Fig. 1. Designed and observed cage symmetry.
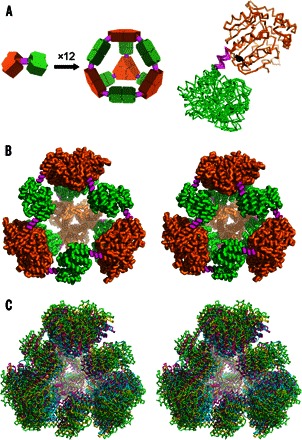
(A) Left: Schematic diagram of the symmetry principles used to design the 12-subunit tetrahedral cage by fusing two oligomeric domains (green and orange) by a semirigid linker (magenta) (14, 15). Right: Single point mutation distinguishing our PCtrip construct from PCquad replaces tyrosine (black sticks) with alanine in the trimeric domain that makes contact with the linker. (B) Side-by-side view of the theoretically designed (perfectly symmetric) model of the protein cage (left) and the most symmetric crystal structure obtained in this work for the PCquad variant (right). (C) Walleyed stereo view of three crystal structures of PCquad (yellow, magenta, and blue) overlaid on the ideal model (green ribbon), showing the agreement of the observed structures and the design.
