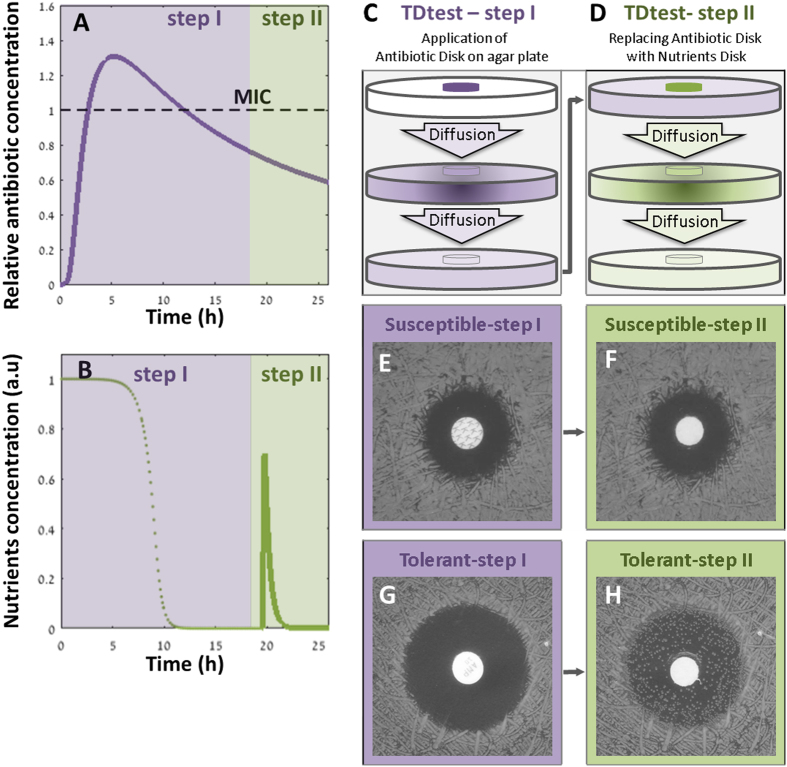
Figure 2. Rationale for the TDtest method for detecting differences in tolerance levels.
(A) Theoretical antibiotic concentration profile over time at a point 0.8 cm from the center of the antibiotic disk, assuming a point-source diffusion profile (Eq. 1). As the antibiotic diffuses in the agar, the concentration increases rapidly and then slowly decreases. After several hours (here: 12 h), the antibiotic concentration falls below the MIC at this point. The concentration is shown in units of the MIC, D = 8.5e-6 cm2 · sec−1 (See Fig. S11) and A = 10 μg, h = 0.4 cm. See Supplementary Fig. S9 for the profile of the antibiotic in the plate over time. (B) Schematic concentration profile over time at the same representative point as shown in (A). Nutrients in the whole plate are consumed by the growing bacteria outside the inhibition zone (dotted line). Due to diffusion, nutrients in the inhibition zone are also depleted after several hours. The lack of nutrients prevents the re-growth of surviving bacteria, even though the antibiotics concentration is below the MIC. Replacing the antibiotic disk with the glucose disk in the TDtest allows re-growth and detection of the surviving bacteria (solid line). (C) TDtest first step: the antibiotic disk is put on top the agar plate. The antibiotic diffuses from the disk. (D) TDtest second step: replacing the antibiotics disk with a glucose disk. (E,F) Susceptible strain: growth inhibition around the antibiotic disk (E), no late growth after glucose addition (F). (G,H) Tolerant strain: growth inhibition around the antibiotic disk (G), colonies inside the inhibition zone after glucose addition, indicate slow or late growing bacteria of this strain (H). Disk diameter: 6 mm.