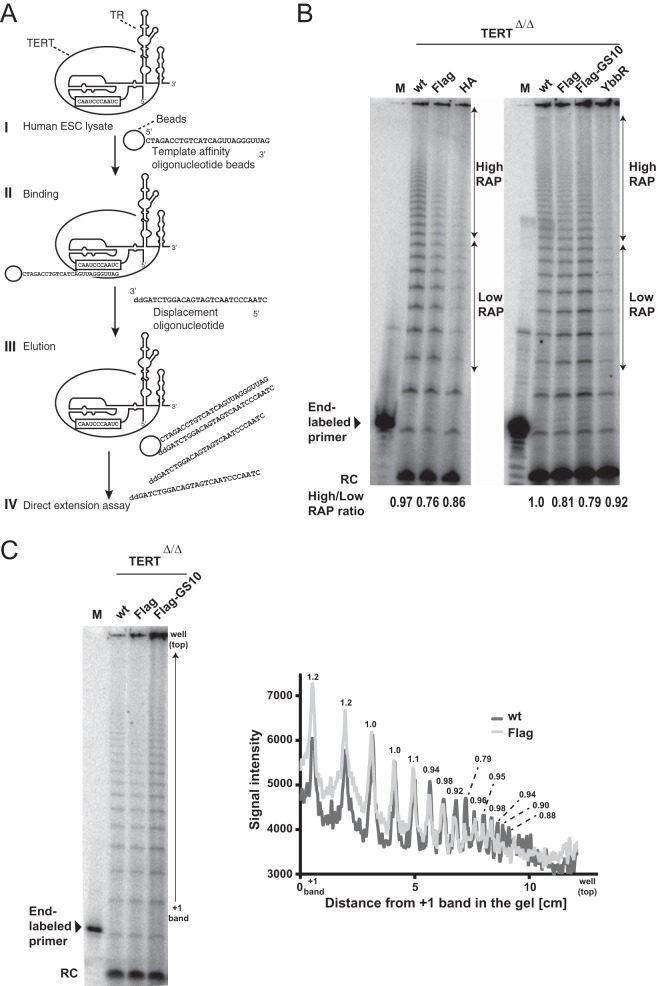
FIG 7.
N-terminally tagged TERT is defective in repeat addition processivity. (A) Schematic overview of purification of endogenous telomerase by template affinity oligonucleotide purification as previously reported in reference 57. (I) Lysate form wt or tagged hESCs were prepared using hypotonic buffer. (II) The lysate was incubated with the telomerase template affinity oligonucleotide-conjugated beads. (III) After unspecific binding on the beads was washed off, enriched telomerase was eluted using the displacement oligonucleotide, which possesses a sequence complementary to the template affinity oligonucleotide but cannot be elongated by telomerase due to its 3′-end 2′-3′-dideoxycytidine. (IV) The elution fraction was used for direct telomerase activity assay. (B) Direct telomerase activity assay for wt or endogenously tagged hESCs. An 18-nt (TTAGGG)3 primer was used as the substrate of the direct extension assay. This 18-nt substrate was used as an end-labeled marker. A 12-nt (TTAGGG)2 primer was used as the recovery control (RC) for ethanol precipitation. Telomerase activity represented in the region indicated by arrows was quantified, and the ratio of high RAP to low RAP is shown at the bottom of the gel. (C) Direct telomerase activity assay as described for panel A. Lysates of wt and tagged TERT hESCs from panel B were prepared independently followed by telomerase template oligonucleotide purification and a direct-extension assay to measure telomerase activity. To visualize the RAP defect, the graph to the right shows the signal intensity of the gel to the left for wild-type cells with the signal intensities for Flag-TERT cells superimposed. Numbers above each peak indicate Flag/wt signal ratios.