponectin stimulates osteoblast differentiation through induction of COX2 in mesenchymal progenitor cells. Stem Cells. 2009;27:2254–2262. doi: 10.1002/stem.144. [DOI] [PubMed] [Google Scholar]
- 8.Clabaut A, Delplace S, Chauveau C, Hardouin P, Broux O. Human osteoblasts derived from mesenchymal stem cells express adipogenic markers upon coculture with bone marrow adipocytes. Differentiation. 2010;80:40–45. doi: 10.1016/j.diff.2010.04.004. [DOI] [PubMed] [Google Scholar]
- 9.Barbier V, Winkler IG, Wadley R, Lévesque JP. Flow cytometry measurement of bone marrow perfusion in the mouse and sorting of progenitors and stems cells according to position relative to blood flow in vivo. Methods Mol Biol. 2012;844:45–63. doi: 10.1007/978-1-61779-527-5_4. [DOI] [PubMed] [Google Scholar]
- 10.Zhang YB, Chen WH, Guo JJ, Fu ZH, Yi C, Zhang M, Na XL. Soy isoflavone supplementation could reduce body weight and improve glucose metabolism in non-Asian postmenopausal women-a meta-analysis. Nutrition. 2013;29:8–14. doi: 10.1016/j.nut.2012.03.019. [DOI] [PubMed] [Google Scholar]
- 11.Xu Z, Wu Q, Godber JS. Stabilities of daidzin, glycitin, genistin, and generation of derivatives during heating. J Agric Food Chem. 2002;50:7402–7406. doi: 10.1021/jf025626i. [DOI] [PubMed] [Google Scholar]
- 12.Robb EL, Stuart JA. Multiple phytoestrogens inhibit cell growth and confer cytoprotection by inducing manganese superoxide dismutase expression. Phytother Res. 2014;28:120–131. doi: 10.1002/ptr.4970. [DOI] [PubMed] [Google Scholar]
- 13.Hsu A, Bray TM, Helferich WG, Doerge DR, Ho E. Differential effects of whole soy extract and soy isoflavones on apoptosis in prostate cancer cells. Exp Biol Med (Maywood) 2010;235:90–97. doi: 10.1258/ebm.2009.009128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Li XH, Zhang JC, Sui SF, Yang MS. Effect of daidzin, genistin, and glycitin on osteogenic and adipogenic differentiation of bone marrow stromal cells and adipocytic transdifferentiation of osteoblasts. Acta Pharmacol Sin. 2005;26:1081–1086. doi: 10.1111/j.1745-7254.2005.00161.x. [DOI] [PubMed] [Google Scholar]
- 15.Zang Y, Igarashi K, Yu C. Anti-obese and anti-diabetic effects of a mixture of daidzin and glycitin on C57BL/6J mice fed with a high-fat diet. Biosci Biotechnol Biochem. 2015;79:117–123. doi: 10.1080/09168451.2014.955453. [DOI] [PubMed] [Google Scholar]
- 16.Li W, Ling W, Teng X, Quan C, Cai S, Hu S. Effect of advanced glycation end products, extracellular matrix metalloproteinase inducer and matrix metalloproteinases on type-I collagen metabolism. Biomed Rep. 2016;4:691–693. doi: 10.3892/br.2016.641. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Peng KY, Horng LY, Sung HC, Huang HC, Wu RT. Antiosteoporotic Activity of Dioscorea alata L. cv. Phyto through driving mesenchymal stem cells differentiation for bone formation. Evid Based Complement Alternat Med. 2011;2011:712892. doi: 10.1155/2011/712892. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Han DS, Chang HK, Kim KR, Woo SM. Consideration of bone regeneration effect of stem cells: Comparison of bone regeneration between bone marrow stem cells and adipose-derived stem cells. J Craniofac Surg. 2014;25:196–201. doi: 10.1097/SCS.0000000000000378. [DOI] [PubMed] [Google Scholar]
- 19.Mosig RA, Martignetti JA. Loss of MMP-2 in murine osteoblasts upregulates osteopontin and bone sialoprotein expression in a circuit regulating bone homeostasis. Dis Model Mech. 2013;6:397–403. doi: 10.1242/dmm.007914. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Gao M, Chen J, Lin G, Li S, Wang L, Qin A, Zhao Z, Ren L, Wang Y, Tang BZ. Long-Term tracking of the osteogenic differentiation of mouse BMSCs by aggregation-induced emission nanoparticles. ACS Appl Mater Interfaces. 2016;8:17878–17884. doi: 10.1021/acsami.6b05471. [DOI] [PubMed] [Google Scholar]
- 21.Wang C, Liu D, Zhang C, Sun J, Feng W, Liang XJ, Wang S, Zhang J. Defect-Related luminescent hydroxyapatite-enhanced osteogenic differentiation of bone mesenchymal stem cells via an ATP-induced cAMP/PKA pathway. ACS Appl Mater Interfaces. 2016;8:11262–11271. doi: 10.1021/acsami.6b01103. [DOI] [PubMed] [Google Scholar]
- 22.Batlle R, Alba-Castellón L, Loubat-Casanovas J, Armenteros E, Francí C, Stanisavljevic J, Banderas R, Martin-Caballero J, Bonilla F, Baulida J, et al. Snail1 controls TGF-β responsiveness and differentiation of mesenchymal stem cells. Oncogene. 2013;32:3381–3389. doi: 10.1038/onc.2012.342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Kumar A, Ruan M, Clifton K, Syed F, Khosla S, Oursler MJ. TGF-β mediates suppression of adipogenesis by estradiol through connective tissue growth factor induction. Endocrinology. 2012;153:254–263. doi: 10.1210/en.2011-1169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Claros S, Rico-Llanos GA, Becerra J, Andrades JA. A novel human TGF-β1 fusion protein in combination with rhBMP-2 increases chondro-osteogenic differentiation of bone marrow mesenchymal stem cells. Int J Mol Sci. 2014;15:11255–11274. doi: 10.3390/ijms150711255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Yin XX, Chen ZQ, Liu ZJ, Ma QJ, Dang GT. Icariine stimulates proliferation and differentiation of human osteoblasts by increasing production of bone morphogenetic protein 2. Chin Med J (Engl) 2007;120:204–210. [PubMed] [Google Scholar]
- 26.Kim YM, Huh JS, Lim Y, Cho M. Soy isoflavone glycitin (4′-Hydroxy-6-Methoxyisoflavone-7-D-Glucoside) promotes human dermal fibroblast cell proliferation and migration via TGF-β signaling. Phytother Res. 2015;29:757–769. doi: 10.1002/ptr.5313. [DOI] [PubMed] [Google Scholar]
- 27.Wang C, Lin K, Chang J, Sun J. Osteogenesis and angiogenesis induced by porous β-CaSiO (3)/PDLGA composite scaffold via activation of AMPK/ERK1/2 and PI3K/Akt pathways. Biomaterials. 2013;34:64–77. doi: 10.1016/j.biomaterials.2012.09.021. [DOI] [PubMed] [Google Scholar]