Abstract
Purpose
To examine the congruence between polysomnography obstructive apnea hypopnea index (OAHI) and parent reported obstructive sleep apnea (OSA) symptoms in 6-to-11 year-old children with juvenile idiopathic arthritis (JIA) and controls; and to compare fatigue and quality of life in JIA and control children based on OAHI and OSA symptoms.
Methods
Sixty-eight children with JIA and 75 controls and a parent participated. Children underwent one night of polysomnography in a sleep laboratory. Parents completed the sleep-related breathing disorders scale - Pediatric Sleep Questionnaire (PSQ), and both children and parents completed the Pediatric Quality of Life Generic Core Scale and the Multidimensional Fatigue scale.
Results
In JIA, 86% who met the OAHI clinical criteria for OSA (≥ 1.5) were above the PSQ OSA symptom cut-off score with a sensitivity of 0.86 and a specificity of 0.28. In the control group, 63% who met the OAHI clinical criteria for OSA, were above the PSQ OSA symptom cut-off score, with a sensitivity of 0.63 and a specificity of 0.42. All children above both the clinical criteria for OAHI and OSA symptom cut-off score had the most impaired quality of life and greater fatigue compared to those below both the clinical criteria for OAHI and the OSA symptom cut-off score.
Conclusion
Children who meet clinical criteria for OSA and also scored high on a parent reported screening tool for OSA symptoms had the most impaired quality of life and more fatigue. The PSQ has potential to identify children at risk for OSA.
Keywords: obstructive sleep apnea, juvenile idiopathic arthritis, health-related quality of life, fatigue, pediatric sleep questionnaire, patient-reported outcomes
INTRODUCTION
Juvenile idiopathic arthritis (JIA) is a common pediatric rheumatologic disease of unknown origin [1]. Sleep disorders, such as obstructive sleep apnea (OSA), [2–5] may play an important role in JIA-related impairments (e.g., pain, fatigue, quality of life). In a prior study [3] we showed that 40% of children with JIA had an elevated apnea hypopnea index (≥ 1.5 events/hr) consistent with the clinical criteria for OSA. The apnea hypopnea index derived from polysomnography is the gold standard for a diagnosis of OSA, [6] but polysomnography is labor-intensive and unsuitable for screening of OSA in routine pediatric clinic practice. Further, the apnea hypopnea index does not highly correlate with OSA-related impairments including behavior problems, daytime sleepiness, and quality of life [7–10]. Several studies in typically developing children provide evidence for the use of the sleep-related breathing disorders subscale of the Pediatric Sleep Questionnaire (PSQ) to screen for OSA [7,10]. This questionnaire is a parent-report measure of OSA symptoms with an empirically established cut-off score (> 0.33) and is used to identify children at risk for OSA. Recent studies show that the pediatric sleep questionnaire predicts OSA-related impairments (e.g., behavior, daytime sleepiness, quality of life) as well or better than the apnea hypopnea index by polysomnography [7, 10–13]. There is a need to establish a reliable screening tool that could be used in routine clinical practice to identify OSA risk in JIA. One aim of the present study was to examine the congruence between the obstructive apnea hypopnea index (OHAI) as measured by polysomnography and parent-reported OSA symptoms as measured by the pediatric sleep questionnaire (PSQ).
In typically developing children, OSA has been associated with increased fatigue and lower quality of life [2, 4, 5, 16–18]. Our previous study reported increased fatigue in JIA, [3,4] but we did not examine fatigue severity as a function of OSA nor did we measure quality of life. Fatigue and quality-of-life are key patient-reported outcomes in JIA. Co-morbid OSA in JIA may contribute to poorer patient-reported outcomes. Considering our prior findings of fatigue in JIA [3–4,19,20] and associations among OSA, fatigue, and quality of life previously reported in typically developing children [2, 4, 5, 16–18], a second aim of this study was to compare fatigue and quality-of-life in JIA and controls based on polysomnography OSA clinical criteria and the cut-off scores for parent report of OSA symptoms.
METHODS
Recruitment and Screening
The Institutional Review Board from a pediatric hospital in the Pacific Northwest approved this study. Written informed consent was obtained from the parents; assent was obtained from the children. Participants were recruited from October 2011 through December 2014. Children with JIA and their parents were recruited from a pediatric rheumatology clinic in a Pacific Northwest. A research coordinator screened the rheumatology clinic records for potential JIA participants. Subsequently, during a routine clinic visit, the research coordinator met with potentially eligible participants to confirm eligibility, discuss the study, and invite participation. Control children were recruited from the Eastern and Western Washington communities via flyers, media advertisements, and from friends and/or relatives of children with JIA. Interested control families contacted a member of the study team who screened participants via a telephone interview, confirmed eligibility, described the study, and invited participation. Each eligible child and a parent were scheduled for an overnight sleep study.
Upon arrival, a nurse practitioner performed a physical examination on the child. Parents completed a demographic survey, and the sleep-related breathing disorders subscale of Pediatric Sleep Questionnaire (PSQ); parents and children completed the Pediatric Quality of Life (PedsQL) Generic Core Scale, and the PedsQL Multidimensional Fatigue Scale.
Participants
A sample of 143 children, 6-to-11 years, with JIA (n=68) and controls (n=75) participated in the study. Both JIA and controls were excluded if they had a psychiatric diagnosis based on DSM-IV-TR criteria (e.g., ADHD), a diagnosed sleep disorder (e.g., OSA) by parent report or medical record, a prior tonsillectomy and/or adenoidectomy, a family history of narcolepsy, obesity, or Tanner stage ≥3. Children with asthma (n=8 [4 JIA]) and allergic rhinitis (n=18 [6 JIA]) were included in the study if they were well-controlled (e.g., required no medications) one month prior to the sleep study.
Of the 120 JIA families screened for the study, 52 declined to participate. Of those who declined, 53% had co-morbid conditions (e.g. Crohn’s disease, diabetes, anxiety) or had time constraints. Of the 140 control participants interested in the study, 65 declined to participate. Of those who declined, 75% had co-morbid conditions (e.g. ADHD, autism) or had time constraints.
Measures
Polysomnography Recordings and Scoring
Polysomnography was obtained for one night in a sleep research laboratory using the Somnologica Science data acquisition recording system (N7000, EMBLA, Broomfield, CO). A registered technologist, blind to group identity, scored the PSG recordings according to AASM standards [21, 22]. Obstructive apnea was defined as cessation of airflow with ongoing thoracoabdominal movement for at least 2 respiratory cycles. Hypopnea was defined as a >50% reduction in airflow with ongoing thoracoabdominal effort resulting in either an arousal or an oxyhemoglobin desaturation of at least 3% or more. The obstructive apnea hyponea index (OAHI) was defined as the number of obstructive apneas and hypopneas per hour of sleep. A board-certified pediatric sleep medicine physician reviewed each polysomnogram and verified OSA based on the number of OAHI per/hour of total sleep time. Clinical criteria for OSA was: 1) mild (OAHI= 1.5 to 4.99); 2) moderate (OAHI 5 to 9.99); and 3) severe (OAHI ≥10) [6]. Children who had an OAHI < 1.5 were classified as OSA negative (OSA −), and those with at least mild OSA (OAHI ≥ 1.5) were classified as OSA positive (OSA+).
Pediatric Sleep Questionnaire (PSQ)
Parents completed the 22-item PSQ sleep-related breathing disorders (SRBD) subscale for OSA symptoms (e.g. snoring, breathing pauses, inattention) [11]. A parent responded “yes=1”, “no=0”, and “don’t know = missing” to each item. A mean score is summed and divided by the number of items rated “yes”. The mean score can vary from 0.0 to 1.0, and cut-off score > 0.33 is considered positive and indicates high risk for OSA [12,23]. Children who had an OSA symptom (Sx) cut-off score of > 0.33 were classified as PSQ positive (PSQ +), and those with an OSA cut-off score < 0.33 were classified as PSQ negative (PSQ −). Reliability and validity of the PSQ is well established (10–12,21). In this study, the Cronbach’s alpha was 0.84.
Pediatric Quality of Life Inventory Generic Core
Quality of life was measured with the Pediatric Quality of Life Generic Core Scales (PedsQL™), a 23-item scale with four domains (physical, social, emotional, school function) [24,25]. A child and his/her parent independently rated each item on a 5-point Likert scale (0=never, 1=almost never, 2=sometimes, 3=often, 4=almost always). Responses for the child and for the parent were summed and divided by the number of items, to yield a Total Score and 2 subscale scores for Physical Health and Psychosocial Health (social, emotional, school function). Higher scores indicate better HRQoL. Reliability and validity are well established in pediatric populations with and without JIA (24–27). The Cronbach’s alpha for overall HRQoL was α = .89 for child report, and α = .92 for parent report.
Fatigue
Fatigue was measured with the PedsQL™ Multidimensional Fatigue Scale, an 18-item scale with 3 domains (general, sleep/rest, cognitive fatigue) [28]. A child and his/her parent independently rated each item on a 5-point Likert scale (0=never, 1=almost never, 2=sometimes, 3=often, 4=almost always). Responses for the child and for the parent were summed and divided by the number of items, to yield a Total Score and 3 subscale scores for general fatigue, sleep/rest fatigue, and cognitive fatigue. Higher scores indicate less fatigue. Reliability and validity are well established in children with and without JIA [28]. In this study, the Cronbach’s alpha for total fatigue was α = .88 for child report, and α = .92 for parent report.
Descriptive Clinical Characteristics
Recognized risk factors for OSA were coded as present (yes) or absent (no). These factors included: nasal turbinate hypertrophy (>50% of nasal cavity in one or both turbinates); high narrow arch palate; TMJ pain; TMJ crepitus (popping or crepitus during mouth opening); retrognathia (visual inspection of retrusion of the mandible); prematurity (<37 weeks of gestation), provider diagnosed asthma, allergies, or reflux; family history of OSA; male gender, and African-American race. Severity of tonsillar hypertrophy was measured by Brodsky score (0–4); higher score indicative of larger tonsils [29]. BMI was adjusted for age and sex according to Center for Disease Control criteria.
JIA disease duration was obtained from the clinic record and measured from the date the child was first diagnosed. At the time children enrolled in the study, a pediatric rheumatologist confirmed JIA disease subtype (e.g., oligioarthritis, polyarticular arthritis, systemic) and disease activity. Disease activity is rated according to active (physician global assessment [PGA] > 1, inflammation of one or more joints with swelling, limited range of motion, or tenderness), or inactive (PGA = 0, no active joints, no uveitis) [30]. The number of active joints was defined as the number of joints with active synovitis during the examination.
Statistical Analyses
Data were analyzed using SPSS for Windows version 17.0 (SPSS Inc, Chicago, Ill). To control for multiple comparisons, a Bonferroni correction was used. The first set of analyses addressed group differences between JIA and controls in demographics, clinical characteristics, risk factors, OAHI, OSA severity (mild, moderate, severe), PSQ total score, and PSQ OSA Sx score (PSQ − [< 0.33], PSQ + [> 0.33]). Data are expressed as mean ± standard deviation or percentage. Student’s t-test was used to test group differences of two continuous variables, and the Mann–Whitney U test was used for variables with non-normal distributions. Chi-square analysis tested group differences in proportions of categorical variables.
The second set of analyses examined the congruence (agreement and disagreement) between the clinical criteria for OSA (OAHI ≥ 1.5) and the PSQ OSA Sx cut-off score (> 0.33). To examine agreement and disagreement between the clinical criteria for OSA and PSQ OSA Sx cut-off score, a two by two contingency table was used to categorize the number of occurrences according to the following classifications: 1) True Positive (TP), coded when OSA+ (≥ 1.5) and PSQ+ (> 0.33); 2) True Negative (TN), coded when OSA− (< 1.5) and PSQ− (< 0.33); 3) False Negative (FN), coded when OSA+ (≥ 1.5) and PSQ− (< 0.33); and 4) False Positive (FP), coded as OSA− (< 1.5) and PSQ+ (> 0.33) (Table 1). JIA and control children were categorized into one of four groups: 1) OSA+/PSQ+ (n=35); 2) OSA−/PSQ− (n=37); 3) OSA−/PSQ+ (n=60); and 4) OSA+/PSQ− (n=8). This analysis provided estimates of sensitivity, defined as the proportion of children above the clinical criteria of OHAI (OSA +) and were also above the PSQ OSA Sx cutoff score (PSQ+) (TP), and specificity, defined as the proportion of children who did not meet the clinical criteria of OHAI (OSA−) and were also below the PSQ OSA Sx cut-off score (TN). Accuracy was defined as the proportion of participants classified as TP and TN divided by TP, TN, FP, and FN (Table 1).
Table 1.
Congruence between polysomnography OSA and PSQ OSA symptom cut-off score
| PSQ OSA Sx cut-off score | Polysomnography OAHI OSA+ | OSA − |
|---|---|---|
| PSQ+ | True Positive (TP) OSA+/PSQ+ |
False Positive (FP) OSA−/PSQ+ |
| PSQ − | False Negative (FN) OSA+/PSQ− |
True Negative (TN) OSA−/PSQ− |
Sensitivity: TP (TP + FP); specificity: TN (FP+ TN); accuracy: (TP + TN) (TP+FP+TN + FN)
PSQ− pediatric sleep questionnaire; OSA− obstructive sleep apnea; Sx- symptom; OAHI− obstructive apnea hypopnea index
The third set of analyses compared the mean scores in HRQoL (overall, physical heath, psychosocial health) and fatigue (total, general, sleep/rest, cognitive) for each group (e.g., OSA+/PSQ+; OSA−/PSQ−; OSA−/PSQ+; and OSA+/PSQ−). The OSA+/PSQ− group included 8 participants, which was too small to be included in the comparative analysis and was removed. The mean scores for HRQoL and fatigue were calculated for each of the remaining groups. Two separate one-way ANOVA’s (one for the child & one for the parent) were used to test for group differences in HRQoL and fatigue.
RESULTS
Demographic and clinical characteristics
Table 2 shows the demographic and clinical characteristics for JIA and controls. There were no statistically significant group differences for any of the variables after correcting for multiple comparisons.
Table 2.
Demographics and clinical characteristics
| JIA (n=68) | Controls (n=75) | p-value | |
|---|---|---|---|
| Age, years, mean (SD) | 8.5 ± 1.8 | 8.9 ± 1.6 | 0.21 |
| Sex, n (%) | |||
| Girls | 38 (55.9%) | 42 (56%) | 0.86 |
| Race, n (%) | 0.23 | ||
| White | 48 (70.6%) | 54 (72%) | |
| Mixed | 11 (16.2%) | 10 (13.3%) | |
| Asian American | 4 (1.5%) | 8 (10.7%) | |
| African American | 0 (0 %) | 2 (2.7%) | |
| Hispanic/Latino | 5 (7.4%) | 1 (1.3%) | |
| Participating parent, n (%) | 0.38 | ||
| Mothers | 63 (92.6%) | 66 (88%) | |
| Fathers | 5 (7.4%) | 9 (12%) | |
| Disease category, n (%) | |||
| Oligoarticular | 19 (27.9%) | N/A | |
| Extended oligoarticular | 13 (19.1%) | ||
| Polyarticular RF negative | 22 (32.4%) | ||
| Polyarticular RF positive | 1 (1.5%) | ||
| Systemic | 7 (10.3%) | ||
| Enthesitis-related | 4 (5.9%) | ||
| Psoriatis | 2 (2.9%) | ||
| Disease Activity, n (%) | |||
| Active | 27 (39.7%) | NA | |
| Inactive | 41 (60.3%) | NA | |
| Disease duration, months, mean (SD) | 37.8 + 28 | NA | |
| Physician Global Assessment of disease activity, (0–10) | 0.88 + 1.4 | NA | |
| Active Joint Count, (#) | 1.57 + 4.5 | NA | |
| OSA Risk factors | |||
| BMI | 17.0 ± 2.8 | 17.3 ± 2.4 | 0.46 |
| Oral & Facial Features | |||
| Tonsil size ( > 3) | 7 (10.3%) | 9 (12%) | 0.68 |
| Nasal Turbinate hypertrophy, n (% yes) | 31 (45.6%) | 39 (52%) | 0.63 |
| High arch narrow palate, n (% yes) | 7 (10.3%) | 5 (6.7%) | 0.44 |
| Retrognathia, n (% yes) | 6 (8.8%) | 2 (2.7%) | 0.09 |
| TMJ pain, n (% yes) | 4 (5.9%) | 0 (0%) | 0.03a |
| TMJ crepitus, n (% yes) | 3 (4.4%) | 0 (0%) | 0.07 |
| History of: | |||
| Prematurity, n (% yes) | 9 (13.2%) | 5 (20%) | 0.27 |
| Allergies, n (% yes) | 6 (8.8%) | 12 (16%) | 0.19 |
| Asthma, n (% yes) | 4 (5.9%) | 4 (5.3%) | 0.89 |
| GERD, n (% yes) | 3 (4.4%) | 0 (0%) | 0.07 |
JIA- juvenile idiopathic arthritis; OSA- obstructive sleep apnea.
Data are mean ± SD or n (%).
Comparison no longer significant after applying α correction.
OAHI and PSQ OSA symptom cut-off score
Table 3 shows OAHI, OSA severity, PSQ total score, and PSQ OSA Sx cut-off scores for JIA and controls. OAHI (p <0.01) was significantly higher in JIA compared to controls and was four times more prevalent in JIA (χ2 = 28.2, p < 0.001).
Table 3.
OSA severity and PSQ OSA symptom cut-off score.
| JIA (n=68) | Controls (n=75) | p value | |
|---|---|---|---|
| Obstructive apnea hypopnea index (/h TST) | 2.9 ± 3.9 | 1.3 ± 1.2 | 0.003 |
| OSA severity, n (%) | 0.001 | ||
| Mild (OAHI ≥ 1.5 to 4.99) | 25 (36.8%) | 7 (9.3%) | |
| Moderate (OAHI≥ 5 to 9.99) | 7 (10.3%) | 1 (1.3%) | |
| Severe (OAHI ≥ 10/h) | 3 (4.4%) | 0 (0%) | |
| Total | 35 (51%) | 8 (10%) | |
| PSQ score, mean (SD) | 1.05 ± .82 | 0.66 ± .75 | 0.004 |
| OSA Sx cut-off score | 0.007 | ||
| PSQ − (≤ 0.33), n (%) | 16 (25.2%) | 30 (40%) | |
| PSQ + (≤ 0.33), n (%) | 52 (76.4%) | 42 (56%) |
JIA- juvenile idiopathic arthritis; OSA- obstructive sleep apnea;/h TST- per hour total sleep time; OAHI- obstructive apnea hypopnea index; OSA symptom (sx) score from the pediatric sleep questionnaire cut-off score, > 0.33.
Data are mean ± SD or n (%).
Congruency between OHAI and PSQ OSA symptom cut-off scores
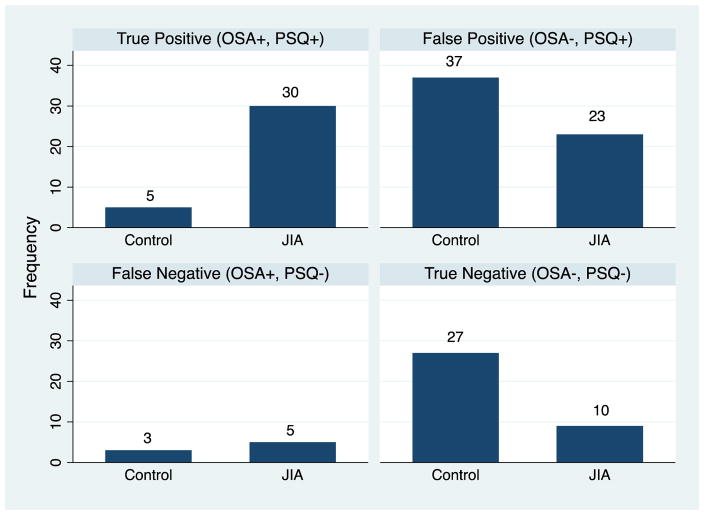
Figure 1 shows the number of participants in the JIA and control groups who were classified by the clinical criteria for OAHI and by PSQ OSA Sx cut-off score into one of four groups (e.g. TP, TN, FP, FN). In JIA 86% had an OAHI ≥ 1.5 and a PSQ OSA Sx cut-off score > 0.33 yielding a sensitivity of 0.86 and a specificity of 0.28. In JIA, 58.2% had an OAHI above or below 1.5 and also had a corresponding PSQ OSA Sx cut-off score above or below 0.33. In controls, 63% had an OAHI ≥ 1.5 (n=5) and had a PSQ OSA Sx cut-off score > 0.33, yielding a sensitivity of 0.63 and a specificity of 0.42. In controls, 44.4% had an OAHI above or below 1.5 and also had a corresponding PSQ OSA Sx cut-off score above or below 0.33. The false positive rates (e.g., OSA−/PSQ+) was 27% for JIA and 73% for controls.
Figure 1. Congruence between OAHI and PSQ OSA symptom cut-off score in JIA and controls.
OSA- obstructive sleep apnea confirmed by polysomnography obstructive apnea hypopnea index ≥ 1.5; PSQ- pediatric sleep questionnaire OSA symptom score cut-off score (> 0.33); JIA- juvenile idiopathic arthritis; Data include the number JIA and control children (n=140).
HRQoL: OAHI and PSQ OSA symptom cut-off scores
Table 4 shows the mean and the mean difference scores both for the child and parent report of overall HRQoL, and physical and psychosocial health subscales for each of the OSA/PSQ groups (OSA+/PSQ+; OSA−/PSQ−; OSA−/PSQ+). ANOVAs both for the child and the parent report revealed significant differences among the 3 groups for overall HRQoL (child, F = 4.7, p < 0.004; parent F = 5.6, p < 0.001), physical health (child F = 4.6, p < 0.005; parent F = 6.2, p <0.001), and psychosocial health (child F = 3.8, p < 0.01; F = 4.6, p < 0.004). Post hoc comparisons showed significantly lower mean scores both for the child and the parent report for all three HRQoL measures in the OSA+/PSQ+ group compared to the OSA−/PSQ+ group.
Table 4.
PedsQL Generic Core Scales: OAHI and PSQ OSA symptom cut-off score
| OSA+/PSQ + (n= 35) Mean (SD) |
OSA−/PSQ− (n=37) Mean (SD) |
OSA−/PSQ+ (n=60) Mean (SD) |
OSA/PSQ− vs. OSA+/PSQ+ Comparison Difference (95%CI) |
OSA+/PSQ+ vs. OSA−/PSQ+ Comparison Difference (95%CI) |
OSA/PSQ− vs. OSA−/PSQ+ Comparison Difference (95%CI) |
|
|---|---|---|---|---|---|---|
| Child report | ||||||
| Overall HRQoL | 74.7 (15.3) | 84.9 (10.1) | 83.4 (9.3) | 10.2 (1.7, 18.7)** | −8.7 (−16.4, -.90)* | |
| Physical health | 77.0 (16.2) | 87.9 (10.8) | 87.9 (12.3) | 10.9 (1.1, 20.7)* | −11.0 (−20.7, −1.7)** | |
| Psychosocial health | 73.9 (15.7) | 83.8 (10.5) | 81.8 (10.8) | 10.2 (.83,19.2)* | ||
| Parent report | ||||||
| Overall HRQoL | 75.5 (13.2) | 87.3 (10.7) | 78.0 (15.2) | 11.9 (3.3, 20.5)*** | 9.3 (1.6, 17.1)** | |
| Physical health | 75.5 (17.9) | 90.9 (10.1) | 82.1 (19.7) | 15.3 (4.8, 25.8)*** | ||
| Psychosocial health | 75.4 (13.3) | 86.2 (11.8) | 76.6 (15.1) | 10.7 (1.9, 19.6)** | 9.5 (1.5,17.5)** | |
OSA- obstructive sleep apnea confirmed by polysomnography obstructive apnea hypopnea index ≥1.5; PSQ - pediatric sleep questionnaire OSA symptom cut-off score > 0.33; HRQoL- health related quality of life.
Higher scores indicate better HRQoL.
Data shows statistically significant mean difference scores and 95% confidence intervals for comparisons of the OSA+/PSQ+, OSA−/PSQ−, and OSA−/PSQ+ groups.
p < 0.05,
p < .01,
p < .005
Fatigue: OAHI and PSQ OSA symptom cut-off scores
Table 5 shows the mean and the mean difference scores both for the child and the parent report for total fatigue, and the general, sleep/rest, and cognitive fatigue subscale scores for each of three groups (OSA+/PSQ+; OSA−/PSQ−; OSA−/PSQ+). ANOVA for child report showed significant group differences for mean total fatigue (F = 2.8, p < 0.04) and the sleep/rest (F = 3.5, p < 0.02) subscale fatigue score. ANOVA for parent report of fatigue showed significant group differences for total fatigue (F =8.3, p < 0.001), and the general (F = 4.8, p < 0.003), and sleep/rest (F = 10.2, p < 0.001) fatigue subscale scores. Post hoc comparisons showed significantly lower mean scores for overall (p < 0.001), general fatigue (p < 0.009), sleep rest (p < 0.001), and cognitive fatigue (p < 0.04) in the OSA+/PSQ+ group compared to the OSA−/PSQ− group.
Table 5.
PedsQL multidimensional fatigue: OAHI and PSQ OSA symptom cut-off score
| OSA+/PSQ+ (n= 35) Mean (SD) |
OSA−/PSQ− (n=37) Mean (SD) |
OSA−/PSQ + (n=60) Mean (SD) |
OSA−/PSQ− vs. OSA+/PSQ+ Comparison Difference (95%CI) |
OSA−/PSQ− vs. OSA−/PSQ+ Comparison Difference (95%CI) |
|
|---|---|---|---|---|---|
| Child report | |||||
| Total fatigue | 68.8 (17.3) | 76.9 (14.1) | 74.7 (14.6) | ||
| General fatigue | 68.4 (17.8) | 74.1 (15.8) | 72.4 (13.6) | ||
| Sleep/rest fatigue | 66.4 (16.0) | 75.2 (18.5) | 71.4 (17.4) | ||
| Cognitive fatigue | 65.4 (24.5) | 74.1 (19.1) | 72.6 (21.5) | ||
| Parent report | |||||
| Total fatigue | 71.9 (16.2) | 86.1 (10.9) | 74.2 (16.0) | 14.2 (5.0, 23.5)*** | 11.9 (3.7, 20.2)*** |
| General fatigue | 69.8 (21.2) | 83.2 (13.3) | 75.3 (17.9) | 13.5 (2.3, 24.6)** | |
| Sleep/rest fatigue | 76.7 (17.1) | 92.5 (9.4) | 75.4 (19.7) | 15.8 (5.5 26.1)*** | 17.1 (7.9, 26.3)*** |
| Cognitive fatigue | 69.2 (19.5) | 82.7 (18.8) | 71.8 (22.6) | 13.5 (.55,26.4)* | |
OSA- obstructive sleep apnea confirmed by polysomnography obstructive apnea hypopnea index ≥1.5; PSQ - pediatric sleep questionnaire OSA symptom cut-off score > 0.33.
Higher scores indicate less fatigue.
Data shows statistically significant mean difference scores and 95% confidence intervals for comparisons of the OSA+/PSQ+, OSA−/PSQ−, and OSA−/PSQ+ groups.
p < 0.05,
p < .01,
p < 0.001.
DISCUSSION
In JIA, cut-off scores on the parent-report Pediatric Sleep Questionnaire showed good congruence with high sensitivity for OSA as defined by clinical criteria derived from polysomnography. In JIA 86% who met OAHI clinical criteria for OSA also had scores above the cut-off for OSA symptoms on the PSQ. Thus, in our sample, the PSQ served as an excellent screening tool for children with JIA at considerable risk for OSA.
Despite the high prevalence of OSA in JIA, none of the children in our studies had been referred to a sleep clinic for an OSA evaluation, suggesting the need for more effective in-clinic screening procedures. The presence of OSA is particularly noteworthy given that fatigue and HRQoL were most impaired in JIA children who met both the clinical criteria for OSA and had a positive parent-reported OSA symptom score. These findings provide evidence that OSA can negatively impact key patient-reported outcomes including fatigue and HRQoL in JIA.
Congruence between OAHI and PSQ OSA symptom cut-off score
The PSQ performed well in JIA, but this was not observed in the control group. The moderate to high sensitivity (86%) in JIA might be attributed to manifestations of symptoms such as TMJ pain and crepitus, and/or from craniofacial structures such as retrognathia that were not present in controls. In the current study, TMJ pain, crepitus, and retrognathia were observed in JIA, but not in controls, and these features are risk factors for OSA [31]. Arthritis of the TMJ involves inflammation, decreased mouth opening, deviation on opening, and crepitus that may result in reduced mandibular growth, negatively impact breathing, and place JIA children at increased risk for OSA [31]. However, risk factors associated with OSA in JIA have not been well documented. Longitudinal studies that examine JIA children shortly after diagnosis are needed to better understand when OSA emerges as children transition across developmental stages. Such knowledge would provide guidance for early detection of OSA in JIA.
Our findings also show high false positive rates (OSA−/PSQ+) for both JIA and control children. This finding may be attributed to the binary (yes or no) responses on the sleep-related breathing disorder subscale questions versus a scale that would ascertain the frequency of OSA symptoms (e.g. 5–7 days/wk vs. 1–2 days/wk). Further, the sleep-related breathing subscale also includes questions about daytime behaviors (e.g., inattention, hyperactivity) which may or may not be associated with OSA symptoms, and could have contributed to the high false positive rates found in this study. Additionally, a high proportion of children in both groups had nasal turbinate hypertrophy, which could contribute to ‘snoring’ or symptom reports, but not necessarily lead to a hyponea or apnea observed on polysomnography.
There is currently no screening tool used in rheumatology practice to identify risk for OSA in JIA. It is impractical to screen all children using polysomnography, which is costly and not readily available. Although, the false positive rates were high, scores from the PSQ showed moderate to good sensitivity for OSA in JIA. Integrating the PSQ into pediatric rheumatology clinics could provide a cost effective screening tool for OSA that may identify patients at risk and prompt referral to a pediatric sleep disorders center for further evaluation. Further research is needed on the utility of the PSQ as a clinical screening tool for OSA in JIA.
HRQoL
Overall HRQoL, physical health, and psychosocial health both in child and parent reports were the most impaired in children who met the clinical criteria for OSA and had a positive parent-reported OSA symptom score. Given that (35/68) 51% of the entire JIA sample had OSA, this finding is clinically important for several reasons. First, it highlights that the impact of OSA extends beyond sleep to affect children’s physical, emotional, and overall wellbeing. Second, in comparison to other studies of HRQoL in JIA with varying disease severity, [2, 5, 20, 32,33] children in the OSA+/PSQ+ group perceived their physical, psychosocial, and overall HRQoL much worse than children in the other groups. Third, children in the OSA+/PSQ+ group, child and parent report were consistent, such that parents also perceived their child’s overall well-being more impaired than the other groups. Our findings suggest children with JIA may be quite vulnerable to adverse effects of OSA, and this could negatively impact disease management and important patient reported outcomes, like HRQoL. Additional research is needed because sleep disorders are often overlooked and attributed to the underlying chronic condition, when in fact, these children may have a co-morbid, treatable sleep disorder.
Fatigue
Fatigue scores were highest in the OSA+/PSQ+ group in comparison to the other groups. This finding suggests that untreated OSA not only impacts quality of life but also fatigue. Similar to previous studies, [2, 36] fatigue was related to DMARD therapy and active disease (data not shown), but these variables were not associated with a higher prevalence of OSA. To our knowledge, this is the first study to examine physiologic measures of OSA in relation to multidimensional fatigue in JIA, and we have no published studies for comparison. Fatigue is a complex multidimensional construct that reflects a number of psychosocial (e.g., stress) and environmental factors, [37,38] which were not examined in this study, and may contribute to our findings.
Limitations
Limitations to this study deserve comment. First, although we excluded children with prior sleep disorders, we cannot account for parent bias as some parents may have suspected their child had a sleep problem and were more likely to agree to participate in the study. Second, we had a small number of control children who screened positive for OSA via the gold standard polysomnography, but not by parent report, and this limited the comparisons we could conduct. Third, we did not include measures of physical and psychosocial function that may contribute to our HRQoL and fatigue findings. Despite these limitations our findings provide new knowledge about OSA in an understudied pediatric population and its impact on important patient reported health outcomes.
Conclusions
In conclusion, study results showed the PSQ was a sensitive screening tool for OSA in JIA children (Aim 1). Further, both fatigue and HRQoL, key patient-reported outcomes in the clinical management of JIA, were most impaired in JIA children who met both the clinical criteria for OSA and screened positive for parent report of OSA symptoms (Aim 2). With the current standard of care, most JIA children with OSA are unrecognized and untreated, which is an additional co-morbidity that may contribute to poor disease management and patient-reported outcomes. Implementing the PSQ screening tool could promote timely diagnosis and treatment of OSA.
Acknowledgments
Funding Support: The study was supported by the National Institute of Nursing Research, NIH, (# RO1NR012734) and the Center for Research on Management of Sleep Disturbances, National Institute of Nursing Research (# P30NR011400).
Additional contributions: The authors thank the children and families who helped with this research. We thank Ching Hung and Lucas Reichley, Research Coordinator in the Rheumatology Clinic for recruiting the participants. We thank Ernie Tolentino, Laboratory Manager, and the sleep laboratory staff, James Rothermel, Alexandra Lexie Miller, Casey Guilland, Philip Celerian, Alexa Celerian, Megha Santhosh, Susie Chen, Anna Duong, Willada Locha, Deenpret Dhaliwal for data collection and processing.
Footnotes
Author contributions: T.M. Ward had full access to all the data in the study and takes responsibility for the integrity of the data, and T.M. Ward and K.C. Pike take responsibility for the accuracy of the data analysis. All authors made substantial contributions to the study and this manuscript. None were compensated for the manuscript preparation.
Conflict of Interest Disclosures: None of the other authors report any conflicts of interest.
Role of the Funding/Sponsor: The funding sources had no role in the designs and conduct of the study; collection, management, analysis, and interpretation of the data; and preparation, review, and approval of the manuscript, and decision to submit the manuscript for publication.
References
- 1.Ravelli A, Martini A. Juvenile idiopathic arthritis. Lancet. 2007;369(9563):767–78. doi: 10.1016/S0140-6736(07)60363-8. [DOI] [PubMed] [Google Scholar]
- 2.Butbul Aviel Y, Stremler R, Benseler SM, Cameron B, Laxer RM, Ota S, et al. Sleep and fatigue and the relationship to pain, disease activity and quality of life in juvenile idiopathic arthritis and juvenile dermatomyositis. (2011) Rheumatology (Oxford) 50(11):2051–60. doi: 10.1093/rheumatology/ker256. [DOI] [PubMed] [Google Scholar]
- 3.Ward TM, Archbold K, Lentz M, Ringold S, Wallace CA, Landis CA. Sleep disturbance, daytime sleepiness, and neurocognitive performance in children with juvenile idiopathic arthritis. Sleep. 2010;33:252–259. doi: 10.1093/sleep/33.2.252. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Ward TM, Brandt P, Archbold K, Lentz M, Ringold S, Wallace CA, Landis CA. Polysomnography and self-reported sleep, pain, fatigue, and anxiety in children with active and inactive juvenile rheumatoid arthritis. J Pediatr Psychol. 2008;33(3):232–41. doi: 10.1093/jpepsy/jsm121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Shyen S, Amine B, Rostom S, Badri EL, Ezzahri DM, Mawani N, et al. Sleep and its relationship to pain, dysfunction, and disease activity in juvenile idiopathic arthritis. Clin Rheumatol. 2014;33(10):1425–31. doi: 10.1007/s10067-013-2409-x. [DOI] [PubMed] [Google Scholar]
- 6.Marcus CL, Brooks LJ, Draper KA, Gozal D, Halbower AC, Jones J, et al. Diagnosis and management of childhood obstructive sleep apnea syndrome. Pediatrics. 2012;130(3):e714–55. doi: 10.1542/peds.2012-1672. [DOI] [PubMed] [Google Scholar]
- 7.Mitchell RB, Garetz S, Moore RH, Rosen CL, Marcus CL, Katz ES, et al. The use of clinical parameters to predict obstructive sleep apnea syndrome severity in children: the Childhood Adenotonsillectomy (CHAT) study randomized clinical trial. JAMA Otolaryngol Head Neck Surg. 2015;141(2):130–6. doi: 10.1001/jamaoto.2014.3049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Chervin RD, Ruzicka DL, Giordani BJ, Weatherly RA, Dillon JE, Hodges EK, Marcus CL, Guire KE. Sleep-disordered breathing, behavior, and cognition in children before and after adenotonsillectomy. Pediatrics. 2006;117(4):e769–78. doi: 10.1542/peds.2005-1837. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Emancipator JL, Storfer-Isser A, Taylor HG, Rosen CL, Kirchner HL, Johnson NL, et al. Variation of cognition and achievement with sleep-disordered breathing in full-term and preterm children. Arch Pediatr Adolesc Med. 2006;160(2):203–10. doi: 10.1001/archpedi.160.2.203. [DOI] [PubMed] [Google Scholar]
- 10.Rosen CL, Wang R, Taylor HG, Marcus CL, Katz ES, Paruthi S, et al. Utility of symptoms to predict treatment outcomes in obstructive sleep apnea syndrome. Pediatrics. 2015;135(3):e662–71. doi: 10.1542/peds.2014-3099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Chervin RD, Hedger K, Dillon JE, Pituch KJ. Pediatric sleep questionnaire (PSQ): validity and reliability of scales for sleep-disordered breathing, snoring, sleepiness, and behavioral problems. Sleep Med. 2000;1(1):21–32. doi: 10.1016/s1389-9457(99)00009-x. [DOI] [PubMed] [Google Scholar]
- 12.Chervin RD, Weatherly RA, Garetz SL, Ruzicka DL, Giodani BJ, Hodges BJ, et al. Pediatric sleep questionnaire: prediction of sleep apnea and outcomes. Arch Otolaryngol Head Neck Surg. 2007;133(3):216–22. doi: 10.1001/archotol.133.3.216. [DOI] [PubMed] [Google Scholar]
- 13.Marcus CL, Moore RH, Rosen CL, Giordani B, Garetz SL, Taylor HG, Mitchell RB, Amin R, Katz ES, Arens R, Paruthi S, Muzumdar H, Gozal D, Thomas NH, Ware J, Beebe D, Snyder K, Elden L, Sprecher RC, Willging P, Jones D, Bent JP, Hoban T, Chervin RD, Ellenberg SS, Redline S. Childhood Adenotonsillectomy Trial (CHAT). A randomized trial of adenotonsillectomy for childhood sleep apnea. N Engl J Med. 2013;368:2366–76. doi: 10.1056/NEJMoa1215881. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Lumeng JC, Chervin RD. Epidemiology of pediatric obstructive sleep apnea. Proc Am Thorac Soc. 2008 Feb 15;5(2):242–252. doi: 10.1513/pats.200708-135MG. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Tarasiuk A, Reuveni H. The economic impact of obstructive sleep apnea. Curr Opin Pulm Med. 2013 Nov;19(6):639–44. doi: 10.1097/MCP.0b013e3283659e1e. Review. [DOI] [PubMed] [Google Scholar]
- 16.Crabtree VM, Varni JW, Gozal D. Health-related quality of life and depressive symptoms in children with suspected sleep-disordered breathing. Sleep. 2004;27(6):1131–8. doi: 10.1093/sleep/27.6.1131. [DOI] [PubMed] [Google Scholar]
- 17.Baldassari CM, Mitchell RB, Schubert C, Rudnick EF. Pediatric obstructive sleep apnea and quality of life: a meta-analysis. Otolaryngol Head Neck Surg. 2008;138(3):265–273. doi: 10.1016/j.otohns.2007.11.003. [DOI] [PubMed] [Google Scholar]
- 18.Rosen CL, Palermo TM, Larkin EK, Redline S. Health-related quality of life and sleep-disordered breathing in children. Sleep. 2002;25(6):657–66. [PubMed] [Google Scholar]
- 19.Ringold S, Ward TM, Wallace CA. Disease activity and fatigue in juvenile idiopathic arthritis. Arthritis Care Res (Hoboken) 2013;5:391–7. doi: 10.1002/acr.21801. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Ringold S, Wallace CA, Rivara FP. Health-related quality of life, physical function, fatigue, and disease activity in children with established polyarticular juvenile idiopathic arthritis. J Rheumatol. 2009;36:1330–6. doi: 10.3899/jrheum.081028. [DOI] [PubMed] [Google Scholar]
- 21.Iber C, Ancoli-Israel S, Chesson AL, Jr, Quan SF American Academy of Sleep Medicine. The AASM manual for the scoring of sleep and associated events: rules, terminology and technical specifications. 1. Westchester, IL: American Academy of Sleep Medicine; 2007. [Google Scholar]
- 22.Grigg-Damberger M, Gozal D, Marcus CL, Quan SF, Rosen CL, Chervin RD, et al. The visual scoring of sleep and arousal in infants and children. J Clin Sleep Med. 2007;3(2):201–40. [PubMed] [Google Scholar]
- 23.Chervin RD, Weatherly RA, Ruzicka DL, Burns JW, Giordani BJ, Dillon JE, et al. Subjective sleepiness and polysomnographic correlates in children scheduled for adenotonsillectomy vs other surgical care. Sleep. 2006;29(4):495–503. [PMC free article] [PubMed] [Google Scholar]
- 24.Varni JW, Seid M, Smith Knight T, Burwinkle T, Brown J, Szer IS. The PedsQL in pediatric rheumatology: reliability, validity, and responsiveness of the Pediatric Quality of Life Inventory Generic Core Scales and Rheumatology Module. Arthritis Rheum. 2002;46(3):714–25. doi: 10.1002/art.10095. [DOI] [PubMed] [Google Scholar]
- 25.Varni JW, Seid M, Kurtin PS. PedsQL™ 4.0: Reliability and validity of the Pediatric Quality of Life Inventory™ Version 4.0 generic core scales in healthy and patient populations. Med Care. 2001;39:800–12. doi: 10.1097/00005650-200108000-00006. [DOI] [PubMed] [Google Scholar]
- 26.Varni JW, Globe DR, Gandra SR, Harrison DJ, Hooper M, Baumgartner S. Health-related quality of life of pediatric patients with moderate to severe plaque psoriasis: comparisons to four common chronic diseases. Eur J Pediatr. 2012 Mar;171(3):485–92. doi: 10.1007/s00431-011-1587-2. [DOI] [PubMed] [Google Scholar]
- 27.Haverman L, Grootenhuis MA, van den Berg JM, van Veenendaal M, Dolman KM, Swart JF, Kuijpers TW, van Rossum MA. Predictors of health-related quality of life in children and adolescents with juvenile idiopathic arthritis: results from a Web-based survey. Arthritis Care Res (Hoboken) 2012 May;64(5):694–703. doi: 10.1002/acr.21609. [DOI] [PubMed] [Google Scholar]
- 28.Varni JW, Burwinkle TM, Szer IS. The PedsQL Multidimensional Fatigue Scale in pediatric rheumatology: reliability and validity. J Rheumatol. 2004;31(12):2494–500. [PubMed] [Google Scholar]
- 29.Brodsky L, Moore l, Stanievich JF. Stanievich, A comparison of tonsillar size and oropharyngeal dimensions in children with obstructive adenotonsillar hypertrophy. Int J Pediatr Otorhinolaryngol. 1987;13(2):149–56. doi: 10.1016/0165-5876(87)90091-7. [DOI] [PubMed] [Google Scholar]
- 30.Wallace CA, Giannini EH, Huang B, Itert L, Ruperto N. American College of Rheumatology provisional criteria for defining clinical inactive disease in select categories of juvenile idiopathic arthritis. Arthritis Care Res (Hoboken) 2011;63(7):929–36. doi: 10.1002/acr.20497. [DOI] [PubMed] [Google Scholar]
- 31.Abad VC, Sarinas PS, Guilleminault C. Sleep and rheumatologic disorders. Sleep Med Rev. 2008;12:211–228. doi: 10.1016/j.smrv.2007.09.001. [DOI] [PubMed] [Google Scholar]
- 32.Seid M, Huang B, Niehaus S, Brunner HI, Lovell DJ. Determinants of health-related quality of life in children newly diagnosed with juvenile idiopathic arthritis. Arthritis Care Res (Hoboken) 2014;66:263–9. doi: 10.1002/acr.22117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Oliveira S, Ravelli A, Pistorio A, Castell E, Malattia C, Prieur AM, et al. The Pediatric Rheumatology International Trials Organization (PRINTO) Proxy-reported health-related quality of life of patients with juvenile idiopathic arthritis: the Pediatric Rheumatology International Trials Organization multinational quality of life cohort study. Arthritis Rheum. 2007;57:35–43. doi: 10.1002/art.22473. [DOI] [PubMed] [Google Scholar]
- 36.Husted JA, Tom BD, Schentag CT, Farewell VT, Gladman DD. Occurrence and correlates of fatigue in psoriatic arthritis. Ann Rheum Dis. 2009;68:1553–8. doi: 10.1136/ard.2008.098202. [DOI] [PubMed] [Google Scholar]
- 37.Bromberg MH, Gil KM, Schanberg LE. Daily sleep quality and mood as predictors of pain in children with juvenile polyarticular arthritis. Health Psychol. 2012;31(2):202–9. doi: 10.1037/a0025075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Schanberg LE, Gil KM, Anthony KK, Yow E, Rochon J. Pain, stiffness, and fatigue in juvenile polyarticular arthritis: contemporaneous stressful events and mood as predictors. Arthritis Rheum. 2005;52:1196–204. doi: 10.1002/art.20952. [DOI] [PubMed] [Google Scholar]