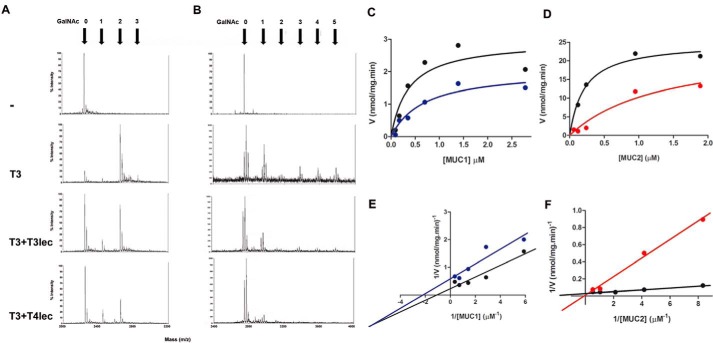
FIGURE 2.
Effects of lectin domains on enzyme activity of ppGalNAc-T3 (T3). MUC1 (A) and MUC5B (B) glycosylation products were analyzed by MALDI-TOF. Numbers of α-GalNAc incorporated were determined on acceptor peptide without ppGalNAc-T3 enzyme (control), with ppGalNAc-T3 (8 nm), and with ppGalNAc-T3 (8 nm) in the presence of 0.8 μm T3lec or T4lec domain. Kinetics plots of peptide glycosylation assay under initial velocity condition of ppGalNAc-T3 (1.6 nm) enzymatic reaction (black) are shown. Effects of 0.08 μm T3lec on ppGalNAc-T3 activity with MUC1 (C) or MUC2 (D) as acceptor peptide were assayed. Plots were fitted to the Michaelis-Menten equation using the GraphPad software program, yielding R2 values of 0.83 (C, black), 0.92 (C, blue), 0.96 (D, black), and 0.95 (D, red). Double reciprocal plots of enzyme activity without lectin domain (black) or in the presence of 0.08 μm T3lec (E, blue and F, red) indicated the type of inhibition on ppGalNAc-T3 activity. Plots were fitted to linear regression using the GraphPad software program, yielding R2 values 0.92 (E, black), 0.88 (E, blue) 0.98 (F, black), and 0.99 (F, red). Results are representative of three independent experiments.