Abstract
Background
Studies evaluating dietary patterns, including the DASH diet, and their relationship with the metabolic syndrome and diabetes may help to understand the role of dairy products (low fat or full fat) in these conditions. Our aim is to identify dietary patterns in Brazilian adults and compare them with the (DASH) diet quality score in terms of their associations with metabolic syndrome and newly diagnosed diabetes in the Brazilian Longitudinal Study of Adult Health-the ELSA-Brasil study.
Methods
The ELSA-Brasil is a multicenter cohort study comprising 15,105 civil servants, aged 35–74 years at baseline (2008–2010). Standardized interviews and exams were carried out, including an OGTT. We analyzed baseline data for 10,010 subjects. Dietary patterns were derived by principal component analysis. Multivariable logistic regression investigated associations of dietary patterns with metabolic syndrome and newly diagnosed diabetes and multivariable linear regression with components of metabolic syndrome.
Results
After controlling for potential confounders, we observed that greater adherence to the Common Brazilian meal pattern (white rice, beans, beer, processed and fresh meats), was associated with higher frequencies of newly diagnosed diabetes, metabolic syndrome and all of its components, except HDL-C. Participants with greater intake of a Common Brazilian fast foods/full fat dairy/milk based desserts pattern presented less newly diagnosed diabetes. An inverse association was also seen between the DASH Diet pattern and the metabolic syndrome, blood pressure and waist circumference. Diet, light foods and beverages/low fat dairy pattern was associated with more prevalence of both outcomes, and higher fasting glucose, HDL-C, waist circumference (among men) and lower blood pressure. Vegetables/fruit dietary pattern did not protect against metabolic syndrome and newly diagnosed diabetes but was associated with lower waist circumference.
Conclusions
The inverse associations found for the dietary pattern characterizing Brazilian fast foods and desserts, typically containing dairy products, with newly diagnosed diabetes, and for the DASH diet with metabolic syndrome, support previously demonstrated beneficial effects of dairy products in metabolism. The positive association with metabolic syndrome and newly diagnosed diabetes found for the pattern characterizing a typical Brazilian meal deserves further investigation, particularly since it is frequently accompanied by processed meat.
Trial registration NCT02320461. Registered 18 December 2014
Electronic supplementary material
The online version of this article (doi:10.1186/s13098-017-0211-7) contains supplementary material, which is available to authorized users.
Keywords: Dietary patterns, Diabetes, Metabolic syndrome, Cohort study
Background
Type 2 diabetes and metabolic syndrome are global public health problems [1, 2], and nutritional interventions have been recommended for their prevention and control [3–5]. Studies investigating dietary patterns provide valuable information to assess healthful or harmful dietary habits, beyond those analyzing individual nutrients or food groups alone [6–8], since foods are consumed in complex combinations and nutrients may have interactive effects [9, 10].
A recent meta-analysis of prospective observational studies including 404,528 individuals, revealed that adherence to ‘healthy’ dietary patterns significantly reduced the risk of diabetes (RR = 0.86; 95% CI 0.82, 0.90), while ‘unhealthy’ dietary patterns (generally emphasizing red meat and refined carbohydrate based foods) increased risk (RR = 1.30; 95% CI 1.18, 1.43) [11]. Additionally, a Western dietary pattern, with strong components of refined carbohydrates, and red and processed meats, is associated with metabolic syndrome and cardiovascular diseases (CVD) [12–14].
The dietary approaches to stop hypertension (DASH) diet, a well-known dietary pattern specifically targeted to lowering blood pressure, was associated with lower CVD risk in US [15, 16] and European populations [17]. The DASH diet also has the potential to prevent type 2 diabetes and stroke [18, 19]. A meta-analysis of intervention studies based on the DASH diet showed reduced fasting insulin concentration and improved insulin sensitivity independently of weight loss [20]. The DASH diet emphasizes high intake of fruits, vegetables, nuts, legumes, whole grains, low-fat dairy products as well as a low intake of sodium, red and processed meats, and sweetened beverages.
The specific benefits of an increased intake of dairy foods have recently received greater attention. A pooled analysis of 7 cohort studies (254,892 participants and 19,082 cases of diabetes) revealed that higher intake of total dairy products was associated with lower risk of type 2 diabetes. Similar inverse associations were found for low-fat dairy products, low-fat or skim milk and cheese, and for yogurt, but not for high-fat dairy products or total milk; results regarding the metabolic syndrome were inconclusive [21]. Additionally, biomarkers of dairy intake are related to a decreased incidence of diabetes [22]. Analyzing data from the Brazilian Longitudinal Study of Adult Health (ELSA-Brasil) study, we found that the intake of total dairy products was inversely associated with measures of glycemia, insulinemia and metabolic syndrome with a linear dose–response pattern. Interestingly, these associations were observed for full-fat dairy products and yogurt but not for low-fat dairy products [23, 24].
Further investigation of dietary patterns and their relationship with diabetes and the metabolic syndrome in distinct populations may provide valuable information with regard to the role of specific combinations of foods and nutrients. Yet, studies evaluating dietary patterns, including the DASH diet, in relation to metabolic syndrome and diabetes are scarce, especially in populations outside of Europe and North America [25]. The present study aims to identify dietary patterns in Brazilian adults and compare their associations, as well as those of the (DASH) diet quality score, with metabolic syndrome and newly diagnosed diabetes.
Methods
Study design
The Brazilian Longitudinal Study of Adult Health is a multicenter cohort study and comprises 15,105 civil servant volunteers, aged 35–74 years at baseline (2008–2010), from universities or research institutions located in six Brazilian capitals (Belo Horizonte, Porto Alegre, Rio de Janeiro, Salvador, São Paulo, Vitoria). Description of study design and sample characteristics have been previously described [26]. The study was approved by the local research and ethics committees of participating institutions, and all participants provided written consent.
Study participants
For the current investigation we used data from the baseline examination. We excluded participants with previously diagnosed diabetes (diabetes status reported during initial interviews, or taking oral hypoglycemic medications or insulin [n = 1473]), self-reported chronic disease as CVD [n = 1280], cancer [n = 695], and other chronic diseases [n = 2649] (stroke, emphysema, bronchitis, chronic obstructive pulmonary disease, cirrhosis, hepatitis, cardiac or bariatric surgery, rheumatic fever, Chagas disease, and thrombosis or emboli), and with unusually low (2nd percentile; ≤1298 kcal/day) or high (98th percentile, ≥6372 kcal/day) reported energy intake [n = 629], which left 10,010 subjects for analysis. For analyses that involves metabolic syndrome, we also excluded participants with a fasting state either <12 or >15 h [n = 542]. Some participants had more than 1 exclusion criteria, resulting in 9835 participants.
Assessment of diet and covariates
Dietary data were collected using a validated food frequency questionnaire (FFQ) [27], with 114 food and drink items and covering the last 12 months. Participants were also asked to provide information on their typical eating habits, including attempts to modify their diet in the 6 months before the baseline examination.
We transformed the frequency options into daily frequencies as follows: 3 for more than 3 times/day; 2.5 for 2–3 times/day; 1 for once/day; 0.8 for 5–6 times/week; 0.4 for 2–4 times/week; 0.1 for once/week; 0.07 for 1–3 times/month; and 0 for never/almost never. Additionally, we obtained daily energy intake in kilocalories and nutrient intake using the University of Minnesota Nutrition Data System for Research database [28]. For these determinations, we calculated the grams/day for each food item from the FFQ as the quantity of servings consumed/day × weight (standard portion in grams) × frequency of consumption.
At baseline, trained interviewers collected demographic characteristics (age, sex, race, educational level, family income, occupational status, study site), family history of diabetes, menopausal status and lifestyle factors: smoking (current and previous), alcohol intake and physical activity. Alcohol intake was estimated as the sum of ethanol (g/week) of all beverages consumed. Physical activity variables were defined by using the leisure activity section of the International Physical Activity Questionnaire (IPAQ), long form, according to the IPAQ guidelines for data processing and analysis. Median (interquartile range) metabolic equivalent min/week were computed for walking, moderate-intensity and vigorous-intensity activities, and summed to obtain a combined leisure-time total physical activity score [29].
We performed anthropometric measurements (weight, height, and waist circumference) with participants standing, dressed in light standard uniforms, without shoes in the fasting state. We measured body weight to the nearest 0.1 kg with a calibrated balance (Toledo 2096PP) and height with a vertical stadiometer (Seca-SE-216) to the nearest 0.1 cm. Waist circumference was measured with a tape measure to the nearest 0.1 cm around the midpoint between the inferior costal border and the iliac crest. BMI was calculated as weight (kg) divided by height squared (m2).
Definition of metabolic syndrome
Blood was drawn by venipuncture after an overnight fast of 8–15 h, and a standard 2-h 75-g oral glucose tolerance test was administered for all participants who did not report a diagnosis or current treatment for diabetes. Glucose was measured by an ADVIA 1200 chemistry hexokinase system (Siemens); glycated hemoglobin using an HPLC assay (Bio-Rad D-10 Dual Program Laboratories) certified by the National Glycohemoglobin Standardization Program; and HDL-cholesterol and triglycerides using enzymatic procedures (ADVIA 1200).
Resting blood pressure was measured 3 times at 1-min intervals using a 765CP oscillometric sphygmomanometer (Omron) with participants seated after a 5-min rest. The average of the second and third measurements was used in the analyses.
We used the joint interim statement consensus criteria [30] for diagnosing metabolic syndrome, which requires the presence of any 3 of the following 5 risk factors: elevated waist circumference (≥102 cm in men and ≥88 cm in women), hypertriglyceridemia (≥1.69 mmol/L/150 mg/dL or drug treatment), reduced HDL-cholesterol (<1.03 mmol/L/<40 mg/dL for men and <1.29 mmol/L/<50 mg/dL for women, or drug treatment), elevated blood pressure (≥130 mmHg; diastolic blood pressure ≥85 mmHg and/or drug treatment), and elevated fasting glucose (≥100 mg/dL/5.6 mmol/L or drug treatment).
Definition of newly diagnosed diabetes
After excluding participants with previously diagnosed diabetes, we considered as newly diagnosed diabetes participants with fasting glucose ≥126 mg/dL, 2-h postload glucose ≥200 mg/dL or HbA1c ≥6.5%.
Statistical analysis
Dietary patterns were derived using principal component analysis (PCA). All 114 foods and beverages, including alcohol, were standardized to per day units. The Kaiser–Meyer–Olkin test (>0.6) and Bartlett’s test of sphericity (p < 0.05) were applied to verify whether the PCA assumptions were all met. The factors were rotated orthogonally by the varimax method, and those with eigenvalues ≥1.5 were retained. To determine the number of factors to retain, we also considered the scree plot and factor interpretability. Factor was interpreted based on food items with loadings ≥0.20. We identified four dietary patterns and named them according to food items showing high loadings (Table 1). Factors extracted did not vary by sex and study site. Factor scores were determined for each participant by multiplying standardized intake of each food item (grams/day) by its respective loading factor on each factor. Factor scores were thus linear variables, representing the weighted sum across all 114 food and beverage items. Individuals were classified into quintiles of each factor score. Results are presented for three groups: high adherence (fifth quintile); mid adherence (third quintile); low adherence (first quintile) to a given pattern quintile.
Table 1.
Dietary patterns extracted using principal component analysis (n = 9835)
| Dietary Patterns (P1–P4) | Loading weights ≥0.4 | Loading weights 0.4–0.3 | Cumulative % of the variance explained |
|---|---|---|---|
| Vegetables/fruits (P1) | Zucchini/chayote/eggplant, collard greens, carrots, green beans, cabbage, lettuce, chicory/watercress/arugula, tomato | Orange/tangerine, pumpkin, apple/pear, beets, cauliflower, broccoli, papaya, banana, okra, pineapple, onion, melon, grape | 4.3 |
| Common Brazilian fast foods/full fat dairy/desserts (P2) | Pizza, chocolate, fries, flan/mousses | Ham/salami, fried fast foods, baked fast foods, layered cake, beef stroganoff, ice cream, hot dog, mozzarella and other yellow cheeses, hamburger patty, regular soda | 8.4 |
| Common Brazilian meal (P3) | White rice, beans, garlic | Beer, sausage, tripe/stomach, bacon, onion, fried chicken, pork | 11.0 |
| Diet, light foods and beverages/low fat dairy (P4) | Low fat turkey breast | Low fat yogurt, coffee with artificial sweetener, diet, light or zero soda, low calorie bread, low fat cream cheese | 13.1 |
Extraction method: principal component analysis using varimax rotation
A DASH Diet quality score was also created to assess adherence to the DASH diet [16]. This score considered intake of fruits, vegetables, nuts and legumes, whole grains, low-fat dairy products as well as that of sodium, red and processed meats, and sweetened beverages. For each of these components, subjects were classified into sex-specific quintiles according to their intake, with values being assigned to each quintile (from 1 for the lowest intake to 5 for the highest, except for the components sodium, and red and processed meats, and sweetened beverages for which the scoring was reversed). We summed up the eight DASH component scores to obtain an overall DASH score ranging from 8 (lowest adherence) to 40 (highest adherence) [17].
Multivariable logistic regression was used to evaluate the adjusted association of the different dietary patterns with metabolic syndrome and newly diagnosed diabetes. A linear trend was tested by modeling categorical quintiles of dietary patterns consumption as a continuous variable in the multivariable regression models. We evaluated interactions between dietary patterns and excess weight (BMI ≥25 kg/m2). We adjusted for covariates (potential confounders) in 2 models: model 1 included demographic covariates, menopausal status, and family history of diabetes; model 2 included all variables from model 1 plus body mass index (BMI), physical activity, current and previous smoking, alcohol intake, and calories/day. Our intention was to separate the adjustment for obesity and related behavioral characteristics. Since obesity may be interpreted as a confounder or as a mediator of the associations, we provide results for both models, facilitating interpretation of potential confounding. Multivariable linear regression was performed to investigate adjusted associations of different dietary patterns with components of metabolic syndrome: fasting glucose, triglycerides, HDL cholesterol, waist circumference, systolic and diastolic blood pressure. We adjusted for covariates using similar models.
All analyses were performed in the statistical package SPSS version 18 (IBM).
Results
The mean ± SD age was 50.7 ± 8.7 year, and 54.8% (n = 5390) were women. Four main dietary patterns were derived from PCA (Table 1). The first pattern, “vegetables/fruits” (P1), had high loadings for vegetables and fruits (not including fruit juice). The second pattern, “Common Brazilian fast foods/full fat dairy/desserts” (P2), had high loadings for fast foods (fried or baked), cakes, milk based desserts, regular cheese and red meats (fresh and processed), which characterizes, in general a common pattern of eating in fast food snack bars/restaurants in Brazil. The third pattern, “Common Brazilian meal” (P3), had high loadings for white rice, beans, beer, processed and fresh meats, which includes the classic Brazilian meal (black beans, rice and meat). The fourth pattern “Diet or light foods and beverages/low fat dairy” (P4) had high loadings for low fat foods, low or zero sugar beverages with artificial sweeteners and low fat dairy, which characterizes a pattern of eating of those interested in improving their health by eating less calories/less sugar or by medical counseling. Mean servings/day for specific food groups according to the quintiles of dietary patterns scores are presented in Additional file 1: Table S1. Participant characteristics according to low, mid and high adherence to dietary patterns are presented in Additional file 1: Table S2. Those with higher vegetables/fruits (P1) scores were older, more often women, less frequently a university graduate, smoked less, drank less alcohol, were more active, and had lower intake of total fat and saturated fat, and greater consumption of carbohydrate. Those with higher Common Brazilian fast foods/full-fat dairy/desserts (P2) scores were predominantly men, younger, white, with a university degree, and had greater intake of total and saturated fat, and less protein consumption. Those with a higher Common Brazilian meal (P3) score were predominantly men, and on average slightly younger, of mixed color-race (“Pardo”), less frequently a university graduate, smoked more, drank more alcohol, were less active and had slightly higher BMI. Those with higher scores in the Diet, light foods and beverages/low fat dairy (P4) pattern were more frequently women, white, with a university degree, smoked less, drank more alcohol, were more active, with high protein consumption, less carbohydrate intake and higher BMI. Finally, those with higher adherence to DASH Diet were more frequently women, elderly, white, with a university degree, smoked less, drank less alcohol, were more active and had a lower BMI. They had less total fat and saturated fat consumption and more carbohydrate intake.
The frequency of metabolic syndrome was 30.7% (n = 3019) and of newly diagnosed diabetes, 10.9% (n = 1044). Table 2 presents unadjusted frequencies of metabolic syndrome and newly diagnosed diabetes according to levels of adherence (low, mid and high) to dietary patterns. Frequency of metabolic syndrome increased with levels of adherence to the Common Brazilian meal and the Diet, light foods and beverages/low fat dairy dietary patterns, but decreased with higher adherence to the DASH Diet. Frequency of newly diagnosed diabetes increased with levels of adherence to the Common Brazilian meal (frequency was twice that of low adherence). In contrast, lower frequency of newly diagnosed diabetes was seen in those of high adherence to the Common Brazilian fast foods/full fat dairy/desserts pattern.
Table 2.
Metabolic syndrome and newly diagnosed diabetes by adherence to dietary patterns (n = 9835)
| Outcomes | Dietary patterns (P1–P4) | Adherence to different dietary patterns (in quintiles) | ||
|---|---|---|---|---|
| Quintile 1—low adherence | Quintile 3—mid adherence | Quintile 5—high adherence | ||
| N (%) | ||||
| Metabolic syndrome, n (%) | P1—Vegetables/fruits | 594 (30.6) | 558 (28.7) | 677 (34.9) |
| P2—Common Brazilian fast food/Full fat dairy/desserts | 704 (36.2) | 545 (28.0) | 714 (36.7) | |
| P3—Common Brazilian meal | 421 (21.6) | 591 (30.4) | 786 (40.4) | |
| P4—Diet, light foods and beverages/low fat dairy | 540 (27.8) | 616 (31.7) | 629 (32.4) | |
| DASH diet | 571 (32.7) | 712 (31.2) | 538 (29.4) | |
| Newly diagnosed diabetes, n (%) | P1—Vegetables/Fruits | 184 (9.5) | 189 (9.7) | 261 (13.4) |
| P2—Common Brazilian fast food/Full fat dairy/desserts | 314 (16.1) | 183 (9.4) | 182 (9.4) | |
| P3—Common Brazilian | 128 (6.6) | 185 (9.5) | 314 (16.1) | |
| P4—Diet, light foods and beverages/low fat dairy | 209 (10.7) | 197 (10.1) | 199 (10.2) | |
| DASH diet | 176 (10.1) | 247 (10.8) | 200 (10.9) | |
Table 3 shows adjusted odds ratios for metabolic syndrome and newly diagnosed diabetes according to the adherence to each pattern when categorized in quintiles. Strong and graded positive association remained for the Common Brazilian meal pattern for both metabolic syndrome and newly diagnosed diabetes after adjustment (model 2), with odds of presenting either outcome being approximately twice as frequent in the highest compared to the lowest quintile. Similarly, for the Diet, light foods and beverages/low fat dairy pattern, we also found positive associations for both outcomes, those in the 5th quintile, compared to the 1st quintile having approximately 50% greater odds of presenting either outcome.
Table 3.
Adjusted associations of metabolic syndrome and newly diagnosed diabetes with adherence to dietary patterns (n = 9835)
| Metabolic syndrome criteriaa (OR and 95% CI) | P for trendb | |||||
|---|---|---|---|---|---|---|
| Q1 | Q2 | Q3 | Q4 | Q5 | ||
| P1—Vegetables/fruits | ||||||
| Model 1 | 1.00 | 0.87 (0.75, 1.00) | 0.87 (0.75, 1.00) | 0.96 (0.83, 1.10) | 1.04 (0.90, 1.20) | 0.229 |
| Model 2 | 1.00 | 0.91 (0.77, 1.06) | 0.88 (0.75, 1.03) | 0.96 (0.82, 1.13) | 1.07 (0.90, 1.28) | 0.366 |
| P2—Common Brazilian fast foods/full fat dairy/milk based desserts | ||||||
| Model 1 | 1.00 | 0.85 (0.74, 0.98) | 0.86 (0.74, 0.99) | 0.89 (0.77, 1.03) | 1.08 (0.94, 1.25) | 0.225 |
| Model 2 | 1.00 | 0.83 (0.71, 0.97) | 0.81 (0.69, 0.95) | 0.77 (0.65, 0.91) | 0.86 (0.71, 1.04) | 0.057 |
| P3—Common Brazilian meal | ||||||
| Model 1 | 1.00 | 1.51 (1.29, 1.75) | 1.81 (1.55, 2.11) | 2.26 (1.93, 2.65) | 2.89 (2.45, 3.42) | <0.001 |
| Model 2 | 1.00 | 1.32 (1.12, 1.56) | 1.48 (1.25, 1.76) | 1.81 (1.52, 2.15) | 2.13 (1.76, 2.58) | <0.001 |
| P4—Diet, light foods and beverages/low fat dairy | ||||||
| Model 1 | 1.00 | 1.31 (1.13, 1.51) | 1.59 (1.37, 1.85) | 1.78 (1.53, 2.08) | 2.02 (1.72, 2.38) | <0.001 |
| Model 2 | 1.00 | 1.24 (1.05, 1.46) | 1.40 (1.18, 1.65) | 1.44 (1.21, 1.71) | 1.47 (1.23, 1.71) | <0.001 |
| DASH diet pattern | ||||||
| Model 1 | 1.00 | 0.90 (0.78, 1.03) | 0.88 (0.77, 1.01) | 0.75 (0.64, 0.86) | 0.72 (0.61, 0.83) | <0.001 |
| Model 2 | 1.00 | 0.94 (0.80, 1.10) | 0.96 (0.82, 1.11) | 0.81 (0.69, 0.95) | 0.88 (0.74, 1.05) | 0.044 |
| Newly diagnosed diabetesc | ||||||
| P1—Vegetables/fruits | ||||||
| Model 1 | 1.00 | 1.01 (0.81, 1.25) | 0.95 (0.76, 1.19) | 0.99 (0.80, 1.24) | 1.19 (0.96, 1.47) | 0.132 |
| Model 2 | 1.00 | 1.05 (0.84, 1.32) | 1.00 (0.79, 1.26) | 1.04 (0.82, 1.31) | 1.29 (1.01, 1.65) | 0.077 |
| P2—Common Brazilian fast foods/Full fat dairy/milk based desserts | ||||||
| Model 1 | 1.00 | 0.70 (0.57, 0.85) | 0.71 (0.57, 0.87) | 0.66 (0.53, 0.82) | 0.76 (0.61, 0.94) | 0.007 |
| Model 2 | 1.00 | 0.67 (0.54, 0.82) | 0.66 (0.53, 0.81) | 0.57 (0.45, 0.72) | 0.59 (0.46, 0.77) | <0.001 |
| P3—Common Brazilian meal | ||||||
| Model 1 | 1.00 | 1.54 (1.21, 1.97) | 1.58 (1.23, 2.02) | 1.92 (1.50, 2.46) | 2.53 (1.96, 3.25) | <0.001 |
| Model 2 | 1.00 | 1.41 (1.09, 1.80) | 1.42 (1.10, 1.82) | 1.64 (1.28, 2.12) | 2.04 (1.56, 2.68) | <0.001 |
| P4—Diet, light foods and beverages/low fat dairy | ||||||
| Model 1 | 1.00 | 1.23 (0.99, 1.52) | 1.29 (1.04, 1.61) | 1.61 (1.28, 2.01) | 1.71 (1.35, 2.18) | <0.001 |
| Model 2 | 1.00 | 1.19 (0.96, 1.48) | 1.21 (0.96, 1.52) | 1.46 (1.15, 1.84) | 1.45 (1.13, 1.88) | 0.001 |
| DASH Diet Pattern | ||||||
| Model 1 | 1.00 | 1.01 (0.82, 1.26) | 1.01 (0.82, 1.25) | 0.94 (0.75, 1.18) | 0.91 (0.72, 1.15) | 0.324 |
| Model 2 | 1.00 | 1.04 (0.83, 1.29) | 1.07 (0.86, 1.32) | 1.01 (0.80, 1.27) | 1.07 (0.84, 1.36) | 0.702 |
Model 1 Adjusted through multiple logistic regression for the following variables: age (y), sex, race, education, family income (amount), occupational status, study center, menopausal status and family history of diabetes
Model 2 Model 1 + BMI (kg/m2), physical activity (metabolic equivalent min/week), current and previous smoking status, alcohol (grams of ethanol/day) and calorie intake (kcal/day)
aDefined by the Joint Interim Statement Consensus Criteria, 2009 (see “Methods”)
bPCA components entered as continuous scores
cDefined by fasting glucose, oral glucose tolerance test or glycated hemoglobin
In contrast, as seen also in Table 3, participants who adhered to the Common Brazilian fast foods/full fat dairy/desserts dietary pattern had a considerably lower prevalence of newly diagnosed diabetes (OR for quintile 5 vs. quintile 1 = 0.59; 0.50–0.86) and a borderline statistically significant lower prevalence of metabolic syndrome. A borderline inverse association was seen also between higher adherence to the DASH Diet pattern and metabolic syndrome. When we adjusted additionally for full fat dairy, greater adherence to the Common Brazilian fast foods/full-fat dairy/desserts pattern was no longer associated with the metabolic syndrome, and the association with new onset diabetes moved slightly towards the null. Those with a greater vegetables/fruits pattern showed no association with the metabolic syndrome and inconsistently increased odds for having diabetes.
Figures 1 and 2 present means of all components of metabolic syndrome according to adherence to dietary patterns and the DASH Diet, adjusted in similar models. We observed graded, positive associations between adherence to the Common Brazilian meal pattern and adjusted mean values for all syndrome components except HDL-cholesterol. Positive associations were also found for the diet, light foods and beverages/low fat dairy pattern with levels of fasting glucose, HDL-cholesterol and, in men only, for waist circumference. However, an inverse association for this dietary pattern was seen with systolic and diastolic blood pressure, similarly with the findings seen for the DASH diet. The vegetables/fruits pattern and the DASH diet were inversely related with waist circumference.
Fig. 1.
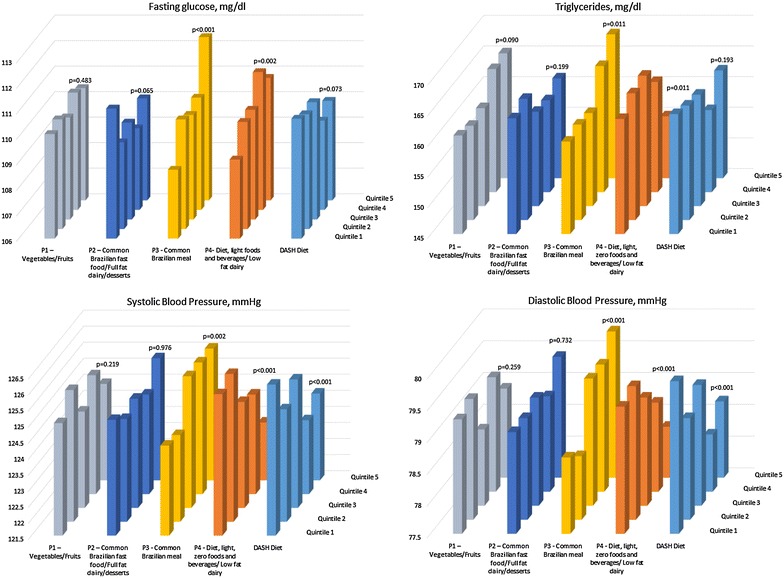
Adjusted* means for fasting glucose (mg/dL), triglycerides (mg/dL), systolic and diastolic blood pressure according to quintiles of adherence to the four dietary patterns identified and to the DASH-diet. ELSA-Brasil 2008–2010 (n = 9835). *Means obtained through multiple linear regression adjusting for: race, age, sex, education, study center, menopause, occupational status, family history of diabetes, BMI, physical activity, smoking, alcohol, calories/day
Fig. 2.
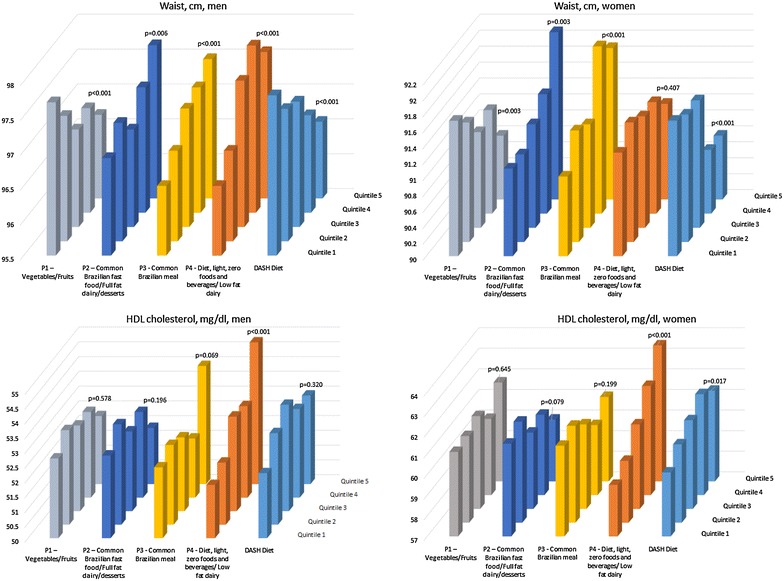
Adjusted* means for waist circumference (cm) in men and women and HDL cholesterol (mg/dL) in men and women according to quintiles of adherence to the four dietary patterns identified and to the DASH-diet. ELSA-Brasil 2008–2010 (n = 9835). *Means obtained through multiple linear regression adjusting for: race, age, sex, education, study center, menopause, occupational status, family history of diabetes, BMI, physical activity, smoking, alcohol, calories/day
Suggestions of multiplicative interaction were seen between BMI categories and Common Brazilian meal dietary pattern (P3) for metabolic syndrome (p = 0.024), Common Brazilian fast foods/full-fat dairy/desserts dietary pattern (P2) for incident diabetes (p = 0.10) and diet/light foods and beverages/low fat dairy pattern for newly diagnosed diabetes (p = 0.157). In stratified analyses the associations between the Common Brazilian meal dietary pattern (P3) and metabolic syndrome were stronger in normal BMI participants than in the overweight or obese. The Common Brazilian fast foods/full-fat dairy/desserts was not associated with newly diagnosed diabetes in normal BMI participants, but was so associated in overweight and obese participants (p = 0.004).
Discussion
In this large cohort of free-living middle-aged and older Brazilian men and women, after controlling for potential confounders, we observed that greater adherence to the Common Brazilian meal pattern (P3: white rice, beans, beer, processed and fresh meats), was associated with higher frequencies of newly diagnosed diabetes, metabolic syndrome and all of its components, except HDL-C. In contrast, those with greater intake of a Common Brazilian fast foods/full fat dairy/milk based desserts pattern (P2) presented less diabetes (although higher waist circumference). An inverse association was also seen between the DASH diet pattern and the metabolic syndrome, blood pressure and waist circumference. Those who adhered more to the diet, light foods and beverages/low fat dairy pattern (P4) had greater prevalences of both outcomes, and higher fasting glucose, HDL-C, waist circumference (among men) and lower blood pressure. Finally, the vegetables/fruit dietary pattern (P1) did not protect against metabolic syndrome and newly diagnosed diabetes but was associated with lower waist circumference.
Our findings concerning the Common Brazilian meal pattern merit discussion. Greater consumption of beans, a typical Brazilian food with important nutrients (fiber, potassium), when combined with white rice, processed and fresh meats, and beer, was not related to a generally better metabolic status. Similar findings were observed in the multi-ethnic study of atherosclerosis (MESA), where “Beans, tomatoes, and refined grains” pattern was associated with greater risk of type 2 diabetes [31]. Three additional US cohorts (Nurses’ Health Study, Nurses’ Health Study 2, and Health Professionals’ Follow-up Study) found that similar dietary patterns, including regular consumption of refined grains, red and processed meats, and sugar-sweetened beverages including fruits juices were associated with increased risk of diabetes [32]. Further, in a northern German cohort, a pattern characterized by high intake of potatoes, various vegetables, beef, pork, processed meat, fats, sauce and bouillon, reflecting a ‘traditional German diet’, was positively associated with metabolic syndrome [33].
Possible mechanistic factors linking empirically derived dietary patterns with measures of metabolic dysfunction have been previously described [34, 35], including associations with some markers of inflammation and endothelial activation. In the MESA Study, a “fats, oils, processed meats, fried potatoes, salty snacks, and desserts” pattern, considered a Westernized dietary pattern, was positively associated with C-reactive protein, interleukin 6, and homocysteine. The “beans, tomatoes, and refined grains” pattern was positively related to soluble intercellular adhesion molecule-1 [34]. A systematic review found positive association between Westernized dietary patterns (higher intakes of processed meats, sweets, fried foods, and refined grains) and inflammation molecules and atherogenic promoters [35, 36].
That greater adherence to the Common Brazilian fast foods/full fat dairy/milk based desserts pattern-P2 was associated with a lower frequency of newly diagnosed diabetes and a DASH diet pattern, as defined a priori, with a lower prevalence of metabolic syndrome might be explained, at least in part, by the dairy products present in these patterns. Dairy products are dense in nutrients, including dietary saturated fatty acids present in full-fat dairy products. It has been postulated that the impact of these saturated fatty acids on metabolic risk may be influenced by the food and nutrient matrix involved with dairy intake, including micronutrients such as calcium, potassium, and magnesium, which may contribute to lower the risk of newly diagnosed diabetes and cardiovascular diseases [37]. In our recent finding from the ELSA-Brasil study, the only nutrient that materially changed the associations of greater dairy intake with lesser frequency of metabolic syndrome was saturated fatty acids from dairy products [22]. Similarly, a population-based prospective cohort study in Sweden found that high intake of full-fat dairy products was associated with decreased incidence of diabetes, compared to low-fat dairy products, whereas the consumption of both low- and high-fat meats was associated with increased incidence [38]. Addition of full fat dairy as a covariate in our adjusted model in fact decreased somewhat the inverse association found, suggesting that full fat dairy may mediate, in part, this association.
It is not clear how dietary fat content and food sources of fat may modulate the effect of saturated fatty acids on risk of diabetes and metabolic syndrome. The major food sources of saturated fatty acid are of animal origin, including meat and dairy products. Ericson et al. [38] observed a decreased incidence of diabetes at high intake of high-fat dairy products but not of low-fat dairy products, and meat intake was associated with increased risk independently of fat content. Analyzing saturated fatty acids with shorter chain lengths (4:0–10:0), mainly found in dairy products, the study indicated protective associations with diabetes. Dairy products are also better sources of lauric acid (12:0) and myristic acid (14:0). In contrast, palmitic acid (16:0) and stearic acid (18:0), which are abundant in both dairy foods and meat, fish, and eggs, showed null associations with diabetes [38]. In our previous ELSA analysis, we found that myristic acid could explain the association between dairy product consumption and glycemia, insulin, and glycated hemoglobin concentrations [23]. A Dutch study with 12 years of follow-up found a lower ischemic heart disease risk with a higher intake of saturated fatty acids, and that this association was apparently mainly driven by short- to medium-chain saturated fatty, myristic acid, the sum of pentadecylic and margaric acids, and saturated fatty acids from dairy sources including butter, cheese, and milk and milk products [39]. A recent meta-analysis relating diabetes with consumption of butter, the dairy product highest in fat content, found that in four cohort studies (201,628 participants and 23,954 incident diabetes cases) butter consumption was associated with lower incidence of type 2 diabetes, with 4% lower risk per 14 g daily intake (RR = 0.96; 95% CI 0.93,0.99) [40].
In the present study we found that blood pressure was lower with increased adherence to diet, light foods and beverages/low fat dairy (P4) and DASH diet patterns, which both include low fat dairy in their composition. We previously observed in ELSA a significant, inverse, graded dose–response association between total dairy intake and adjusted systolic and diastolic blood pressure [24]. In addition to the saturated fatty acids present in dairy, other nutritional components may be involved in the reduction of the metabolic syndrome. Milk proteins have an angiotensin-converting enzyme-inhibitory effect and the inhibition of the renin angiotensin system in adipocytes can potentially reduce hypertension [41]. Calcium reduces blood pressure by modulation of smooth muscle reactivity and vitamin D may reduce dyslipidemia and improve blood pressure through maintenance of calcium homeostasis, stimulation of insulin production and release, and regulation of the renin–angiotensin–aldosterone system [40]. In contrast, a recent Mendelian randomization study using a lactase persistence genotype as an instrumental variable found that while this genotype was associated with greater dairy consumption, it showed no association with blood pressure [42].
Our findings of increased frequencies of newly diagnosed diabetes and the metabolic syndrome among those with greater adherence to the dietary pattern diet, light foods and beverages/low fat dairy (P4) might be explained by reverse causality, since a presumed knowledge of increased risk of diabetes could have led some participants to this pattern of “dieting”, or due to residual confounding, as previously suggested [43, 44]. Two additional interpretations are worth considering. First, habitual consumption of artificially sweeteners may increase the risk of diabetes and the metabolic syndrome [45–47], perhaps by alterations in the intestinal microbiota [48] or by imbalance of sweetness and caloric content (simulating a fasting state), which may trigger sensory and behavioral responses to increase caloric consumption [49]. Second, very low dietary fat intake (<15%) has been related with a higher incidence of metabolic syndrome [50].
Our findings that greater adherence to the vegetables/fruits (P1) dietary pattern did not associate with lesser frequencies of newly diagnosed diabetes and the metabolic syndrome, and was only associated with slightly lower waist circumferences are puzzling. Interestingly, in the PREDIMED trial, a significant inverse association for fiber and fruits with risk of CVD was seen only in minimally adjusted models, but not in fully adjusted models [51].
It is reassuring that the dietary patterns here derived were similar to those generated using a different methodology [52]. In the previous analyses consumption of fast-foods (and other unhealthy foods) were mixed with beans, fruits and vegetables, and most likely decreased the potential nutritional benefits of the latter foods. In this regard, it is important to highlight the emphasis on natural rather than processed foods given in the new Brazilian Dietary Guideline for the Brazilian population.
A major strength of this study is the direct measurements of the outcomes, including measurements of the cardiometabolic risk factors and detection of diabetes by a standardized 75-g oral-glucose-tolerance test and glycated hemoglobin. However, this study has some limitations. We measured dietary intake with a FFQ, a typical choice in large epidemiologic studies. This method is subject to random and systematic errors, although this limitation would most likely have resulted in a nondifferential misclassification with respect to outcomes and a likely underestimation of associations. Another limitation is that the factor analyses used to identify the dietary patterns depend on several decisions made by the researcher, such as number of factors to be retained. Furthermore, we used cross-sectional data to identify the association of dietary patterns with the metabolic syndrome and diabetes, and because of the observational nature of our study, it is possible that our findings result from reverse causality or residual confounding, the latter despite the fact that we extensively adjusted for other dietary variables and risk factors. It is also possible that some of the participants for whom we ascertained newly diagnosed diabetes reported a higher adherence to the vegetable/fruits and to the diet/light food patterns in response to their perceived propensity to develop diabetes (e.g. family history, knowledge of intermediate hyperglycemia). Although the exclusions we made aimed to prevent some of these potential biases, they may also introduce selectin bias. However, previously reported comparisons suggest that this is unlikely [23].
In conclusion, the inverse associations found for the dietary pattern characterizing Brazilian fast foods and desserts, typically containing dairy products, with newly diagnosed diabetes, and for the DASH diet with metabolic syndrome, support previously demonstrated beneficial effects of dairy products in metabolism. The positive association with metabolic syndrome and newly diagnosed diabetes found for the pattern characterizing a typical Brazilian meal deserves further investigation, particularly since it is frequently accompanied by processed meat.
Authors’ contributions
MD, MIS, BBD, SA, LOC and MDM designed and conducted research and provided essential materials; MD, AO and MAP analyzed data and wrote paper; MD, AO, MAP, MIS, BBD, SA, LOC and MDM had primary responsibility for final content. All authors read and approved the final manuscript.
Acknowledgements
The authors thank the staff and participants of the ELSA-Brasil for their important contributions.
Competing interests
The authors declare that they have no competing interests.
Availability of data and materials
The datasets generated and analyzed during the current study are not publicly available . According to ELSA-Brasil protocol, any analysis proposal must be evaluated and approved by the Study´s Publication´s Committee before beginning analyses.
Ethics approval and consent to participate
The study was approved by Hospital de Clínicas de Porto Alegre Ethic Committee, with reference number 06-194, and all participants provided written consent.
Funding
The ELSA-Brasil Study was supported by the Brazilian Ministry of Health (Science and Technology Department) and the Brazilian Ministry of Science, Technology and Innovation (Financiadora de Estudos e Projetos-FINEP and Conselho Nacional de Desenvolvimento Científico e Tecnológico-CNPq), grants 01 06 0010.00 RS, 01 06 0212.00 BA, 01 06 0300.00 ES, 01 06 0278.00 MG, 01 06 0115.00SP, 01 06 0071.00 RJ and postdoctoral grant 249320/2013.
Additional file
Additional file 1: Table S1. Means of servings/day (sv/d) for food groups according to quintiles of adherence to each dietary pattern. ELSA-Brasil 2008–2010 (n=10,010). Table S2. Characteristics of Brazilian participants without previously diagnosed diabetes by categories of high adherence to dietary patterns: ELSA-Brasil 2008–2010 (n=10,010).
Contributor Information
Michele Drehmer, Phone: + 55 51 3308 5347, Email: michele.drehmer@gmail.com.
Andrew O. Odegaard, Email: aodegaar@uci.edu
Maria Inês Schmidt, Email: maria.schmidt@ufrgs.br.
Bruce B. Duncan, Email: bbduncan@ufrgs.br
Letícia de Oliveira Cardoso, Email: leticiaocar@ensp.fiocruz.br.
Sheila M. Alvim Matos, Email: sheilaalvim@gmail.com.
Maria del Carmen B. Molina, Email: mdmolina@uol.com.br
Sandhi M. Barreto, Email: sandhi.barreto@gmail.com
Mark A. Pereira, Email: perei004@umn.edu
References
- 1.da Rocha Fernandes J, Ogurtsova K, Linnenkamp U, Guariguata L, Seuring T, Zhang P, Cavan D, Makaroff LE. IDF diabetes Atlas estimates of 2014 global health expenditures on diabetes. Diabetes Res Clin Pract. 2016;117:48–54. doi: 10.1016/j.diabres.2016.04.016. [DOI] [PubMed] [Google Scholar]
- 2.O’Neill S, O’Driscoll L. Metabolic syndrome: a closer look at the growing epidemic and its associated pathologies. Obes Rev. 2015;16(1):1–12. doi: 10.1111/obr.12229. [DOI] [PubMed] [Google Scholar]
- 3.Waxman A, World Health Assembly WHO global strategy on diet, physical activity and health. Food Nutr Bull. 2004;25(3):292–302. doi: 10.1177/156482650402500310. [DOI] [PubMed] [Google Scholar]
- 4.Knowler WC, Barrett-Connor E, Fowler SE, Hamman RF, Lachin JM, Walker EA, Nathan DM, Diabetes Prevention Program Research G Reduction in the incidence of type 2 diabetes with lifestyle intervention or metformin. N Engl J Med. 2002;346(6):393–403. doi: 10.1056/NEJMoa012512. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Neuhouser ML, Howard B, Lu J, Tinker LF, Van Horn L, Caan B, Rohan T, Stefanick ML, Thomson CA. A low-fat dietary pattern and risk of metabolic syndrome in postmenopausal women: the women’s health Initiative. Metabolism. 2012;61(11):1572–1581. doi: 10.1016/j.metabol.2012.04.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Batis C, Mendez MA, Sotres-Alvarez D, Gordon-Larsen P, Popkin B. Dietary pattern trajectories during 15 years of follow-up and HbA1c, insulin resistance and diabetes prevalence among Chinese adults. J Epidemiol Commun Health. 2014;68(8):773–779. doi: 10.1136/jech-2013-203560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Odegaard AOP. Dietary patterns and type 2 diabetes. In: Press C, editor. Nutrition and type 2 diabetes etiology and prevention. New York; 2014: p. 173–92.
- 8.Newby PK, Muller D, Tucker KL. Associations of empirically derived eating patterns with plasma lipid biomarkers: a comparison of factor and cluster analysis methods. Am J Clin Nutr. 2004;80(3):759–767. doi: 10.1093/ajcn/80.3.759. [DOI] [PubMed] [Google Scholar]
- 9.Jacobs DR, Jr, Steffen LM. Nutrients, foods, and dietary patterns as exposures in research: a framework for food synergy. Am J Clin Nutr. 2003;78(3 Suppl):508S–513S. doi: 10.1093/ajcn/78.3.508S. [DOI] [PubMed] [Google Scholar]
- 10.McNaughton SA, Mishra GD, Brunner EJ. Dietary patterns, insulin resistance, and incidence of type 2 diabetes in the Whitehall II Study. Diabetes Care. 2008;31(7):1343–1348. doi: 10.2337/dc07-1946. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Maghsoudi Z, Ghiasvand R, Salehi-Abargouei A. Empirically derived dietary patterns and incident type 2 diabetes mellitus: a systematic review and meta-analysis on prospective observational studies. Public Health Nutr. 2015:1–12. [DOI] [PMC free article] [PubMed]
- 12.Nettleton JA, Polak JF, Tracy R, Burke GL, Jacobs DR., Jr Dietary patterns and incident cardiovascular disease in the multi-ethnic study of atherosclerosis. Am J Clin Nutr. 2009;90(3):647–654. doi: 10.3945/ajcn.2009.27597. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Gadgil MD, Anderson CA, Kandula NR, Kanaya AM. Dietary patterns are associated with metabolic risk factors in South Asians living in the United States. J Nutr. 2015;145(6):1211–1217. doi: 10.3945/jn.114.207753. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Liu L, Nettleton JA, Bertoni AG, Bluemke DA, Lima JA, Szklo M. Dietary pattern, the metabolic syndrome, and left ventricular mass and systolic function: the multi-ethnic study of atherosclerosis. Am J Clin Nutr. 2009;90(2):362–368. doi: 10.3945/ajcn.2009.27538. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Appel LJ, Moore TJ, Obarzanek E, Vollmer WM, Svetkey LP, Sacks FM, Bray GA, Vogt TM, Cutler JA, Windhauser MM, DASH Collaborative Research Group et al. A clinical trial of the effects of dietary patterns on blood pressure. N Engl J Med. 1997;336(16):1117–1124. doi: 10.1056/NEJM199704173361601. [DOI] [PubMed] [Google Scholar]
- 16.Fung TT, Chiuve SE, McCullough ML, Rexrode KM, Logroscino G, Hu FB. Adherence to a DASH-style diet and risk of coronary heart disease and stroke in women. Arch Intern Med. 2008;168(7):713–720. doi: 10.1001/archinte.168.7.713. [DOI] [PubMed] [Google Scholar]
- 17.Struijk EA, May AM, Wezenbeek NL, Fransen HP, Soedamah-Muthu SS, Geelen A, Boer JM, van der Schouw YT, Bueno-de-Mesquita HB, Beulens JW. Adherence to dietary guidelines and cardiovascular disease risk in the EPIC-NL cohort. Int J Cardiol. 2014;176(2):354–359. doi: 10.1016/j.ijcard.2014.07.017. [DOI] [PubMed] [Google Scholar]
- 18.Liese AD, Nichols M, Sun X, D’Agostino RB, Jr, Haffner SM. Adherence to the DASH Diet is inversely associated with incidence of type 2 diabetes: the insulin resistance atherosclerosis study. Diabetes Care. 2009;32(8):1434–1436. doi: 10.2337/dc09-0228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Lin PH, Yeh WT, Svetkey LP, Chuang SY, Chang YC, Wang C, Pan WH. Dietary intakes consistent with the DASH dietary pattern reduce blood pressure increase with age and risk for stroke in a Chinese population. Asia Pac J Clin Nutr. 2013;22(3):482–491. [PubMed] [Google Scholar]
- 20.Shirani F, Salehi-Abargouei A, Azadbakht L. Effects of dietary approaches to stop hypertension (DASH) diet on some risk for developing type 2 diabetes: a systematic review and meta-analysis on controlled clinical trials. Nutrition. 2013;29(7–8):939–947. doi: 10.1016/j.nut.2012.12.021. [DOI] [PubMed] [Google Scholar]
- 21.Aune D, Norat T, Romundstad P, Vatten LJ. Dairy products and the risk of type 2 diabetes: a systematic review and dose-response meta-analysis of cohort studies. Am J Clin Nutr. 2013;98(4):1066–1083. doi: 10.3945/ajcn.113.059030. [DOI] [PubMed] [Google Scholar]
- 22.Yakoob MY, Shi P, Willett WC, Rexrode KM, Campos H, Orav EJ, Hu FB, Mozaffarian D. Circulating biomarkers of dairy fat and risk of incident diabetes mellitus among men and women in the United States in two large prospective cohorts. Circulation. 2016;133(17):1645–1654. doi: 10.1161/CIRCULATIONAHA.115.018410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Drehmer M, Pereira MA, Schmidt MI, Del Carmen BMM, Alvim S, Lotufo PA, Duncan BB. Associations of dairy intake with glycemia and insulinemia, independent of obesity, in Brazilian adults: the Brazilian Longitudinal Study of Adult Health (ELSA-Brasil) Am J Clin Nutr. 2015;101(4):775–782. doi: 10.3945/ajcn.114.102152. [DOI] [PubMed] [Google Scholar]
- 24.Drehmer M, Pereira MA, Schmidt MI, Alvim S, Lotufo PA, Luft VC, Duncan BB. Total and full-fat, but not low-fat, dairy product intakes are inversely associated with metabolic syndrome in adults. J Nutr. 2016;146(1):81–89. doi: 10.3945/jn.115.220699. [DOI] [PubMed] [Google Scholar]
- 25.Stricker MD, Onland-Moret NC, Boer JM, van der Schouw YT, Verschuren WM, May AM, Peeters PH, Beulens JW. Dietary patterns derived from principal component- and k-means cluster analysis: long-term association with coronary heart disease and stroke. Nutr Metab Cardiovasc Dis. 2013;23(3):250–256. doi: 10.1016/j.numecd.2012.02.006. [DOI] [PubMed] [Google Scholar]
- 26.Schmidt MI, Duncan BB, Mill JG, Lotufo PA, Chor D, Barreto SM, Aquino EM, Passos VM, Matos SM, Molina Mdel C, et al. Cohort profile: Longitudinal Study of Adult Health (ELSA-Brasil) Int J Epidemiol. 2015;44(1):68–75. doi: 10.1093/ije/dyu027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Molina Mdel C, Bensenor IM, Cardoso Lde O, Velasquez-Melendez G, Drehmer M, Pereira TS, Faria CP, Melere C, Manato L, Gomes AL, et al. Reproducibility and relative validity of the Food frequency questionnaire used in the ELSA-Brasil. Cad Saude Publica. 2013;29(2):379–389. doi: 10.1590/S0102-311X2013000600024. [DOI] [PubMed] [Google Scholar]
- 28.Center NC. Nutrition data system for research software. Minneapolis: University of Minnesota; 2010. [Google Scholar]
- 29.Craig CL, Marshall AL, Sjostrom M, Bauman AE, Booth ML, Ainsworth BE, Pratt M, Ekelund U, Yngve A, Sallis JF, et al. International physical activity questionnaire: 12-country reliability and validity. Med Sci Sports Exerc. 2003;35(8):1381–1395. doi: 10.1249/01.MSS.0000078924.61453.FB. [DOI] [PubMed] [Google Scholar]
- 30.Alberti KG, Eckel RH, Grundy SM, Zimmet PZ, Cleeman JI, Donato KA, Fruchart JC, James WP, Loria CM, Smith SC, Jr, et al. Harmonizing the metabolic syndrome: a joint interim statement of the International Diabetes Federation Task Force on Epidemiology and Prevention; National Heart, Lung, and Blood Institute; American Heart Association; World Heart Federation; International Atherosclerosis Society; and International Association for the Study of Obesity. Circulation. 2009;120(16):1640–1645. doi: 10.1161/CIRCULATIONAHA.109.192644. [DOI] [PubMed] [Google Scholar]
- 31.Nettleton JA, Steffen LM, Ni H, Liu K, Jacobs DR., Jr Dietary patterns and risk of incident type 2 diabetes in the Multi-ethnic study of atherosclerosis (MESA) Diabetes Care. 2008;31(9):1777–1782. doi: 10.2337/dc08-0760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Ardisson Korat AV, Willett WC, Hu FB. Diet, lifestyle, and genetic risk factors for type 2 diabetes: a review from the Nurses’ Health Study, Nurses’ Health Study 2, and Health Professionals’ Follow-up Study. Curr Nutr Rep. 2014;3(4):345–354. doi: 10.1007/s13668-014-0103-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Barbaresko J, Siegert S, Koch M, Aits I, Lieb W, Nikolaus S, Laudes M, Jacobs G, Nothlings U. Comparison of two exploratory dietary patterns in association with the metabolic syndrome in a Northern German population. Br J Nutr. 2014;112(8):1364–1372. doi: 10.1017/S0007114514002098. [DOI] [PubMed] [Google Scholar]
- 34.Nettleton JA, Steffen LM, Mayer-Davis EJ, Jenny NS, Jiang R, Herrington DM, Jacobs DR., Jr Dietary patterns are associated with biochemical markers of inflammation and endothelial activation in the Multi-Ethnic Study of Atherosclerosis (MESA) Am J Clin Nutr. 2006;83(6):1369–1379. doi: 10.1093/ajcn/83.6.1369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Defago MD, Elorriaga N, Irazola VE, Rubinstein AL. Influence of food patterns on endothelial biomarkers: a systematic review. J Clin Hypertens (Greenwich) 2014;16(12):907–913. doi: 10.1111/jch.12431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Goldin A, Beckman JA, Schmidt AM, Creager MA. Advanced glycation end products: sparking the development of diabetic vascular injury. Circulation. 2006;114(6):597–605. doi: 10.1161/CIRCULATIONAHA.106.621854. [DOI] [PubMed] [Google Scholar]
- 37.Sacks FM, Svetkey LP, Vollmer WM, Appel LJ, Bray GA, Harsha D, Obarzanek E, Conlin PR, Miller ER, 3rd, Simons-Morton DG, et al. Effects on blood pressure of reduced dietary sodium and the dietary approaches to stop hypertension (DASH) diet. N Engl J Med. 2001;344(1):3–10. doi: 10.1056/NEJM200101043440101. [DOI] [PubMed] [Google Scholar]
- 38.Ericson U, Hellstrand S, Brunkwall L, Schulz CA, Sonestedt E, Wallstrom P, Gullberg B, Wirfalt E, Orho-Melander M. Food sources of fat may clarify the inconsistent role of dietary fat intake for incidence of type 2 diabetes. Am J Clin Nutr. 2015;101(5):1065–1080. doi: 10.3945/ajcn.114.103010. [DOI] [PubMed] [Google Scholar]
- 39.Praagman J, Beulens JW, Alssema M, Zock PL, Wanders AJ, Sluijs I, van der Schouw YT. The association between dietary saturated fatty acids and ischemic heart disease depends on the type and source of fatty acid in the European Prospective Investigation into Cancer and Nutrition-Netherlands cohort. Am J Clin Nutr. 2016;103(2):356–365. doi: 10.3945/ajcn.115.122671. [DOI] [PubMed] [Google Scholar]
- 40.Pimpin L, Wu JH, Haskelberg H, Del Gobbo L, Mozaffarian D. Is butter back? A systematic review and meta-analysis of butter consumption and risk of cardiovascular disease, diabetes, and total mortality. PLoS ONE. 2016;11(6):e0158118. doi: 10.1371/journal.pone.0158118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Pihlanto-Leppala A, Koskinen P, Piilola K, Tupasela T, Korhonen H. Angiotensin I-converting enzyme inhibitory properties of whey protein digests: concentration and characterization of active peptides. J Dairy Res. 2000;67(1):53–64. doi: 10.1017/S0022029999003982. [DOI] [PubMed] [Google Scholar]
- 42.Hartwig FP, Horta BL, Smith GD, deMola CL, Victora CG. Association of lactase persistence genotype with milk consumption, obesity and blood pressure: a Mendelian randomization study in the 1982 Pelotas (Brazil) Birth Cohort, with a systematic review and meta-analysis. Int J Epidemiol. 2016;45:1573–1587. doi: 10.1093/ije/dyw074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Pereira MA. Sugar-sweetened and artificially-sweetened beverages in relation to obesity risk. Adv Nutr. 2014;5(6):797–808. doi: 10.3945/an.114.007062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Pereira MA, Odegaard AO. Artificially sweetened beverages—do they influence cardiometabolic risk? Curr Atheroscler Rep. 2013;15(12):375. doi: 10.1007/s11883-013-0375-z. [DOI] [PubMed] [Google Scholar]
- 45.Ferreira-Pego C, Babio N, Bes-Rastrollo M, Corella D, Estruch R, Ros E, Fito M, Serra-Majem L, Aros F, Fiol M, et al. Frequent consumption of sugar- and artificially sweetened beverages and natural and bottled fruit juices is associated with an increased risk of metabolic syndrome in a Mediterranean population at high cardiovascular disease risk. J Nutr. 2016;146:1528–1536. doi: 10.3945/jn.116.230367. [DOI] [PubMed] [Google Scholar]
- 46.Imamura F, O’Connor L, Ye Z, Mursu J, Hayashino Y, Bhupathiraju SN, Forouhi NG. Consumption of sugar sweetened beverages, artificially sweetened beverages, and fruit juice and incidence of type 2 diabetes: systematic review, meta-analysis, and estimation of population attributable fraction. BMJ. 2015;351:h3576. doi: 10.1136/bmj.h3576. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Yarmolinsky J, Duncan BB, Chambless LE, Bensenor IM, Barreto SM, Goulart AC, Santos IS, Diniz Mde F, Schmidt MI. Artificially sweetened beverage consumption is positively associated with newly diagnosed diabetes in normal-weight but not in overweight or obese Brazilian adults. J Nutr. 2016;146(2):290–297. doi: 10.3945/jn.115.220194. [DOI] [PubMed] [Google Scholar]
- 48.Suez J, Korem T, Zeevi D, Zilberman-Schapira G, Thaiss CA, Maza O, Israeli D, Zmora N, Gilad S, Weinberger A, et al. Artificial sweeteners induce glucose intolerance by altering the gut microbiota. Nature. 2014;514(7521):181–186. doi: 10.1038/nature13793. [DOI] [PubMed] [Google Scholar]
- 49.Wang QP, Lin YQ, Zhang L, Wilson YA, Oyston LJ, Cotterell J, Qi Y, Khuong TM, Bakhshi N, Planchenault Y, et al. Sucralose promotes food intake through NPY and a neuronal fasting response. Cell Metab. 2016;24(1):75–90. doi: 10.1016/j.cmet.2016.06.010. [DOI] [PubMed] [Google Scholar]
- 50.Park S, Ahn J, Lee BK. Very-low-fat diets may be associated with increased risk of metabolic syndrome in the adult population. Clin Nutr. 2015. [DOI] [PubMed]
- 51.Buil-Cosiales P, Toledo E, Salas-Salvado J, Zazpe I, Farras M, Basterra-Gortari FJ, Diez-Espino J, Estruch R, Corella D, Ros E, et al. Association between dietary fibre intake and fruit, vegetable or whole-grain consumption and the risk of CVD: results from the PREvencion con DIeta MEDiterranea (PREDIMED) trial. Br J Nutr. 2016;116(3):534–546. doi: 10.1017/S0007114516002099. [DOI] [PubMed] [Google Scholar]
- 52.Cardoso Lde O, Carvalho MS, Cruz OG, Melere C, Luft VC, Molina Mdel C, Faria CP, Bensenor IM, Matos SM, Fonseca Mde J, et al. Eating patterns in the Brazilian Longitudinal Study of Adult Health (ELSA-Brasil): an exploratory analysis. Cad Saude Publica. 2016;32(5):e00066215. doi: 10.1590/0102-311X00066215. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets generated and analyzed during the current study are not publicly available . According to ELSA-Brasil protocol, any analysis proposal must be evaluated and approved by the Study´s Publication´s Committee before beginning analyses.


