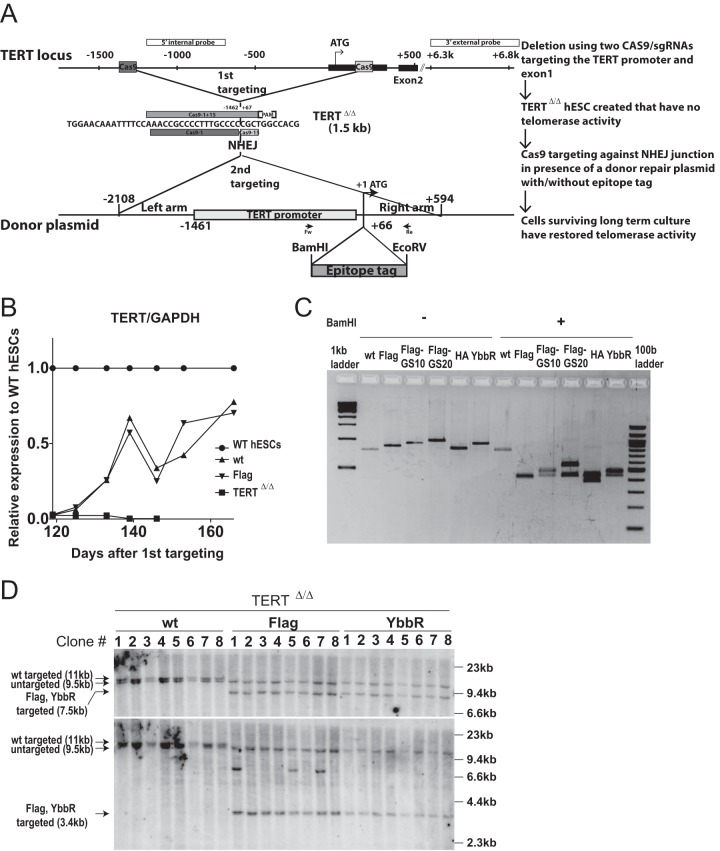
FIG 1.
Generation of endogenously N-terminally tagged hESCs using scarless two-step genome editing. (A) Schematic overview of the two-step genome-editing approach used to insert epitope tags into the N terminus of endogenous TERT. First, a TERT knockout cell line (TERTΔ/Δ) that lacks the region between 1.5 kb upstream and 66 bp downstream of the first ATG was established using two Cas9/sgRNAs (sg-1 and sg-15). Second, an sgRNA against the newly synthesized NHEJ-derived junction (−1462 and +67: sg1+15) was coelectroporated with donor plasmids containing the deleted regions with, or without, epitope tags after the first ATG of TERT. After the second targeting, the cells were passaged until all of the parental TERTΔ/Δ hESCs died due to telomere shortening. (B) Relative expression levels of TERT mRNA measured by quantitative RT-PCR over a time course after genome editing (day 0, first editing; day 86, second editing). Levels of expression of targeted wild-type (wt) and Flag-TERT hESCs are shown relative to those seen with nontargeted original wild-type (WT) hESCs. TERTΔ/Δ cells are plotted until the last time point before the TERTΔ/Δ cultures died. Expression of TERT was normalized to GAPDH. WT and TERTΔ/Δ cell data are identical to the data in reference 52. (C) PCR genotyping of the targeted cells after selection. Bulk DNA was used for PCR using the first ATG, and the products were digested with BamHI to confirm the insertion of epitope tags. wt, wild type. (D) Southern blot genotyping of targeted hESCs. After all TERTΔ/Δ hESCs died, single-cell-derived colonies were isolated from bulk populations. Genomic DNA was digested with BamHI and blotted using either 3′-external (top) or 5′-internal (bottom) probes. An untargeted allele appears as a 9.5-kb band. The sizes of the correctly targeted allele were 11 kb (wild type) or 7.5 kb (Flag, YbbR), respectively, for the external probe and 11 kb (wild type) or 3.4 kb (Flag, YbbR), respectively, for the internal probe.